

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phospholipase C (Bacillus cereus) | BDBM50332115 (3-(N-(benzyloxy)-2-(tert-butyloxycarbonylamino)dod...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Advance Chemistry of Catalonia (IQAC-CSIC) Curated by ChEMBL | Assay Description Mixed-type inhibition of Bacillus cereus phosphatidylcholine preferred phospholipase C by Dixon plot analysis | Bioorg Med Chem 18: 8549-55 (2010) Article DOI: 10.1016/j.bmc.2010.10.031 BindingDB Entry DOI: 10.7270/Q2VH5P32 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phospholipase C (Bacillus cereus) | BDBM50332109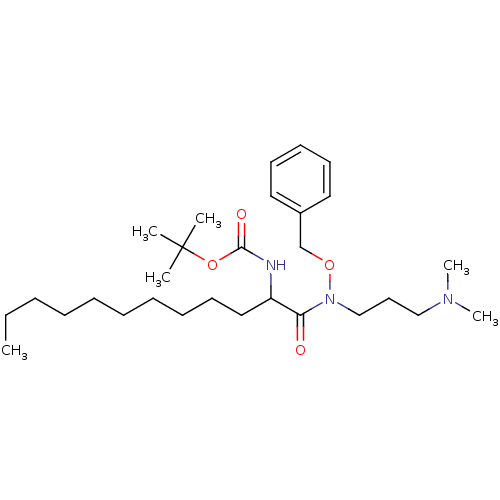 (CHEMBL1287861 | Tert-butyl 1-(benzyloxy[3-(dimethy...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Advance Chemistry of Catalonia (IQAC-CSIC) Curated by ChEMBL | Assay Description Non-competitive inhibition of Bacillus cereus phosphatidylcholine preferred phospholipase C by Dixon plot analysis | Bioorg Med Chem 18: 8549-55 (2010) Article DOI: 10.1016/j.bmc.2010.10.031 BindingDB Entry DOI: 10.7270/Q2VH5P32 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phospholipase C (Bacillus cereus) | BDBM50332105 (1-(hydroxy(2-(trimethylammonio)ethyl)amino)-1-oxod...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Advance Chemistry of Catalonia (IQAC-CSIC) Curated by ChEMBL | Assay Description Uncompetitive inhibition of Bacillus cereus phosphatidylcholine preferred phospholipase C by Dixon plot analysis | Bioorg Med Chem 18: 8549-55 (2010) Article DOI: 10.1016/j.bmc.2010.10.031 BindingDB Entry DOI: 10.7270/Q2VH5P32 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Phospholipase C (Bacillus cereus) | BDBM50332119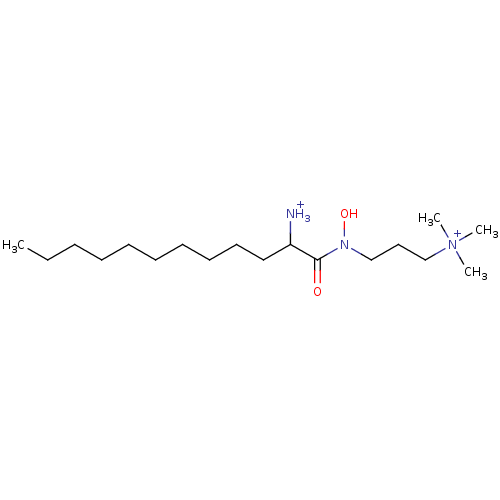 (1-(hydroxy[3-(trimethylammonio)propyl]amino)-1-oxo...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Advance Chemistry of Catalonia (IQAC-CSIC) Curated by ChEMBL | Assay Description Mixed-type inhibition of Bacillus cereus phosphatidylcholine preferred phospholipase C by Dixon plot analysis | Bioorg Med Chem 18: 8549-55 (2010) Article DOI: 10.1016/j.bmc.2010.10.031 BindingDB Entry DOI: 10.7270/Q2VH5P32 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||