 Found 502 hits of ic50 data for polymerid = 5163,9712
Found 502 hits of ic50 data for polymerid = 5163,9712 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50147093
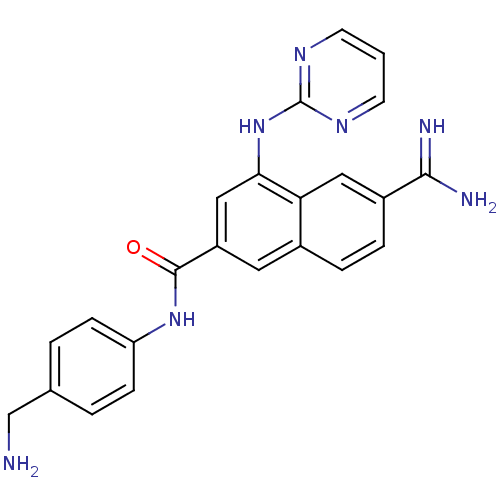
(6-Carbamimidoyl-4-(pyrimidin-2-ylamino)-naphthalen...)Show SMILES NCc1ccc(NC(=O)c2cc(Nc3ncccn3)c3cc(ccc3c2)C(N)=N)cc1 Show InChI InChI=1S/C23H21N7O/c24-13-14-2-6-18(7-3-14)29-22(31)17-10-15-4-5-16(21(25)26)11-19(15)20(12-17)30-23-27-8-1-9-28-23/h1-12H,13,24H2,(H3,25,26)(H,29,31)(H,27,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 0.620 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wollongong
Curated by ChEMBL
| Assay Description
Inhibition of uPA (unknown origin) |
J Med Chem 61: 8299-8320 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00838
BindingDB Entry DOI: 10.7270/Q2QF8WH4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194745
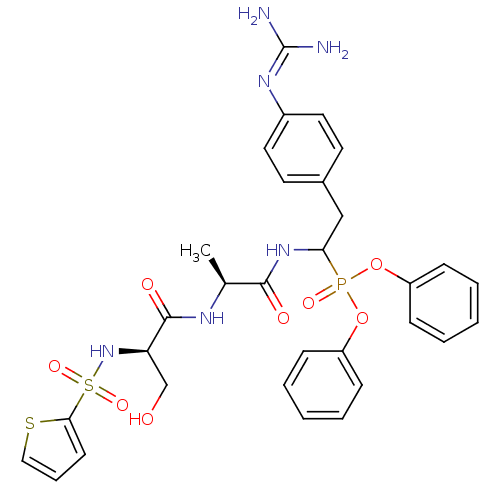
(CHEMBL385900 | diphenyl 1-[(N-2-thiophenesulfonyl-...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1cccs1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C31H35N6O8PS2/c1-21(34-30(40)26(20-38)37-48(42,43)28-13-8-18-47-28)29(39)36-27(19-22-14-16-23(17-15-22)35-31(32)33)46(41,44-24-9-4-2-5-10-24)45-25-11-6-3-7-12-25/h2-18,21,26-27,37-38H,19-20H2,1H3,(H,34,40)(H,36,39)(H4,32,33,35)/t21-,26+,27?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194741
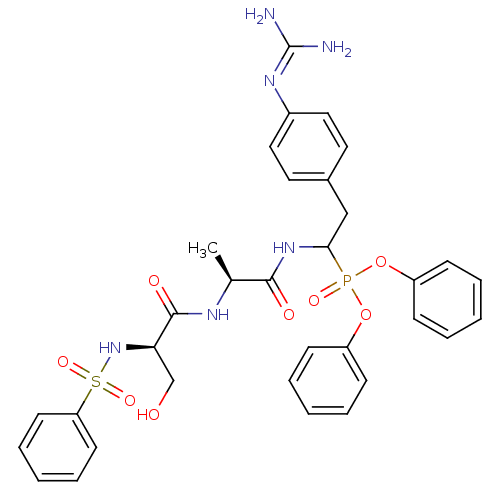
(CHEMBL385158 | diphenyl 1-[(N-benzenesulfonyl-D-se...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C33H37N6O8PS/c1-23(36-32(42)29(22-40)39-49(44,45)28-15-9-4-10-16-28)31(41)38-30(21-24-17-19-25(20-18-24)37-33(34)35)48(43,46-26-11-5-2-6-12-26)47-27-13-7-3-8-14-27/h2-20,23,29-30,39-40H,21-22H2,1H3,(H,36,42)(H,38,41)(H4,34,35,37)/t23-,29+,30?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228418
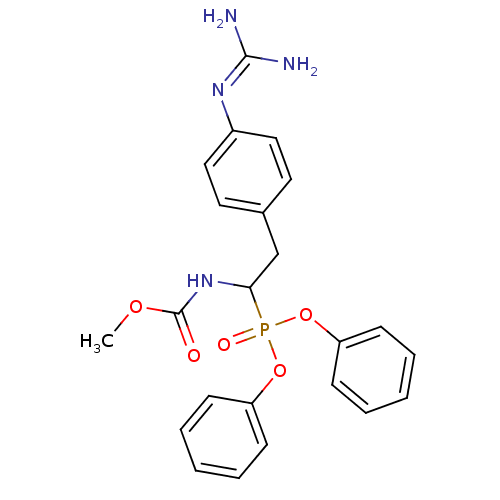
(CHEMBL393591 | methyl 1-(diphenoxyphosphoryl)-2-(4...)Show SMILES [#6]-[#8]-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C23H25N4O5P/c1-30-23(28)27-21(16-17-12-14-18(15-13-17)26-22(24)25)33(29,31-19-8-4-2-5-9-19)32-20-10-6-3-7-11-20/h2-15,21H,16H2,1H3,(H,27,28)(H4,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088982
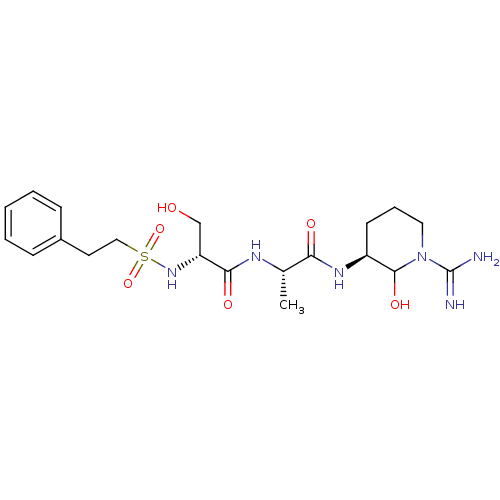
(CHEMBL160253 | CHEMBL367004 | N-[1-(1-Carbamimidoy...)Show SMILES C[C@H](NC(=O)[C@@H](CO)NS(=O)(=O)CCc1ccccc1)C(=O)N[C@H]1CCCN(C1O)C(N)=N Show InChI InChI=1S/C20H32N6O6S/c1-13(17(28)24-15-8-5-10-26(19(15)30)20(21)22)23-18(29)16(12-27)25-33(31,32)11-9-14-6-3-2-4-7-14/h2-4,6-7,13,15-16,19,25,27,30H,5,8-12H2,1H3,(H3,21,22)(H,23,29)(H,24,28)/t13-,15-,16+,19?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228412

(CHEMBL393979 | methyl 1-(bis(4-acetamidophenoxy)ph...)Show SMILES [#6]-[#8]-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccc(-[#7]-[#6](-[#6])=O)cc1)[#8]-c1ccc(-[#7]-[#6](-[#6])=O)cc1 Show InChI InChI=1S/C27H31N6O7P/c1-17(34)30-20-8-12-23(13-9-20)39-41(37,40-24-14-10-21(11-15-24)31-18(2)35)25(33-27(36)38-3)16-19-4-6-22(7-5-19)32-26(28)29/h4-15,25H,16H2,1-3H3,(H,30,34)(H,31,35)(H,33,36)(H4,28,29,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194743
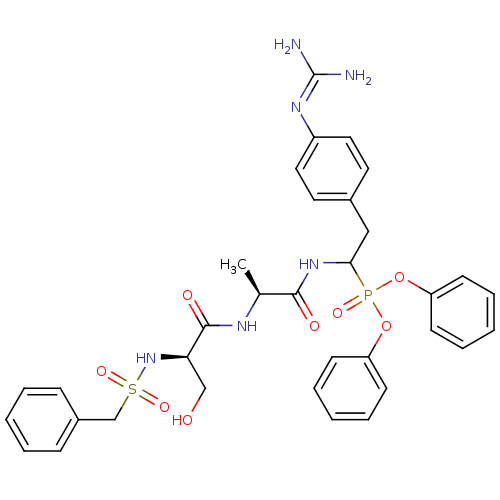
(CHEMBL214814 | diphenyl 1-[(N-alpha-toluenesulfony...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H39N6O8PS/c1-24(37-33(43)30(22-41)40-50(45,46)23-26-11-5-2-6-12-26)32(42)39-31(21-25-17-19-27(20-18-25)38-34(35)36)49(44,47-28-13-7-3-8-14-28)48-29-15-9-4-10-16-29/h2-20,24,30-31,40-41H,21-23H2,1H3,(H,37,43)(H,39,42)(H4,35,36,38)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228425

(CHEMBL239118 | di-(4-acetamidophenyl) 1-(methylsul...)Show SMILES [#6]-[#6](=O)-[#7]-c1ccc(-[#8]P(=O)([#8]-c2ccc(-[#7]-[#6](-[#6])=O)cc2)[#6](-[#6]-c2ccc(cc2)\[#7]=[#6](\[#7])-[#7])-[#7]S([#6])(=O)=O)cc1 Show InChI InChI=1S/C26H31N6O7PS/c1-17(33)29-20-8-12-23(13-9-20)38-40(35,39-24-14-10-21(11-15-24)30-18(2)34)25(32-41(3,36)37)16-19-4-6-22(7-5-19)31-26(27)28/h4-15,25,32H,16H2,1-3H3,(H,29,33)(H,30,34)(H4,27,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194743

(CHEMBL214814 | diphenyl 1-[(N-alpha-toluenesulfony...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H39N6O8PS/c1-24(37-33(43)30(22-41)40-50(45,46)23-26-11-5-2-6-12-26)32(42)39-31(21-25-17-19-27(20-18-25)38-34(35)36)49(44,47-28-13-7-3-8-14-28)48-29-15-9-4-10-16-29/h2-20,24,30-31,40-41H,21-23H2,1H3,(H,37,43)(H,39,42)(H4,35,36,38)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228420
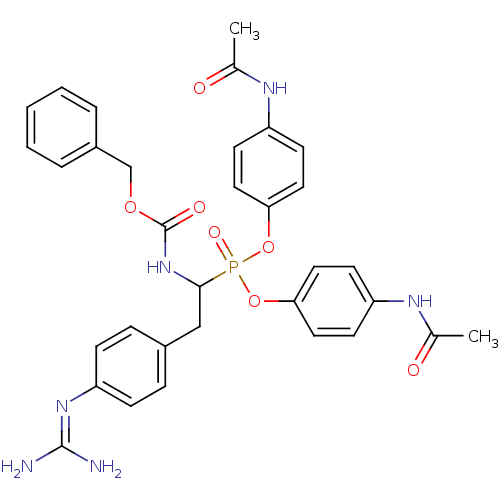
(CHEMBL391968 | di-(4-acetamidophenyl) 1-(benzyloxy...)Show SMILES [#6]-[#6](=O)-[#7]-c1ccc(-[#8]P(=O)([#8]-c2ccc(-[#7]-[#6](-[#6])=O)cc2)[#6](-[#6]-c2ccc(cc2)\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#8]-[#6]-c2ccccc2)cc1 Show InChI InChI=1S/C33H35N6O7P/c1-22(40)36-26-12-16-29(17-13-26)45-47(43,46-30-18-14-27(15-19-30)37-23(2)41)31(20-24-8-10-28(11-9-24)38-32(34)35)39-33(42)44-21-25-6-4-3-5-7-25/h3-19,31H,20-21H2,1-2H3,(H,36,40)(H,37,41)(H,39,42)(H4,34,35,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50145689

(CHEMBL80844 | [1-[(S)-2-((R)-2-Benzyloxycarbonylam...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-[#8]-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](/[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C35H39N6O8P/c1-24(38-33(44)30(22-42)40-35(45)47-23-26-11-5-2-6-12-26)32(43)41-31(21-25-17-19-27(20-18-25)39-34(36)37)50(46,48-28-13-7-3-8-14-28)49-29-15-9-4-10-16-29/h2-20,24,30-31,42H,21-23H2,1H3,(H,38,44)(H,40,45)(H,41,43)(H4,36,37,39)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194737
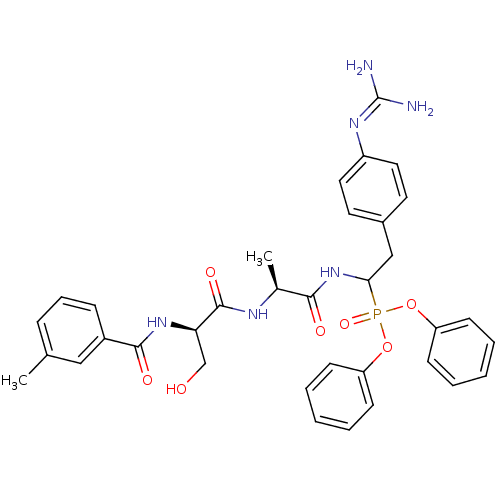
(CHEMBL384263 | diphenyl 1-[(N-o-methylbenzoyl-D-se...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1cccc(-[#6])c1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C35H39N6O7P/c1-23-10-9-11-26(20-23)33(44)40-30(22-42)34(45)38-24(2)32(43)41-31(21-25-16-18-27(19-17-25)39-35(36)37)49(46,47-28-12-5-3-6-13-28)48-29-14-7-4-8-15-29/h3-20,24,30-31,42H,21-22H2,1-2H3,(H,38,45)(H,40,44)(H,41,43)(H4,36,37,39)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194742
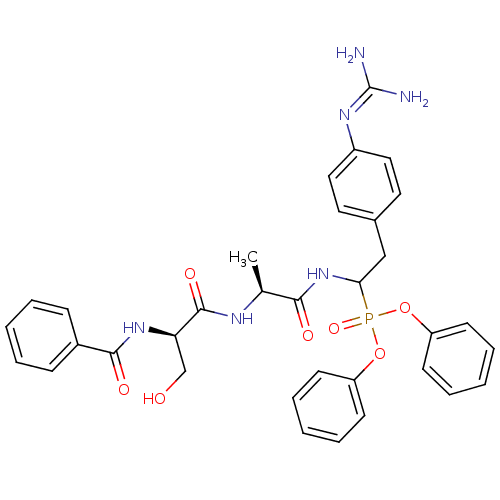
(CHEMBL263977 | diphenyl 1-[(N-benzyl-D-seryl)-L-al...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H37N6O7P/c1-23(37-33(44)29(22-41)39-32(43)25-11-5-2-6-12-25)31(42)40-30(21-24-17-19-26(20-18-24)38-34(35)36)48(45,46-27-13-7-3-8-14-27)47-28-15-9-4-10-16-28/h2-20,23,29-30,41H,21-22H2,1H3,(H,37,44)(H,39,43)(H,40,42)(H4,35,36,38)/t23-,29+,30?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194746
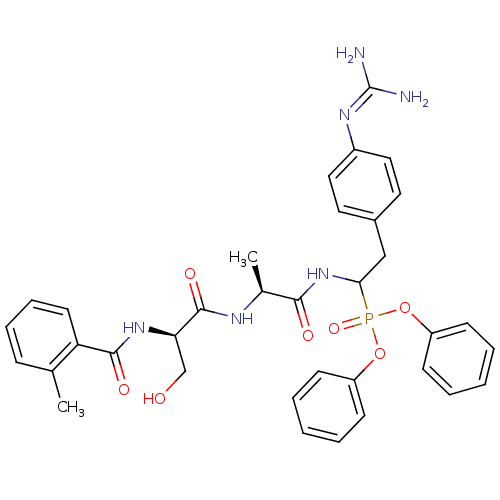
(CHEMBL405546 | diphenyl 1-[(N-o-methylbenzoyl-D-se...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1ccccc1-[#6])-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C35H39N6O7P/c1-23-11-9-10-16-29(23)33(44)40-30(22-42)34(45)38-24(2)32(43)41-31(21-25-17-19-26(20-18-25)39-35(36)37)49(46,47-27-12-5-3-6-13-27)48-28-14-7-4-8-15-28/h3-20,24,30-31,42H,21-22H2,1-2H3,(H,38,45)(H,40,44)(H,41,43)(H4,36,37,39)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088978
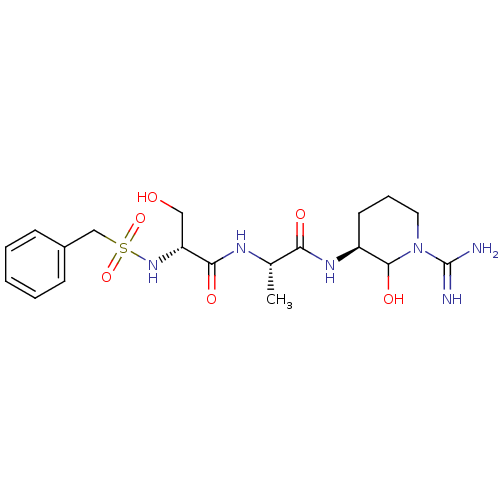
(CHEMBL176515 | N-[1-(1-Carbamimidoyl-2-hydroxy-pip...)Show SMILES C[C@H](NC(=O)[C@@H](CO)NS(=O)(=O)Cc1ccccc1)C(=O)N[C@H]1CCCN(C1O)C(N)=N Show InChI InChI=1S/C19H30N6O6S/c1-12(16(27)23-14-8-5-9-25(18(14)29)19(20)21)22-17(28)15(10-26)24-32(30,31)11-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,18,24,26,29H,5,8-11H2,1H3,(H3,20,21)(H,22,28)(H,23,27)/t12-,14-,15+,18?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194744

(CHEMBL385897 | diphenyl 1-[(N-naphthalenesulfonyl-...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1cccc2ccccc12)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C37H39N6O8PS/c1-25(40-36(46)32(24-44)43-53(48,49)33-18-10-12-27-11-8-9-17-31(27)33)35(45)42-34(23-26-19-21-28(22-20-26)41-37(38)39)52(47,50-29-13-4-2-5-14-29)51-30-15-6-3-7-16-30/h2-22,25,32,34,43-44H,23-24H2,1H3,(H,40,46)(H,42,45)(H4,38,39,41)/t25-,32+,34?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194734

(CHEMBL386275 | diphenyl 1-[(N-p-methoxybenzenesulf...)Show SMILES [#6]-[#8]-c1ccc(cc1)S(=O)(=O)[#7]-[#6@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H39N6O9PS/c1-23(37-33(43)30(22-41)40-51(45,46)29-19-17-26(47-2)18-20-29)32(42)39-31(21-24-13-15-25(16-14-24)38-34(35)36)50(44,48-27-9-5-3-6-10-27)49-28-11-7-4-8-12-28/h3-20,23,30-31,40-41H,21-22H2,1-2H3,(H,37,43)(H,39,42)(H4,35,36,38)/t23-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194736

(CHEMBL189823 | diphenyl 1-[(N-o,o-dimethylbenzoyl-...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1c(-[#6])cccc1-[#6])-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C36H41N6O7P/c1-23-11-10-12-24(2)32(23)35(46)41-30(22-43)34(45)39-25(3)33(44)42-31(21-26-17-19-27(20-18-26)40-36(37)38)50(47,48-28-13-6-4-7-14-28)49-29-15-8-5-9-16-29/h4-20,25,30-31,43H,21-22H2,1-3H3,(H,39,45)(H,41,46)(H,42,44)(H4,37,38,40)/t25-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194735

(CHEMBL215961 | diphenyl 1-[(N-p-cyanobenzenesulfon...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1ccc(cc1)C#N)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C34H36N7O8PS/c1-23(38-33(44)30(22-42)41-51(46,47)29-18-14-25(21-35)15-19-29)32(43)40-31(20-24-12-16-26(17-13-24)39-34(36)37)50(45,48-27-8-4-2-5-9-27)49-28-10-6-3-7-11-28/h2-19,23,30-31,41-42H,20,22H2,1H3,(H,38,44)(H,40,43)(H4,36,37,39)/t23-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194738
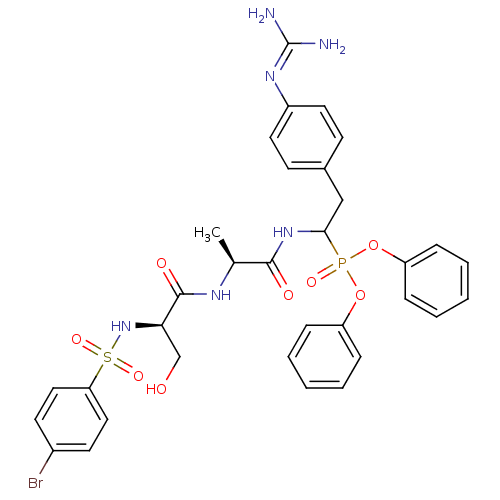
(CHEMBL386249 | diphenyl 1-[(N-p-bromobenzenesulfon...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)c1ccc(Br)cc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C33H36BrN6O8PS/c1-22(37-32(43)29(21-41)40-50(45,46)28-18-14-24(34)15-19-28)31(42)39-30(20-23-12-16-25(17-13-23)38-33(35)36)49(44,47-26-8-4-2-5-9-26)48-27-10-6-3-7-11-27/h2-19,22,29-30,40-41H,20-21H2,1H3,(H,37,43)(H,39,42)(H4,35,36,38)/t22-,29+,30?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM92479
(Tannic Acid, A | Tannic acid)Show SMILES Oc1cc(cc(O)c1O)C(=O)Oc1cc(cc(O)c1O)C(=O)OCC1OC(OC(=O)c2cc(O)c(O)c(OC(=O)c3cc(O)c(O)c(O)c3)c2)C(OC(=O)c2cc(O)c(O)c(OC(=O)c3cc(O)c(O)c(O)c3)c2)C(OC(=O)c2cc(O)c(O)c(OC(=O)c3cc(O)c(O)c(O)c3)c2)C1OC(=O)c1cc(O)c(O)c(OC(=O)c2cc(O)c(O)c(O)c2)c1 Show InChI InChI=1S/C76H52O46/c77-32-1-22(2-33(78)53(32)92)67(103)113-47-16-27(11-42(87)58(47)97)66(102)112-21-52-63(119-72(108)28-12-43(88)59(98)48(17-28)114-68(104)23-3-34(79)54(93)35(80)4-23)64(120-73(109)29-13-44(89)60(99)49(18-29)115-69(105)24-5-36(81)55(94)37(82)6-24)65(121-74(110)30-14-45(90)61(100)50(19-30)116-70(106)25-7-38(83)56(95)39(84)8-25)76(118-52)122-75(111)31-15-46(91)62(101)51(20-31)117-71(107)26-9-40(85)57(96)41(86)10-26/h1-20,52,63-65,76-101H,21H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.60 | n/a | n/a | n/a | n/a | 7.8 | 23 |
University of Michigan
| Assay Description
Enzyme activity assay using human and murine PAI-1. |
J Biol Chem 285: 7892-902 (2010)
Article DOI: 10.1074/jbc.M109.067967
BindingDB Entry DOI: 10.7270/Q2ZC81FC |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228421
(CHEMBL238493 | diphenyl 1-(methylsulfonylamino)-2-...)Show SMILES [#6]S(=O)(=O)[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 |w:5.4| Show InChI InChI=1S/C22H25N4O5PS/c1-33(28,29)26-21(16-17-12-14-18(15-13-17)25-22(23)24)32(27,30-19-8-4-2-5-9-19)31-20-10-6-3-7-11-20/h2-15,21,26H,16H2,1H3,(H4,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194732
(CHEMBL214539 | di-(4-acetamidophenyl) 1-[(N-benzyl...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-[#8]-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccc(-[#7]-[#6](-[#6])=O)cc1)[#8]-c1ccc(-[#7]-[#6](-[#6])=O)cc1 Show InChI InChI=1S/C39H45N8O10P/c1-24(42-37(52)34(22-48)46-39(53)55-23-28-7-5-4-6-8-28)36(51)47-35(21-27-9-11-31(12-10-27)45-38(40)41)58(54,56-32-17-13-29(14-18-32)43-25(2)49)57-33-19-15-30(16-20-33)44-26(3)50/h4-20,24,34-35,48H,21-23H2,1-3H3,(H,42,52)(H,43,49)(H,44,50)(H,46,53)(H,47,51)(H4,40,41,45)/t24-,34+,35?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotin-linked K-ras decapeptide (CVIM) by bovine farnesyltransferase |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194739

(CHEMBL213216 | diphenyl 1-[(N-2-thienyl-D-seryl)-L...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1cccs1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C32H35N6O7PS/c1-21(35-30(41)26(20-39)37-31(42)27-13-8-18-47-27)29(40)38-28(19-22-14-16-23(17-15-22)36-32(33)34)46(43,44-24-9-4-2-5-10-24)45-25-11-6-3-7-12-25/h2-18,21,26,28,39H,19-20H2,1H3,(H,35,41)(H,37,42)(H,38,40)(H4,33,34,36)/t21-,26+,28?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228410
(CHEMBL239535 | diphenyl 1-(benzyloxycarbonylamino)...)Show SMILES [#7]\[#6](-[#7])=[#7]\c1ccc(-[#6]-[#6](-[#7]-[#6](=O)-[#8]-[#6]-c2ccccc2)P(=O)([#8]-c2ccccc2)[#8]-c2ccccc2)cc1 Show InChI InChI=1S/C29H29N4O5P/c30-28(31)32-24-18-16-22(17-19-24)20-27(33-29(34)36-21-23-10-4-1-5-11-23)39(35,37-25-12-6-2-7-13-25)38-26-14-8-3-9-15-26/h1-19,27H,20-21H2,(H,33,34)(H4,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194740
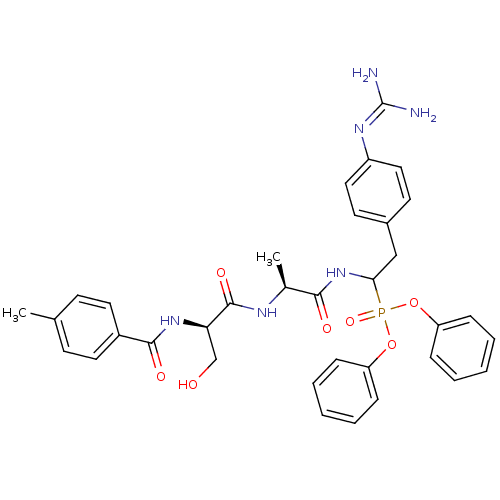
(CHEMBL404226 | diphenyl 1-[(N-p-methylbenzoyl-D-se...)Show SMILES [#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-c1ccc(-[#6])cc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C35H39N6O7P/c1-23-13-17-26(18-14-23)33(44)40-30(22-42)34(45)38-24(2)32(43)41-31(21-25-15-19-27(20-16-25)39-35(36)37)49(46,47-28-9-5-3-6-10-28)48-29-11-7-4-8-12-29/h3-20,24,30-31,42H,21-22H2,1-2H3,(H,38,45)(H,40,44)(H,41,43)(H4,36,37,39)/t24-,30+,31?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228414
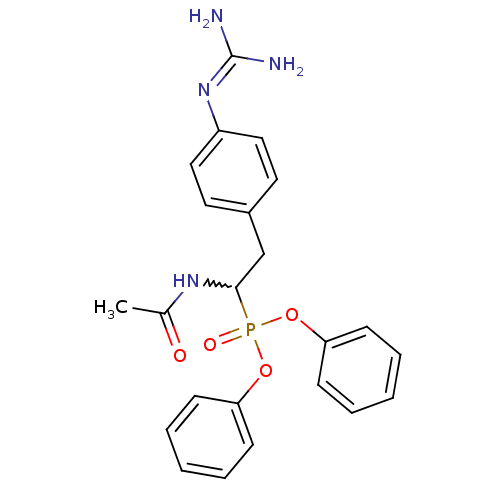
(CHEMBL239747 | diphenyl 1-acetamido-2-(4-guanidino...)Show SMILES [#6]-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 |w:4.3| Show InChI InChI=1S/C23H25N4O4P/c1-17(28)26-22(16-18-12-14-19(15-13-18)27-23(24)25)32(29,30-20-8-4-2-5-9-20)31-21-10-6-3-7-11-21/h2-15,22H,16H2,1H3,(H,26,28)(H4,24,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.20 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
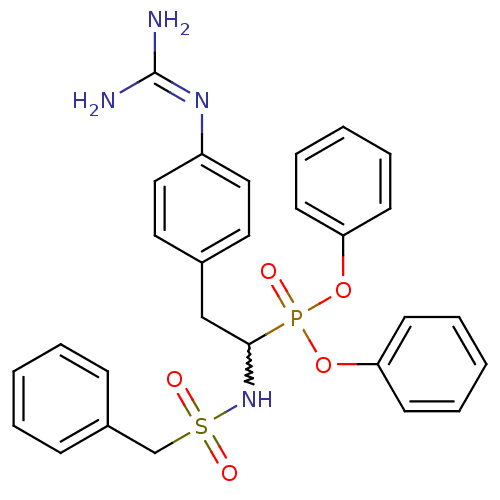
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228417
(CHEMBL238491 | diphenyl 1-(o-toluenesulfonylamino)...)Show SMILES [#7]\[#6](-[#7])=[#7]\c1ccc(-[#6]-[#6](-[#7]S(=O)(=O)[#6]-c2ccccc2)P(=O)([#8]-c2ccccc2)[#8]-c2ccccc2)cc1 |w:9.9| Show InChI InChI=1S/C28H29N4O5PS/c29-28(30)31-24-18-16-22(17-19-24)20-27(32-39(34,35)21-23-10-4-1-5-11-23)38(33,36-25-12-6-2-7-13-25)37-26-14-8-3-9-15-26/h1-19,27,32H,20-21H2,(H4,29,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.70 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228423
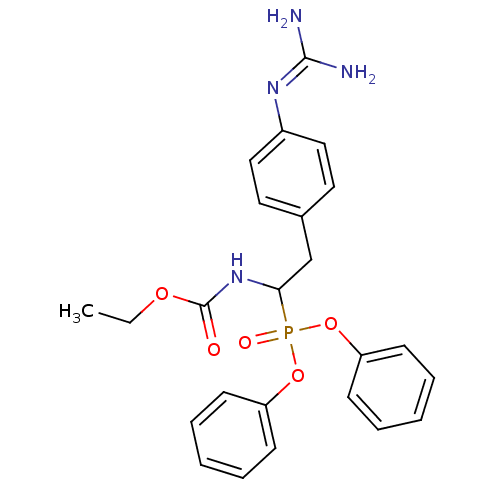
(CHEMBL239536 | ethyl 1-(diphenoxyphosphoryl)-2-(4-...)Show SMILES [#6]-[#6]-[#8]-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1 Show InChI InChI=1S/C24H27N4O5P/c1-2-31-24(29)28-22(17-18-13-15-19(16-14-18)27-23(25)26)34(30,32-20-9-5-3-6-10-20)33-21-11-7-4-8-12-21/h3-16,22H,2,17H2,1H3,(H,28,29)(H4,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50194733

(CHEMBL411215 | diphenyl 1-[(N-1-adamantanyl-D-sery...)Show SMILES C[C@H](NC(=O)[C@@H](CO)NC(=O)C1C2CC3CC(C2)CC1C3)C(=O)NC(Cc1ccc(NC(N)=N)cc1)P(=O)(Oc1ccccc1)Oc1ccccc1 |TLB:17:16:20:13.12.11,THB:18:16:13:20.19.11| Show InChI InChI=1S/C38H47N6O7P/c1-23(41-36(47)32(22-45)43-37(48)34-27-17-25-16-26(19-27)20-28(34)18-25)35(46)44-33(21-24-12-14-29(15-13-24)42-38(39)40)52(49,50-30-8-4-2-5-9-30)51-31-10-6-3-7-11-31/h2-15,23,25-28,32-34,45H,16-22H2,1H3,(H,41,47)(H,43,48)(H,44,46)(H4,39,40,42)/t23-,25?,26?,27?,28?,32+,33?,34?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 49: 5785-93 (2006)
Article DOI: 10.1021/jm060622g
BindingDB Entry DOI: 10.7270/Q2SF2VT3 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088984
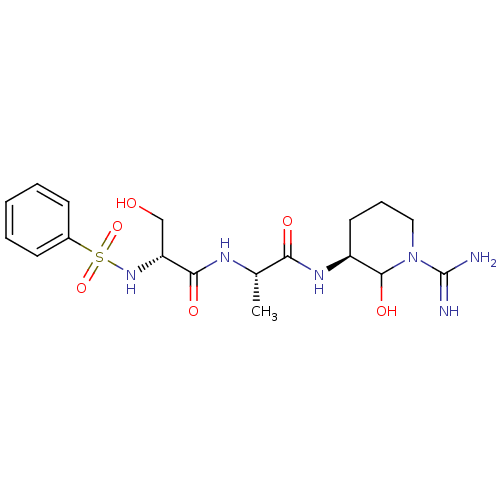
(2-Benzenesulfonylamino-N-[1-(1-carbamimidoyl-2-hyd...)Show SMILES C[C@H](NC(=O)[C@@H](CO)NS(=O)(=O)c1ccccc1)C(=O)N[C@H]1CCCN(C1O)C(N)=N Show InChI InChI=1S/C18H28N6O6S/c1-11(15(26)22-13-8-5-9-24(17(13)28)18(19)20)21-16(27)14(10-25)23-31(29,30)12-6-3-2-4-7-12/h2-4,6-7,11,13-14,17,23,25,28H,5,8-10H2,1H3,(H3,19,20)(H,21,27)(H,22,26)/t11-,13-,14+,17?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM92485
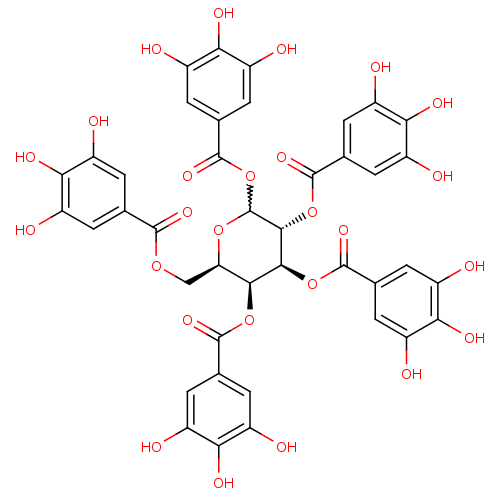
(CDE-066 | US9120744, CDE-066)Show SMILES Oc1cc(cc(O)c1O)C(=O)OC[C@H]1OC(OC(=O)c2cc(O)c(O)c(O)c2)[C@H](OC(=O)c2cc(O)c(O)c(O)c2)[C@@H](OC(=O)c2cc(O)c(O)c(O)c2)[C@H]1OC(=O)c1cc(O)c(O)c(O)c1 |r,w:15.16| Show InChI InChI=1S/C41H32O26/c42-17-1-12(2-18(43)28(17)52)36(57)62-11-27-33(64-37(58)13-3-19(44)29(53)20(45)4-13)34(65-38(59)14-5-21(46)30(54)22(47)6-14)35(66-39(60)15-7-23(48)31(55)24(49)8-15)41(63-27)67-40(61)16-9-25(50)32(56)26(51)10-16/h1-10,27,33-35,41-56H,11H2/t27-,33+,34+,35-,41?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | 7.8 | 23 |
University of Michigan
| Assay Description
Enzyme activity assay using human and murine PAI-1. |
J Biol Chem 285: 7892-902 (2010)
Article DOI: 10.1074/jbc.M109.067967
BindingDB Entry DOI: 10.7270/Q2ZC81FC |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088979

(CHEMBL366666 | {(R)-2-[2-((S)-1-Carbamimidoyl-2-hy...)Show SMILES NC(=N)N1CCC[C@H](NC(=O)C2CCN2C(=O)[C@@H](CO)NC(=O)OCc2ccccc2)C1O Show InChI InChI=1S/C21H30N6O6/c22-20(23)27-9-4-7-14(18(27)30)24-17(29)16-8-10-26(16)19(31)15(11-28)25-21(32)33-12-13-5-2-1-3-6-13/h1-3,5-6,14-16,18,28,30H,4,7-12H2,(H3,22,23)(H,24,29)(H,25,32)/t14-,15+,16?,18?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088985
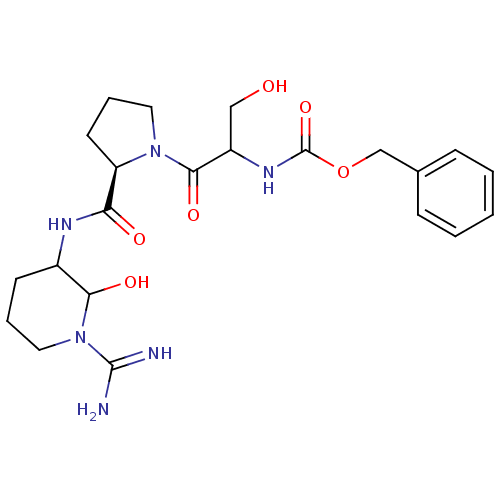
(CHEMBL369042 | {2-[2-(1-Carbamimidoyl-2-hydroxy-pi...)Show SMILES NC(=N)N1CCCC(NC(=O)[C@H]2CCCN2C(=O)C(CO)NC(=O)OCc2ccccc2)C1O Show InChI InChI=1S/C22H32N6O6/c23-21(24)28-11-4-8-15(19(28)31)25-18(30)17-9-5-10-27(17)20(32)16(12-29)26-22(33)34-13-14-6-2-1-3-7-14/h1-3,6-7,15-17,19,29,31H,4-5,8-13H2,(H3,23,24)(H,25,30)(H,26,33)/t15?,16?,17-,19?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM92486
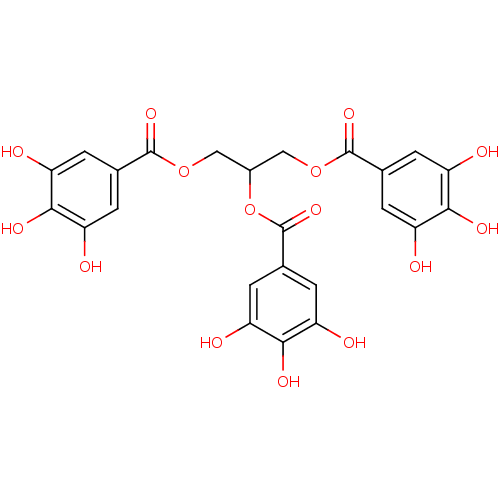
(CDE-082 | US9120744, CDE-082)Show SMILES Oc1cc(cc(O)c1O)C(=O)OCC(COC(=O)c1cc(O)c(O)c(O)c1)OC(=O)c1cc(O)c(O)c(O)c1 Show InChI InChI=1S/C24H20O15/c25-13-1-9(2-14(26)19(13)31)22(34)37-7-12(39-24(36)11-5-17(29)21(33)18(30)6-11)8-38-23(35)10-3-15(27)20(32)16(28)4-10/h1-6,12,25-33H,7-8H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | 7.8 | 23 |
University of Michigan
| Assay Description
Enzyme activity assay using human and murine PAI-1. |
J Biol Chem 285: 7892-902 (2010)
Article DOI: 10.1074/jbc.M109.067967
BindingDB Entry DOI: 10.7270/Q2ZC81FC |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50301972
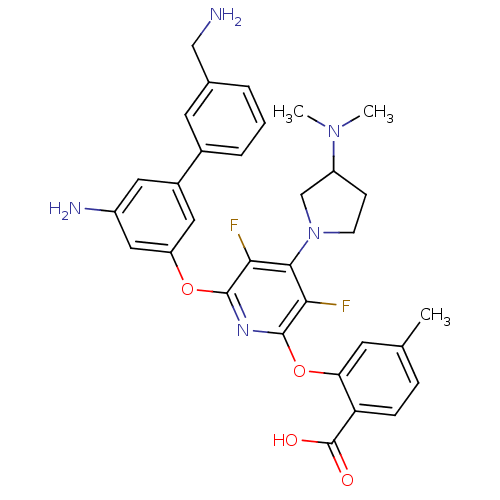
(2-(6-(5-amino-3'-(aminomethyl)biphenyl-3-yloxy)-4-...)Show SMILES CN(C)C1CCN(C1)c1c(F)c(Oc2cc(N)cc(c2)-c2cccc(CN)c2)nc(Oc2cc(C)ccc2C(O)=O)c1F Show InChI InChI=1S/C32H33F2N5O4/c1-18-7-8-25(32(40)41)26(11-18)43-31-28(34)29(39-10-9-23(17-39)38(2)3)27(33)30(37-31)42-24-14-21(13-22(36)15-24)20-6-4-5-19(12-20)16-35/h4-8,11-15,23H,9-10,16-17,35-36H2,1-3H3,(H,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibition of uPA |
Bioorg Med Chem Lett 19: 5712-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.008
BindingDB Entry DOI: 10.7270/Q2JQ113Z |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50542728
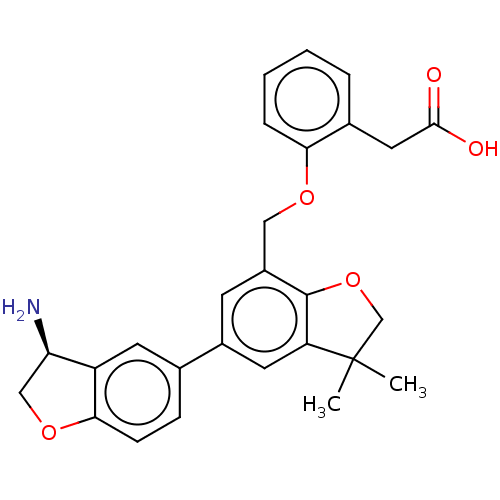
(CHEMBL4635912)Show SMILES CC1(C)COc2c1cc(cc2COc1ccccc1CC(O)=O)-c1ccc2OC[C@@H](N)c2c1 |r| Show InChI InChI=1S/C27H27NO5/c1-27(2)15-33-26-19(13-31-23-6-4-3-5-17(23)12-25(29)30)9-18(11-21(26)27)16-7-8-24-20(10-16)22(28)14-32-24/h3-11,22H,12-15,28H2,1-2H3,(H,29,30)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human urokinase using fluorescent peptide as substrate by florescence assay |
J Med Chem 63: 8088-8113 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00279
BindingDB Entry DOI: 10.7270/Q2KS6W36 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088977
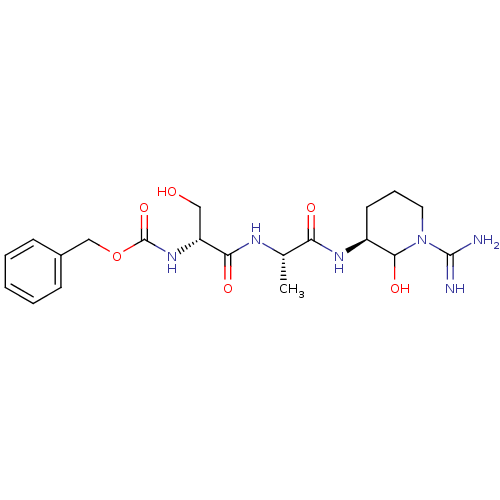
(CHEMBL177557 | {1-[1-(1-Carbamimidoyl-2-hydroxy-pi...)Show SMILES C[C@H](NC(=O)[C@@H](CO)NC(=O)OCc1ccccc1)C(=O)N[C@H]1CCCN(C1O)C(N)=N Show InChI InChI=1S/C20H30N6O6/c1-12(16(28)24-14-8-5-9-26(18(14)30)19(21)22)23-17(29)15(10-27)25-20(31)32-11-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,18,27,30H,5,8-11H2,1H3,(H3,21,22)(H,23,29)(H,24,28)(H,25,31)/t12-,14-,15+,18?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50542732
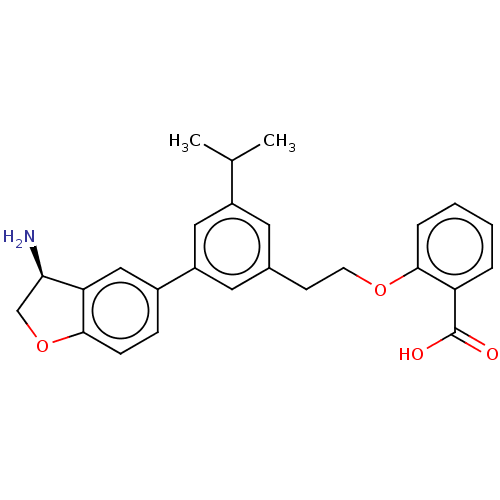
(CHEMBL4647925)Show SMILES CC(C)c1cc(CCOc2ccccc2C(O)=O)cc(c1)-c1ccc2OC[C@@H](N)c2c1 |r| Show InChI InChI=1S/C26H27NO4/c1-16(2)19-11-17(9-10-30-24-6-4-3-5-21(24)26(28)29)12-20(13-19)18-7-8-25-22(14-18)23(27)15-31-25/h3-8,11-14,16,23H,9-10,15,27H2,1-2H3,(H,28,29)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human uPA using fluorescent peptide as substrate by florescence assay |
J Med Chem 63: 8088-8113 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00279
BindingDB Entry DOI: 10.7270/Q2KS6W36 |
More data for this
Ligand-Target Pair | |
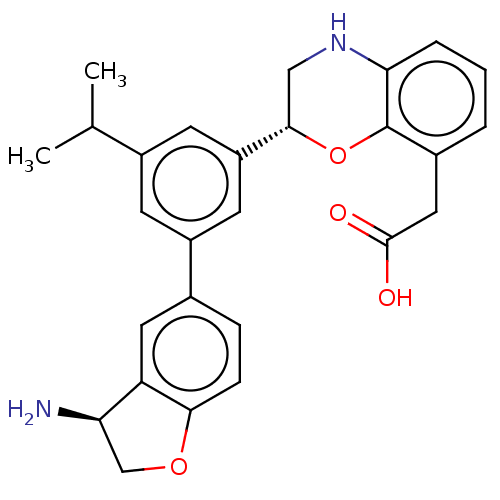
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50542731
(CHEMBL4642845)Show SMILES CC(C)c1cc(cc(c1)-c1ccc2OC[C@@H](N)c2c1)[C@@H]1CNc2cccc(CC(O)=O)c2O1 |r| Show InChI InChI=1S/C27H28N2O4/c1-15(2)18-8-19(16-6-7-24-21(11-16)22(28)14-32-24)10-20(9-18)25-13-29-23-5-3-4-17(12-26(30)31)27(23)33-25/h3-11,15,22,25,29H,12-14,28H2,1-2H3,(H,30,31)/t22-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human uPA using fluorescent peptide as substrate by florescence assay |
J Med Chem 63: 8088-8113 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00279
BindingDB Entry DOI: 10.7270/Q2KS6W36 |
More data for this
Ligand-Target Pair | |
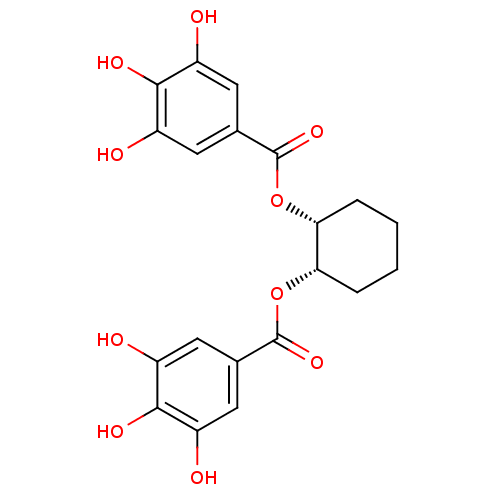
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM92487
(CDE-031 | US9120744, CDE-031)Show SMILES Oc1cc(cc(O)c1O)C(=O)O[C@@H]1CCCC[C@@H]1OC(=O)c1cc(O)c(O)c(O)c1 Show InChI InChI=1S/C20H20O10/c21-11-5-9(6-12(22)17(11)25)19(27)29-15-3-1-2-4-16(15)30-20(28)10-7-13(23)18(26)14(24)8-10/h5-8,15-16,21-26H,1-4H2/t15-,16+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | 7.8 | 23 |
University of Michigan
| Assay Description
Enzyme activity assay using human and murine PAI-1. |
J Biol Chem 285: 7892-902 (2010)
Article DOI: 10.1074/jbc.M109.067967
BindingDB Entry DOI: 10.7270/Q2ZC81FC |
More data for this
Ligand-Target Pair | |
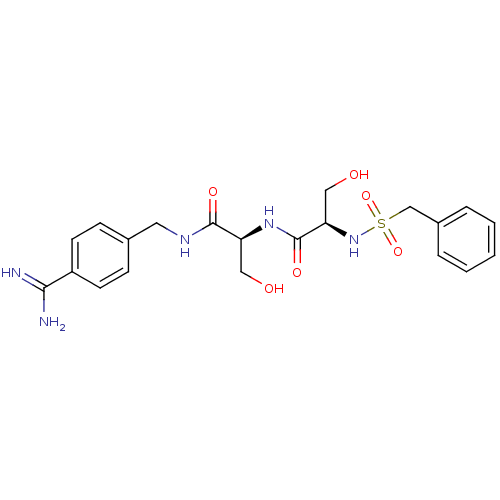
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50231520
((R)-N-[(S)-1-(4-carbamimidoyl-benzylcarbamoyl)-2-h...)Show SMILES NC(=N)c1ccc(CNC(=O)[C@H](CO)NC(=O)[C@@H](CO)NS(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C21H27N5O6S/c22-19(23)16-8-6-14(7-9-16)10-24-20(29)17(11-27)25-21(30)18(12-28)26-33(31,32)13-15-4-2-1-3-5-15/h1-9,17-18,26-28H,10-13H2,(H3,22,23)(H,24,29)(H,25,30)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Astex Therapeutics Ltd
Curated by ChEMBL
| Assay Description
Inhibition of uPA |
J Med Chem 51: 183-6 (2008)
Article DOI: 10.1021/jm701359z
BindingDB Entry DOI: 10.7270/Q2XG9QWK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50301973
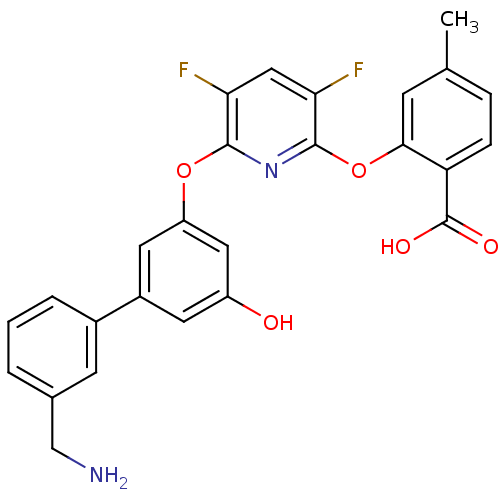
(2-(6-(3'-(aminomethyl)-5-hydroxybiphenyl-3-yloxy)-...)Show SMILES Cc1ccc(C(O)=O)c(Oc2nc(Oc3cc(O)cc(c3)-c3cccc(CN)c3)c(F)cc2F)c1 Show InChI InChI=1S/C26H20F2N2O5/c1-14-5-6-20(26(32)33)23(7-14)35-25-22(28)12-21(27)24(30-25)34-19-10-17(9-18(31)11-19)16-4-2-3-15(8-16)13-29/h2-12,31H,13,29H2,1H3,(H,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibition of uPA |
Bioorg Med Chem Lett 19: 5712-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.008
BindingDB Entry DOI: 10.7270/Q2JQ113Z |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50467209
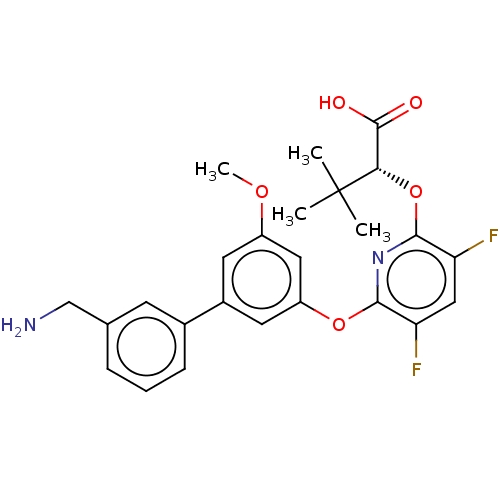
(CHEMBL4291446)Show SMILES COc1cc(Oc2nc(O[C@@H](C(O)=O)C(C)(C)C)c(F)cc2F)cc(c1)-c1cccc(CN)c1 |r| Show InChI InChI=1S/C25H26F2N2O5/c1-25(2,3)21(24(30)31)34-23-20(27)12-19(26)22(29-23)33-18-10-16(9-17(11-18)32-4)15-7-5-6-14(8-15)13-28/h5-12,21H,13,28H2,1-4H3,(H,30,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Abdulaziz University for Health Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
Bioorg Med Chem Lett 28: 3372-3375 (2018)
Article DOI: 10.1016/j.bmcl.2018.09.001
BindingDB Entry DOI: 10.7270/Q24Q7XNF |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50088987
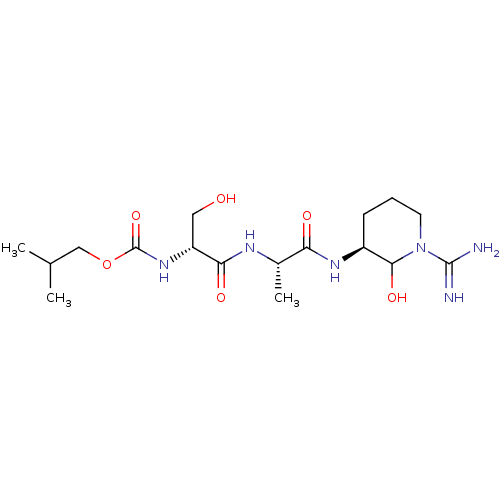
(CHEMBL174813 | {1-[1-(1-Carbamimidoyl-2-hydroxy-pi...)Show SMILES CC(C)COC(=O)N[C@H](CO)C(=O)N[C@@H](C)C(=O)N[C@H]1CCCN(C1O)C(N)=N Show InChI InChI=1S/C17H32N6O6/c1-9(2)8-29-17(28)22-12(7-24)14(26)20-10(3)13(25)21-11-5-4-6-23(15(11)27)16(18)19/h9-12,15,24,27H,4-8H2,1-3H3,(H3,18,19)(H,20,26)(H,21,25)(H,22,28)/t10-,11-,12+,15?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Corvas International, Inc.
Curated by ChEMBL
| Assay Description
The compound was tested in vitro for its inhibitory activity against human urokinase enzyme, activity expressed as IC50 |
Bioorg Med Chem Lett 10: 983-7 (2000)
BindingDB Entry DOI: 10.7270/Q2765DKB |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50301982
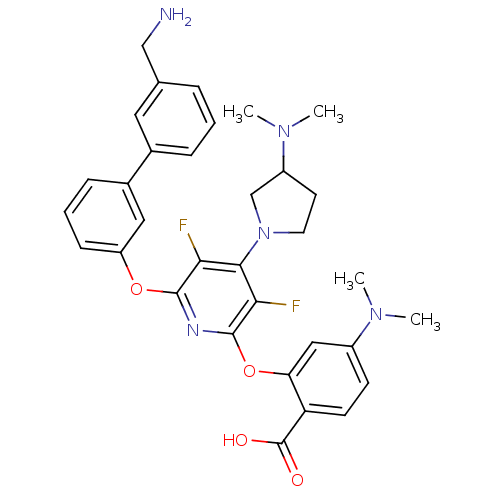
(2-(6-(3'-(aminomethyl)biphenyl-3-yloxy)-4-(3-(dime...)Show SMILES CN(C)C1CCN(C1)c1c(F)c(Oc2cccc(c2)-c2cccc(CN)c2)nc(Oc2cc(ccc2C(O)=O)N(C)C)c1F Show InChI InChI=1S/C33H35F2N5O4/c1-38(2)23-11-12-26(33(41)42)27(17-23)44-32-29(35)30(40-14-13-24(19-40)39(3)4)28(34)31(37-32)43-25-10-6-9-22(16-25)21-8-5-7-20(15-21)18-36/h5-12,15-17,24H,13-14,18-19,36H2,1-4H3,(H,41,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibition of uPA |
Bioorg Med Chem Lett 19: 5712-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.008
BindingDB Entry DOI: 10.7270/Q2JQ113Z |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50184648
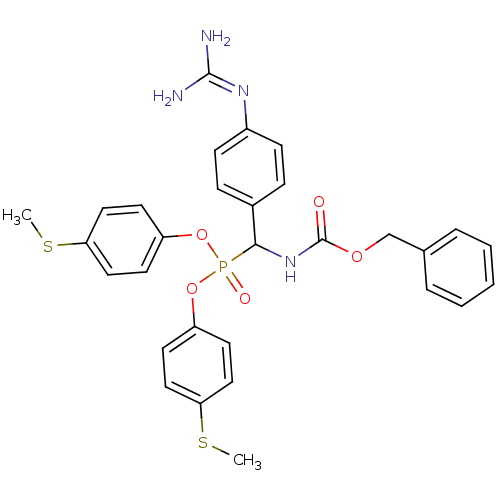
(CHEMBL378976 | [benzyloxycarbonylamino-(4-guanidin...)Show SMILES [#6]-[#16]-c1ccc(-[#8]P(=O)([#8]-c2ccc(-[#16]-[#6])cc2)[#6](-[#7]-[#6](=O)-[#8]-[#6]-c2ccccc2)-c2ccc(cc2)\[#7]=[#6](\[#7])-[#7])cc1 Show InChI InChI=1S/C30H31N4O5PS2/c1-41-26-16-12-24(13-17-26)38-40(36,39-25-14-18-27(42-2)19-15-25)28(22-8-10-23(11-9-22)33-29(31)32)34-30(35)37-20-21-6-4-3-5-7-21/h3-19,28H,20H2,1-2H3,(H,34,35)(H4,31,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Wroc£?aw University of Technology
Curated by ChEMBL
| Assay Description
Inhibition of human uPA by chromogenic assay using Cbz-Val-Gly-Arg-pNA as chromogenic substrate |
Bioorg Med Chem Lett 16: 2886-90 (2006)
Article DOI: 10.1016/j.bmcl.2006.03.002
BindingDB Entry DOI: 10.7270/Q22N51WR |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50228426
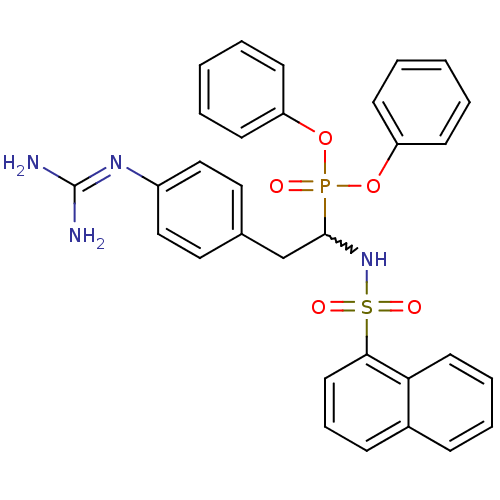
(CHEMBL391344 | diphenyl 1-(naphthalenesulfonylamin...)Show SMILES [#7]\[#6](-[#7])=[#7]\c1ccc(-[#6]-[#6](-[#7]S(=O)(=O)c2cccc3ccccc23)P(=O)([#8]-c2ccccc2)[#8]-c2ccccc2)cc1 |w:9.9| Show InChI InChI=1S/C31H29N4O5PS/c32-31(33)34-25-20-18-23(19-21-25)22-30(35-42(37,38)29-17-9-11-24-10-7-8-16-28(24)29)41(36,39-26-12-3-1-4-13-26)40-27-14-5-2-6-15-27/h1-21,30,35H,22H2,(H4,32,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Antwerp
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
J Med Chem 50: 6638-46 (2007)
Article DOI: 10.1021/jm700962j
BindingDB Entry DOI: 10.7270/Q2MW2J0J |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50144551
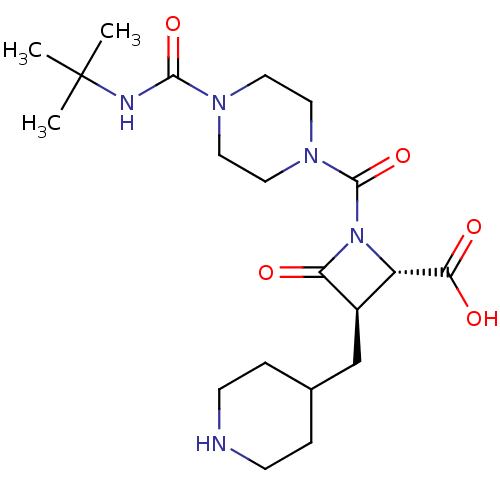
((2S,3R)-1-(4-tert-Butylcarbamoyl-piperazine-1-carb...)Show SMILES CC(C)(C)NC(=O)N1CCN(CC1)C(=O)N1[C@@H]([C@@H](CC2CCNCC2)C1=O)C(O)=O Show InChI InChI=1S/C20H33N5O5/c1-20(2,3)22-18(29)23-8-10-24(11-9-23)19(30)25-15(17(27)28)14(16(25)26)12-13-4-6-21-7-5-13/h13-15,21H,4-12H2,1-3H3,(H,22,29)(H,27,28)/t14-,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
The Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity against Urokinase-type plasminogen activator |
Bioorg Med Chem Lett 14: 2233-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.012
BindingDB Entry DOI: 10.7270/Q2SB4697 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50467207

(CHEMBL4288828)Show SMILES CC(C)(C)[C@@H](Oc1nc(Oc2cc(cc(c2)-c2cccc(CN)c2)C#N)c(F)cc1F)C(O)=O |r| Show InChI InChI=1S/C25H23F2N3O4/c1-25(2,3)21(24(31)32)34-23-20(27)11-19(26)22(30-23)33-18-9-15(13-29)8-17(10-18)16-6-4-5-14(7-16)12-28/h4-11,21H,12,28H2,1-3H3,(H,31,32)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Abdulaziz University for Health Sciences
Curated by ChEMBL
| Assay Description
Inhibition of human uPA |
Bioorg Med Chem Lett 28: 3372-3375 (2018)
Article DOI: 10.1016/j.bmcl.2018.09.001
BindingDB Entry DOI: 10.7270/Q24Q7XNF |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data