

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 2E1 (Homo sapiens (Human)) | BDBM50240850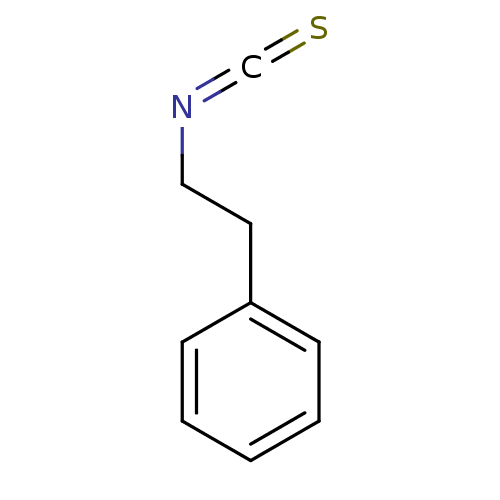 ((2-Isothiocyanato-ethyl)-benzene | CHEMBL151649 | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents | PubMed | 9.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Mechanism based inhibition of human cytochrome P450 2E1 | Curr Drug Metab 6: 413-54 (2005) BindingDB Entry DOI: 10.7270/Q2VQ33X3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2E1 (Homo sapiens (Human)) | BDBM50240850 ((2-Isothiocyanato-ethyl)-benzene | CHEMBL151649 | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents | Article PubMed | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Michigan Medical School Curated by ChEMBL | Assay Description Inhibition of human CYP2E1 expressed in Escherichia coli MV1304 assessed as reduction in 7-EFC O-de-ethylation activity by spectrofluorometry based d... | Drug Metab Dispos 41: 858-69 (2013) Article DOI: 10.1124/dmd.112.050609 BindingDB Entry DOI: 10.7270/Q2V69MBS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2E1 (Homo sapiens (Human)) | BDBM50088490 (CHEMBL3526979) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc Curated by ChEMBL | Assay Description Inhibition of CYP2E1 in human liver microsomes using chlorzoxazone as substrate after 5 to 30 mins by LC-MS/MS analysis | Drug Metab Dispos 40: 2239-49 (2012) Article DOI: 10.1124/dmd.112.047928 BindingDB Entry DOI: 10.7270/Q2Z039WT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2E1 (Homo sapiens (Human)) | BDBM50363928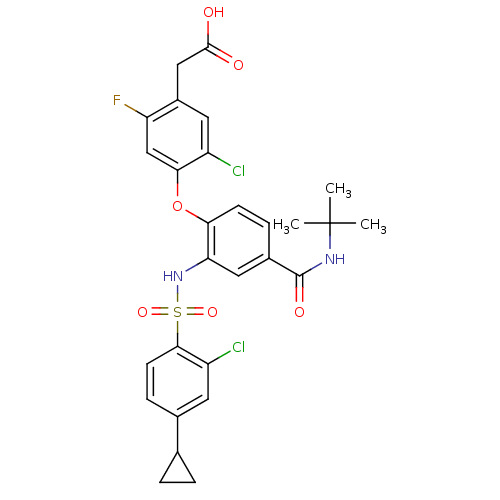 (CHEMBL1951575) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc Curated by ChEMBL | Assay Description Inhibition of CYP2E1 in human liver microsomes using chlorzoxazone as substrate after 5 to 30 mins by LC-MS/MS analysis | Drug Metab Dispos 40: 2239-49 (2012) Article DOI: 10.1124/dmd.112.047928 BindingDB Entry DOI: 10.7270/Q2Z039WT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2E1 (Homo sapiens (Human)) | BDBM50310823 (CHEMBL1078442 | bergamottin) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd. Curated by ChEMBL | Assay Description Mechanism based inhibition of human cytochrome P450 2E1 | Curr Drug Metab 6: 413-54 (2005) BindingDB Entry DOI: 10.7270/Q2VQ33X3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2E1 (Homo sapiens (Human)) | BDBM50088436 (BS-749 | CHEBI:76987 | Metacetamol) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Patents | Article PubMed | 9.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pacific University Oregon Curated by ChEMBL | Assay Description Inhibition of human CYP2E1 assessed as chlorzoxazone 6-hydroxylase activity by HPLC analysis | Drug Metab Dispos 40: 1460-5 (2012) Article DOI: 10.1124/dmd.112.045492 BindingDB Entry DOI: 10.7270/Q27H1M9T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||