 Found 495 hits of ki data for polymerid = 5309
Found 495 hits of ki data for polymerid = 5309 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50346873

(CHEMBL1797652)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7]-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](-[#8])=O |r| Show InChI InChI=1S/C94H173N37O28/c1-46(2)42-63(84(151)116-50(6)91(158)159)126-81(148)61(30-32-66(99)136)122-76(143)55(23-11-15-35-96)120-79(146)59(27-19-38-109-93(103)104)124-86(153)65-29-21-41-131(65)90(157)49(5)115-75(142)54(22-10-14-34-95)117-69(139)44-111-68(138)43-112-87(154)70(51(7)133)128-85(152)64(45-132)127-80(147)56(24-12-16-36-97)121-78(145)58(26-18-37-108-92(101)102)119-74(141)48(4)114-88(155)71(52(8)134)129-83(150)62(31-33-67(100)137)123-77(144)57(25-13-17-40-113-107)125-89(156)72(53(9)135)130-82(149)60(118-73(140)47(3)98)28-20-39-110-94(105)106/h46-65,70-72,113,132-135H,10-45,95-98,107H2,1-9H3,(H2,99,136)(H2,100,137)(H,111,138)(H,112,154)(H,114,155)(H,115,142)(H,116,151)(H,117,139)(H,118,140)(H,119,141)(H,120,146)(H,121,145)(H,122,143)(H,123,144)(H,124,153)(H,125,156)(H,126,148)(H,127,147)(H,128,152)(H,129,150)(H,130,149)(H,158,159)(H4,101,102,108)(H4,103,104,109)(H4,105,106,110)/t47-,48-,49-,50-,51+,52+,53+,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,70-,71-,72-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of LSD1 |
J Med Chem 54: 8236-50 (2011)
Article DOI: 10.1021/jm201048w
BindingDB Entry DOI: 10.7270/Q2D50NFR |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50346873

(CHEMBL1797652)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7]-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](-[#8])=O |r| Show InChI InChI=1S/C94H173N37O28/c1-46(2)42-63(84(151)116-50(6)91(158)159)126-81(148)61(30-32-66(99)136)122-76(143)55(23-11-15-35-96)120-79(146)59(27-19-38-109-93(103)104)124-86(153)65-29-21-41-131(65)90(157)49(5)115-75(142)54(22-10-14-34-95)117-69(139)44-111-68(138)43-112-87(154)70(51(7)133)128-85(152)64(45-132)127-80(147)56(24-12-16-36-97)121-78(145)58(26-18-37-108-92(101)102)119-74(141)48(4)114-88(155)71(52(8)134)129-83(150)62(31-33-67(100)137)123-77(144)57(25-13-17-40-113-107)125-89(156)72(53(9)135)130-82(149)60(118-73(140)47(3)98)28-20-39-110-94(105)106/h46-65,70-72,113,132-135H,10-45,95-98,107H2,1-9H3,(H2,99,136)(H2,100,137)(H,111,138)(H,112,154)(H,114,155)(H,115,142)(H,116,151)(H,117,139)(H,118,140)(H,119,141)(H,120,146)(H,121,145)(H,122,143)(H,123,144)(H,124,153)(H,125,156)(H,126,148)(H,127,147)(H,128,152)(H,129,150)(H,130,149)(H,158,159)(H4,101,102,108)(H4,103,104,109)(H4,105,106,110)/t47-,48-,49-,50-,51+,52+,53+,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,70-,71-,72-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina
Curated by ChEMBL
| Assay Description
Inhibition of recombinant LSD1 (178 to 831) (unknown origin) expressed in baculovirus infected insect Sf9 cells using diMeK4H3-21 as substrate by per... |
ACS Med Chem Lett 5: 29-33 (2014)
Article DOI: 10.1021/ml4002997
BindingDB Entry DOI: 10.7270/Q2HT2QS9 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50361478
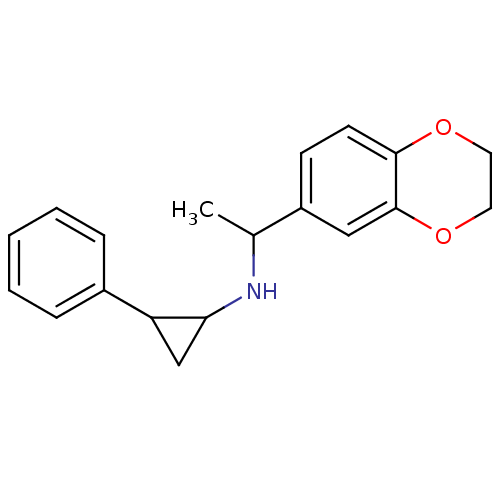
(CHEMBL1938897)Show InChI InChI=1S/C19H21NO2/c1-13(15-7-8-18-19(11-15)22-10-9-21-18)20-17-12-16(17)14-5-3-2-4-6-14/h2-8,11,13,16-17,20H,9-10,12H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of LSD1 |
J Med Chem 54: 8236-50 (2011)
Article DOI: 10.1021/jm201048w
BindingDB Entry DOI: 10.7270/Q2D50NFR |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50446141
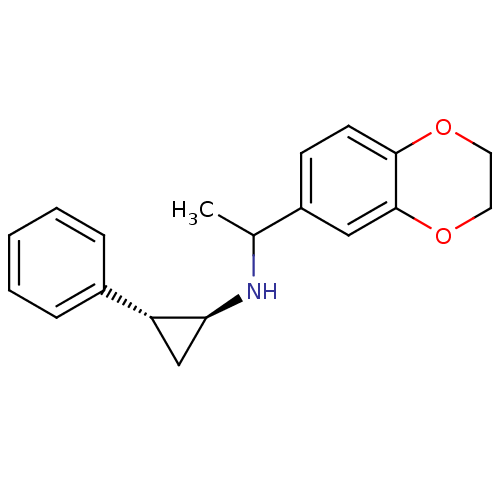
(CHEMBL3108901)Show SMILES CC(N[C@H]1C[C@@H]1c1ccccc1)c1ccc2OCCOc2c1 |r| Show InChI InChI=1S/C19H21NO2/c1-13(15-7-8-18-19(11-15)22-10-9-21-18)20-17-12-16(17)14-5-3-2-4-6-14/h2-8,11,13,16-17,20H,9-10,12H2,1H3/t13?,16-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant LSD1 using di-methylated H3-K4 peptide as substrate assessed as release of H2O2 preincubated for 15 mins followed by ... |
ACS Med Chem Lett 5: 29-33 (2014)
Article DOI: 10.1021/ml4002997
BindingDB Entry DOI: 10.7270/Q2HT2QS9 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50586369

(CHEMBL5094265)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human LSD1 using K4me2 peptide as substrate measured after 10 mins by peroxidase-coupled reaction assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01371
BindingDB Entry DOI: 10.7270/Q2KS6WFP |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM424912
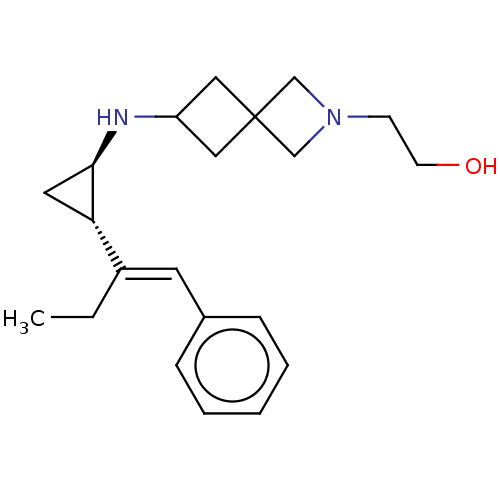
(2-(6-(((1R,2S)-2-((E)-1-phenylbut-1-en-2-yl)cyclop...)Show SMILES CC\C(=C/c1ccccc1)[C@@H]1C[C@H]1NC1CC2(C1)CN(CCO)C2 |r| Show InChI InChI=1S/C21H30N2O/c1-2-17(10-16-6-4-3-5-7-16)19-11-20(19)22-18-12-21(13-18)14-23(15-21)8-9-24/h3-7,10,18-20,22,24H,2,8-9,11-15H2,1H3/b17-10+/t19-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 6.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Constellation Pharmaceuticals
Curated by ChEMBL
| Assay Description
Time dependant inhibition of recombinant N-terminal His-tagged LSD1 (unknown origin) expressed in Escherichia coli expression system assessed as inhi... |
ACS Med Chem Lett 11: 1213-1220 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00060
BindingDB Entry DOI: 10.7270/Q22N55VZ |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50586368

(CHEMBL5073848)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(O)=O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human LSD1 using K4me2 peptide as substrate measured after 10 mins by peroxidase-coupled reaction assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01371
BindingDB Entry DOI: 10.7270/Q2KS6WFP |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101302
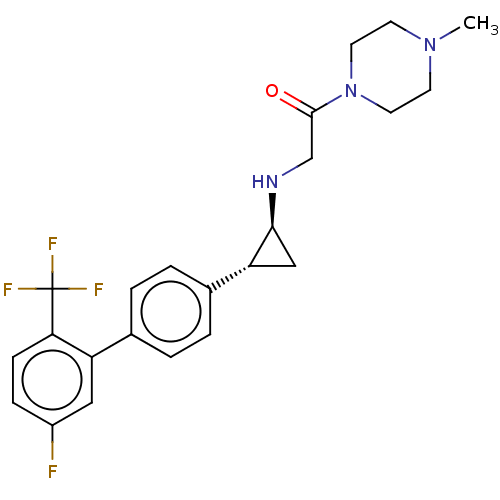
(CHEMBL3108900 | US8524717, 57)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1cc(F)ccc1C(F)(F)F |r| Show InChI InChI=1S/C23H25F4N3O/c1-29-8-10-30(11-9-29)22(31)14-28-21-13-19(21)16-4-2-15(3-5-16)18-12-17(24)6-7-20(18)23(25,26)27/h2-7,12,19,21,28H,8-11,13-14H2,1H3/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101302

(CHEMBL3108900 | US8524717, 57)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1cc(F)ccc1C(F)(F)F |r| Show InChI InChI=1S/C23H25F4N3O/c1-29-8-10-30(11-9-29)22(31)14-28-21-13-19(21)16-4-2-15(3-5-16)18-12-17(24)6-7-20(18)23(25,26)27/h2-7,12,19,21,28H,8-11,13-14H2,1H3/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant LSD1 using di-methylated H3-K4 peptide as substrate assessed as release of H2O2 preincubated for 15 mins followed by ... |
ACS Med Chem Lett 5: 29-33 (2014)
Article DOI: 10.1021/ml4002997
BindingDB Entry DOI: 10.7270/Q2HT2QS9 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50361481
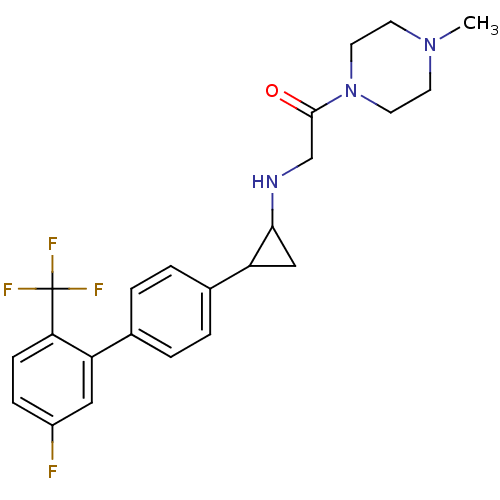
(CHEMBL1938898)Show SMILES CN1CCN(CC1)C(=O)CNC1CC1c1ccc(cc1)-c1cc(F)ccc1C(F)(F)F Show InChI InChI=1S/C23H25F4N3O/c1-29-8-10-30(11-9-29)22(31)14-28-21-13-19(21)16-4-2-15(3-5-16)18-12-17(24)6-7-20(18)23(25,26)27/h2-7,12,19,21,28H,8-11,13-14H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of LSD1 |
J Med Chem 54: 8236-50 (2011)
Article DOI: 10.1021/jm201048w
BindingDB Entry DOI: 10.7270/Q2D50NFR |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101261
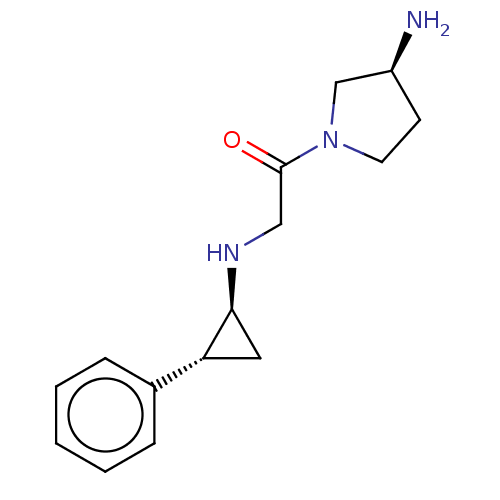
(US8524717, 16)Show SMILES N[C@H]1CCN(C1)C(=O)CN[C@H]1C[C@@H]1c1ccccc1 |r| Show InChI InChI=1S/C15H21N3O/c16-12-6-7-18(10-12)15(19)9-17-14-8-13(14)11-4-2-1-3-5-11/h1-5,12-14,17H,6-10,16H2/t12-,13+,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50158869
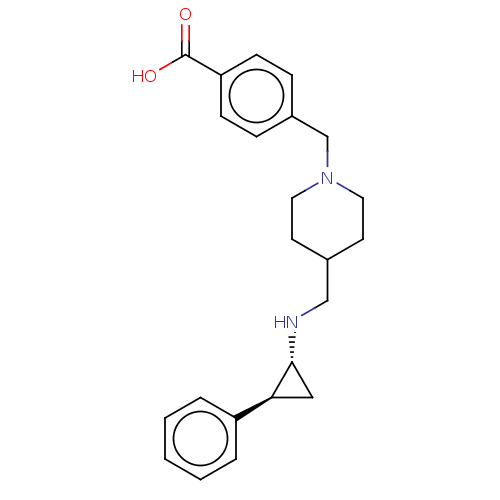
(CHEMBL3786182 | US10836743, Compound GSK-2879552 |...)Show SMILES OC(=O)c1ccc(CN2CCC(CN[C@@H]3C[C@H]3c3ccccc3)CC2)cc1 |r| Show InChI InChI=1S/C23H28N2O2/c26-23(27)20-8-6-18(7-9-20)16-25-12-10-17(11-13-25)15-24-22-14-21(22)19-4-2-1-3-5-19/h1-9,17,21-22,24H,10-16H2,(H,26,27)/t21-,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Constellation Pharmaceuticals
Curated by ChEMBL
| Assay Description
Time dependant inhibition of recombinant N-terminal His-tagged LSD1 (unknown origin) expressed in Escherichia coli expression system assessed as inhi... |
ACS Med Chem Lett 11: 1213-1220 (2020)
Article DOI: 10.1021/acsmedchemlett.0c00060
BindingDB Entry DOI: 10.7270/Q22N55VZ |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50597540
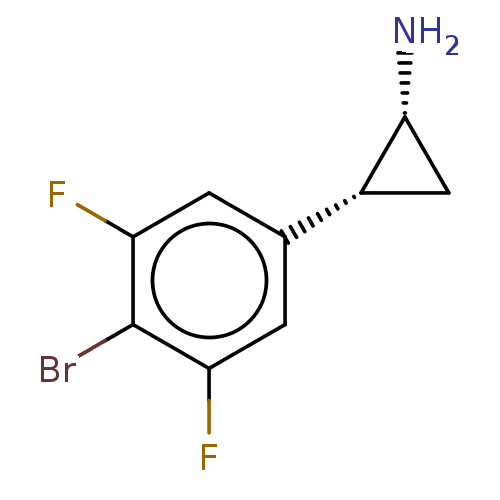
(CHEMBL5201156) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.2c00294
BindingDB Entry DOI: 10.7270/Q28S4TZ1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50597542

(CHEMBL5201621) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.2c00294
BindingDB Entry DOI: 10.7270/Q28S4TZ1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50445346
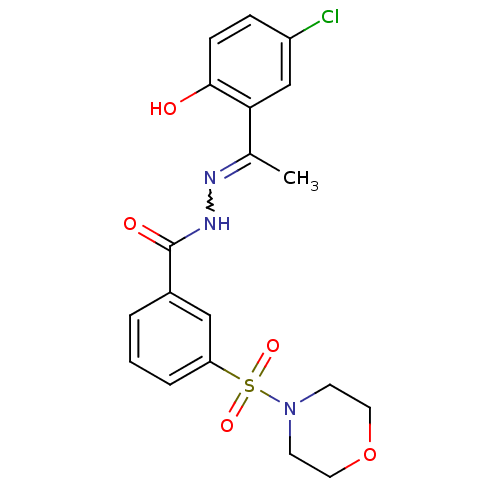
(CHEMBL3104250)Show SMILES CC(=NNC(=O)c1cccc(c1)S(=O)(=O)N1CCOCC1)c1cc(Cl)ccc1O |w:2.2| Show InChI InChI=1S/C19H20ClN3O5S/c1-13(17-12-15(20)5-6-18(17)24)21-22-19(25)14-3-2-4-16(11-14)29(26,27)23-7-9-28-10-8-23/h2-6,11-12,24H,7-10H2,1H3,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of recombinant full-length His6-tagged LSD1 (unknown origin) using dimethylated K4N-terminal H3 peptide as substrate at 1 ... |
J Med Chem 56: 9496-508 (2014)
Article DOI: 10.1021/jm400870h
BindingDB Entry DOI: 10.7270/Q2Z60QJ7 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101276
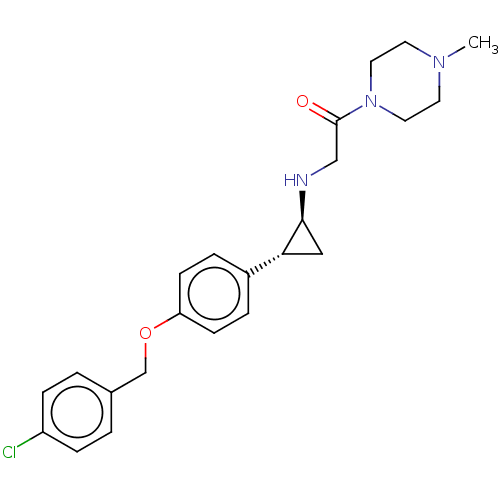
(US8524717, 31)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2ccc(Cl)cc2)cc1 |r| Show InChI InChI=1S/C23H28ClN3O2/c1-26-10-12-27(13-11-26)23(28)15-25-22-14-21(22)18-4-8-20(9-5-18)29-16-17-2-6-19(24)7-3-17/h2-9,21-22,25H,10-16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101278
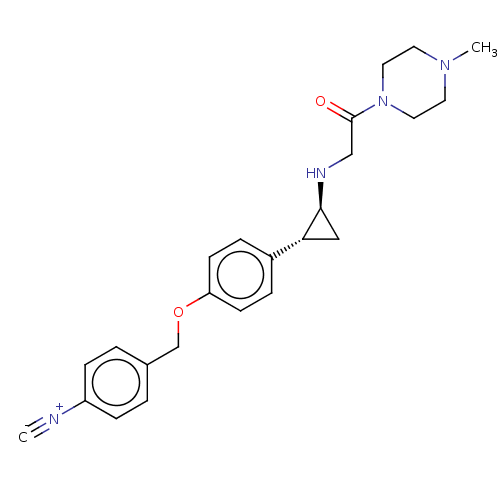
(US8524717, 33)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2ccc(cc2)[N+]#[C-])cc1 |r| Show InChI InChI=1S/C24H28N4O2/c1-25-20-7-3-18(4-8-20)17-30-21-9-5-19(6-10-21)22-15-23(22)26-16-24(29)28-13-11-27(2)12-14-28/h3-10,22-23,26H,11-17H2,2H3/t22-,23+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101305
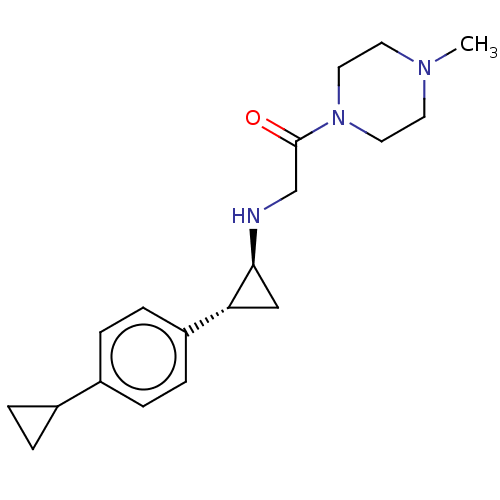
(US8524717, 60)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)C1CC1 |r| Show InChI InChI=1S/C19H27N3O/c1-21-8-10-22(11-9-21)19(23)13-20-18-12-17(18)16-6-4-15(5-7-16)14-2-3-14/h4-7,14,17-18,20H,2-3,8-13H2,1H3/t17-,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101273
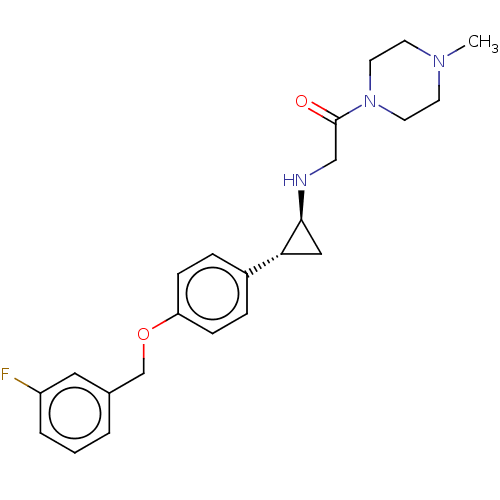
(US8524717, 28)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2cccc(F)c2)cc1 |r| Show InChI InChI=1S/C23H28FN3O2/c1-26-9-11-27(12-10-26)23(28)15-25-22-14-21(22)18-5-7-20(8-6-18)29-16-17-3-2-4-19(24)13-17/h2-8,13,21-22,25H,9-12,14-16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101300
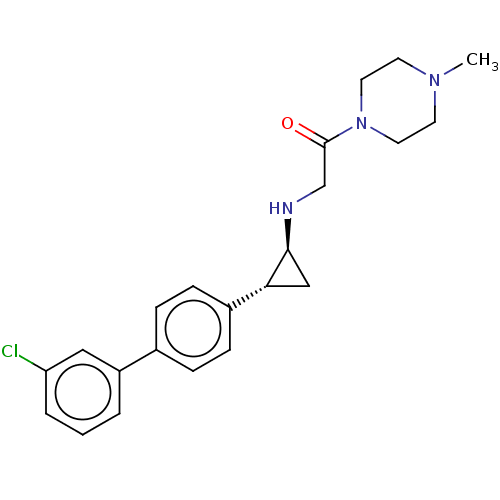
(US8524717, 55)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1cccc(Cl)c1 |r| Show InChI InChI=1S/C22H26ClN3O/c1-25-9-11-26(12-10-25)22(27)15-24-21-14-20(21)17-7-5-16(6-8-17)18-3-2-4-19(23)13-18/h2-8,13,20-21,24H,9-12,14-15H2,1H3/t20-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101274
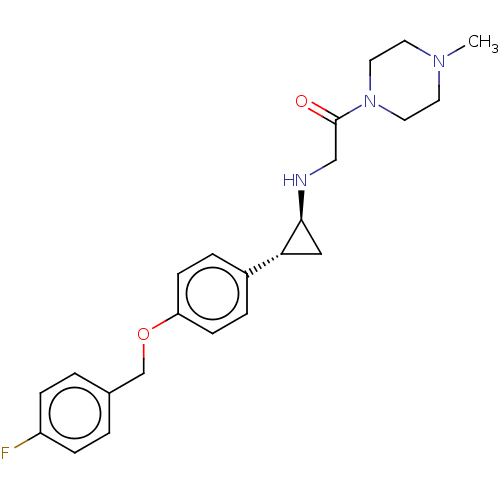
(US8524717, 29)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2ccc(F)cc2)cc1 |r| Show InChI InChI=1S/C23H28FN3O2/c1-26-10-12-27(13-11-26)23(28)15-25-22-14-21(22)18-4-8-20(9-5-18)29-16-17-2-6-19(24)7-3-17/h2-9,21-22,25H,10-16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101289
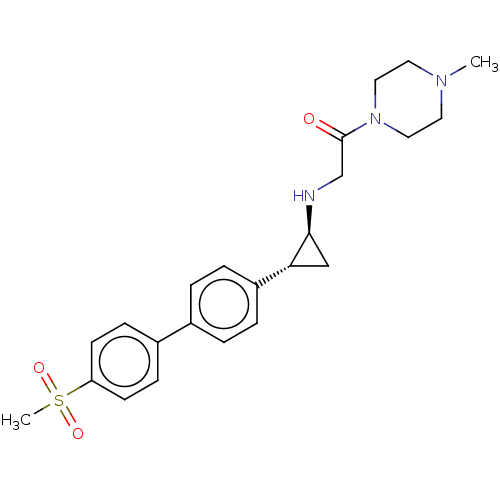
(US8524717, 44)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1ccc(cc1)S(C)(=O)=O |r| Show InChI InChI=1S/C23H29N3O3S/c1-25-11-13-26(14-12-25)23(27)16-24-22-15-21(22)19-5-3-17(4-6-19)18-7-9-20(10-8-18)30(2,28)29/h3-10,21-22,24H,11-16H2,1-2H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101291
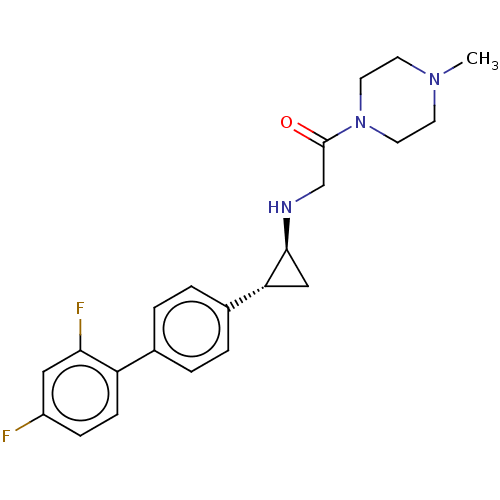
(US8524717, 46)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1ccc(F)cc1F |r| Show InChI InChI=1S/C22H25F2N3O/c1-26-8-10-27(11-9-26)22(28)14-25-21-13-19(21)16-4-2-15(3-5-16)18-7-6-17(23)12-20(18)24/h2-7,12,19,21,25H,8-11,13-14H2,1H3/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101283
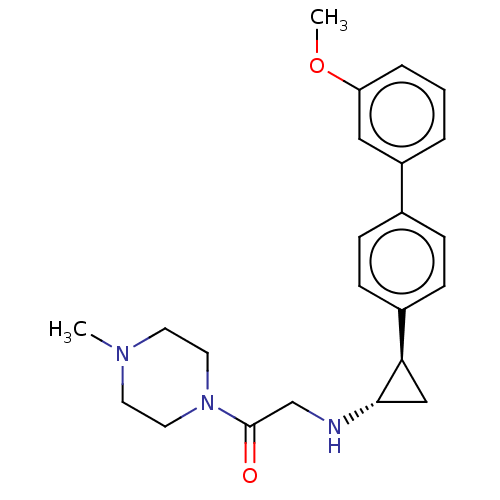
(US8524717, 38)Show SMILES COc1cccc(c1)-c1ccc(cc1)[C@H]1C[C@@H]1NCC(=O)N1CCN(C)CC1 |r| Show InChI InChI=1S/C23H29N3O2/c1-25-10-12-26(13-11-25)23(27)16-24-22-15-21(22)18-8-6-17(7-9-18)19-4-3-5-20(14-19)28-2/h3-9,14,21-22,24H,10-13,15-16H2,1-2H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101277
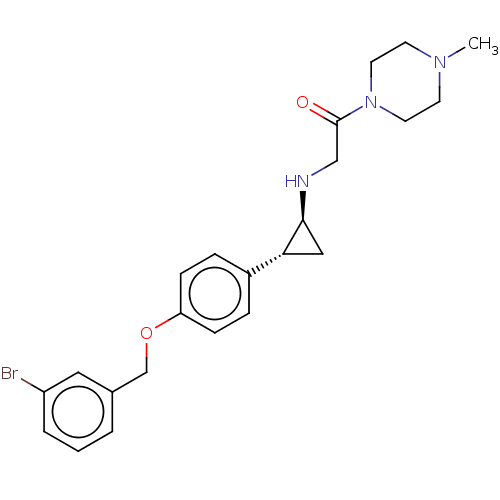
(US8524717, 32)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2cccc(Br)c2)cc1 |r| Show InChI InChI=1S/C23H28BrN3O2/c1-26-9-11-27(12-10-26)23(28)15-25-22-14-21(22)18-5-7-20(8-6-18)29-16-17-3-2-4-19(24)13-17/h2-8,13,21-22,25H,9-12,14-16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101304
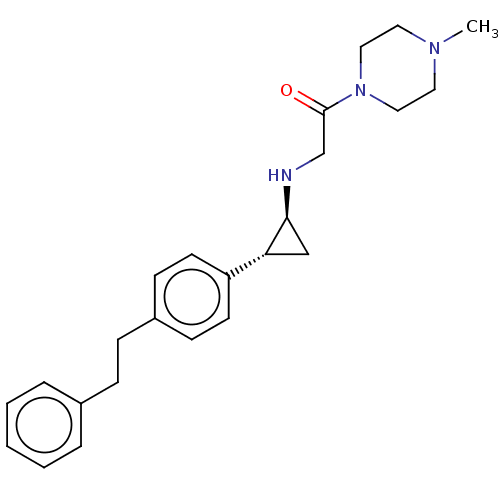
(US8524717, 59)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(CCc2ccccc2)cc1 |r| Show InChI InChI=1S/C24H31N3O/c1-26-13-15-27(16-14-26)24(28)18-25-23-17-22(23)21-11-9-20(10-12-21)8-7-19-5-3-2-4-6-19/h2-6,9-12,22-23,25H,7-8,13-18H2,1H3/t22-,23+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101290
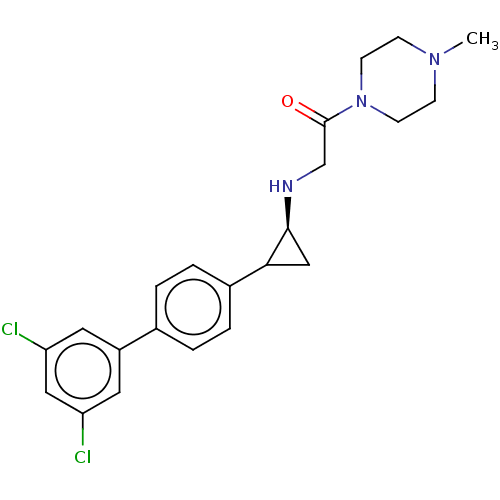
(US8524717, 45)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1CC1c1ccc(cc1)-c1cc(Cl)cc(Cl)c1 |r| Show InChI InChI=1S/C22H25Cl2N3O/c1-26-6-8-27(9-7-26)22(28)14-25-21-13-20(21)16-4-2-15(3-5-16)17-10-18(23)12-19(24)11-17/h2-5,10-12,20-21,25H,6-9,13-14H2,1H3/t20?,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101285
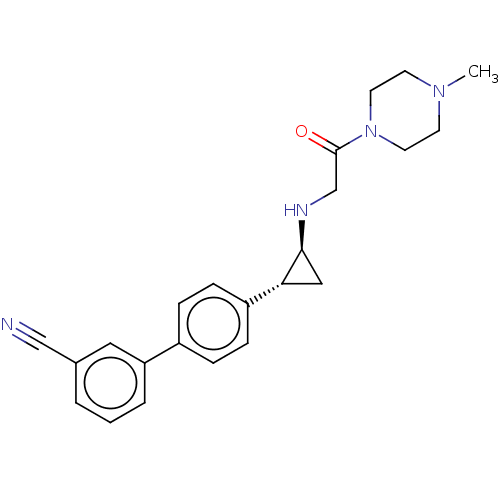
(US8524717, 40)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1cccc(c1)C#N |r| Show InChI InChI=1S/C23H26N4O/c1-26-9-11-27(12-10-26)23(28)16-25-22-14-21(22)19-7-5-18(6-8-19)20-4-2-3-17(13-20)15-24/h2-8,13,21-22,25H,9-12,14,16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50597543

(CHEMBL5182939) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.2c00294
BindingDB Entry DOI: 10.7270/Q28S4TZ1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101286
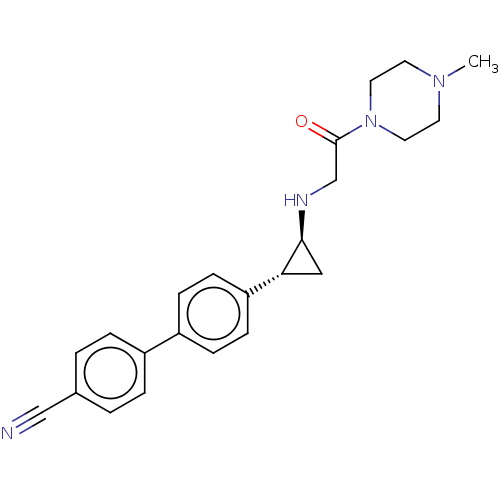
(US8524717, 41)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1ccc(cc1)C#N |r| Show InChI InChI=1S/C23H26N4O/c1-26-10-12-27(13-11-26)23(28)16-25-22-14-21(22)20-8-6-19(7-9-20)18-4-2-17(15-24)3-5-18/h2-9,21-22,25H,10-14,16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50142238
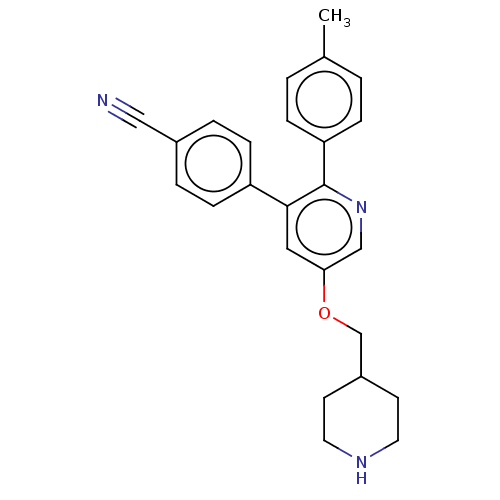
(CHEMBL3759201)Show SMILES Cc1ccc(cc1)-c1ncc(OCC2CCNCC2)cc1-c1ccc(cc1)C#N Show InChI InChI=1S/C25H25N3O/c1-18-2-6-22(7-3-18)25-24(21-8-4-19(15-26)5-9-21)14-23(16-28-25)29-17-20-10-12-27-13-11-20/h2-9,14,16,20,27H,10-13,17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhengzhou University
Curated by ChEMBL
| Assay Description
Dissociation constant against galectin-3 using competitive fluorescence polarization |
Eur J Med Chem 125: 940-951 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.021
BindingDB Entry DOI: 10.7270/Q2ZC8597 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101293
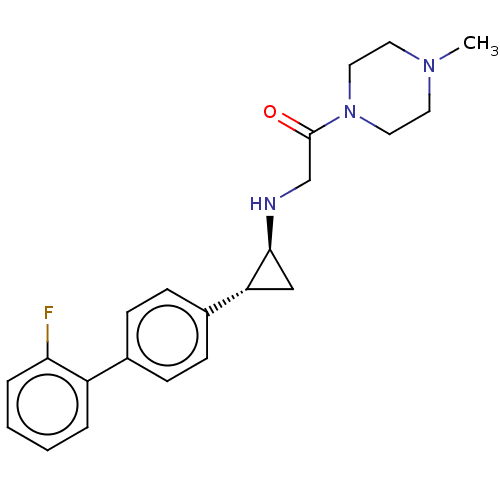
(US8524717, 48)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1ccccc1F |r| Show InChI InChI=1S/C22H26FN3O/c1-25-10-12-26(13-11-25)22(27)15-24-21-14-19(21)17-8-6-16(7-9-17)18-4-2-3-5-20(18)23/h2-9,19,21,24H,10-15H2,1H3/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50142238

(CHEMBL3759201)Show SMILES Cc1ccc(cc1)-c1ncc(OCC2CCNCC2)cc1-c1ccc(cc1)C#N Show InChI InChI=1S/C25H25N3O/c1-18-2-6-22(7-3-18)25-24(21-8-4-19(15-26)5-9-21)14-23(16-28-25)29-17-20-10-12-27-13-11-20/h2-9,14,16,20,27H,10-13,17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Baylor College of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human GST-tagged LSD1 catalytic domain (172 to 833 residues) using dimethylated H3K4 peptide substrate preincubated for 10 ... |
J Med Chem 59: 253-63 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01361
BindingDB Entry DOI: 10.7270/Q23J3FSW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240130
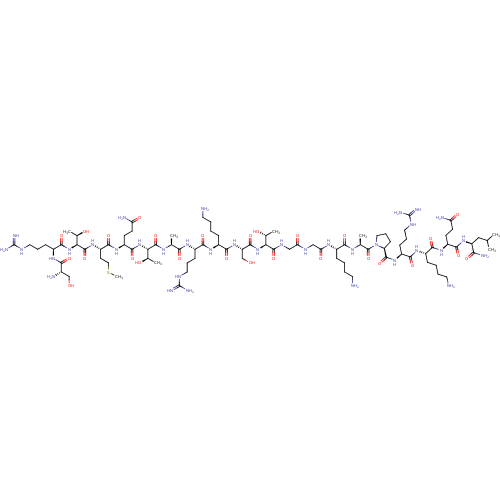
(CHEMBL4060961)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C90H165N35O27S/c1-44(2)39-60(70(97)135)120-78(143)57(26-28-63(95)131)116-74(139)52(20-10-13-32-92)114-76(141)55(23-16-35-105-89(100)101)118-83(148)62-25-18-37-125(62)87(152)46(4)110-73(138)51(19-9-12-31-91)111-66(134)41-107-65(133)40-108-84(149)67(47(5)128)122-82(147)61(43-127)121-77(142)53(21-11-14-33-93)115-75(140)54(22-15-34-104-88(98)99)112-71(136)45(3)109-85(150)68(48(6)129)123-81(146)58(27-29-64(96)132)117-79(144)59(30-38-153-8)119-86(151)69(49(7)130)124-80(145)56(24-17-36-106-90(102)103)113-72(137)50(94)42-126/h44-62,67-69,126-130H,9-43,91-94H2,1-8H3,(H2,95,131)(H2,96,132)(H2,97,135)(H,107,133)(H,108,149)(H,109,150)(H,110,138)(H,111,134)(H,112,136)(H,113,137)(H,114,141)(H,115,140)(H,116,139)(H,117,144)(H,118,148)(H,119,151)(H,120,143)(H,121,142)(H,122,147)(H,123,146)(H,124,145)(H4,98,99,104)(H4,100,101,105)(H4,102,103,106)/t45-,46-,47+,48+,49+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,67-,68-,69-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101284
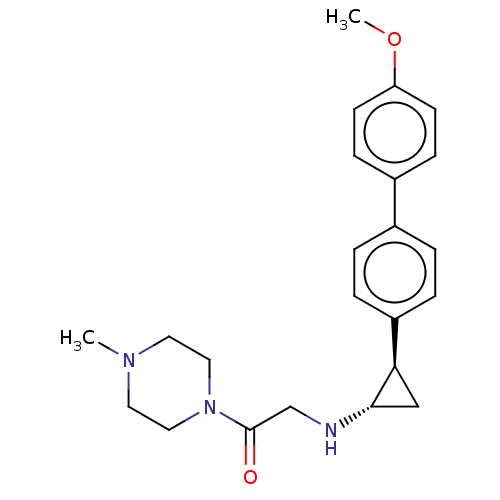
(US8524717, 39)Show SMILES COc1ccc(cc1)-c1ccc(cc1)[C@H]1C[C@@H]1NCC(=O)N1CCN(C)CC1 |r| Show InChI InChI=1S/C23H29N3O2/c1-25-11-13-26(14-12-25)23(27)16-24-22-15-21(22)19-5-3-17(4-6-19)18-7-9-20(28-2)10-8-18/h3-10,21-22,24H,11-16H2,1-2H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101271
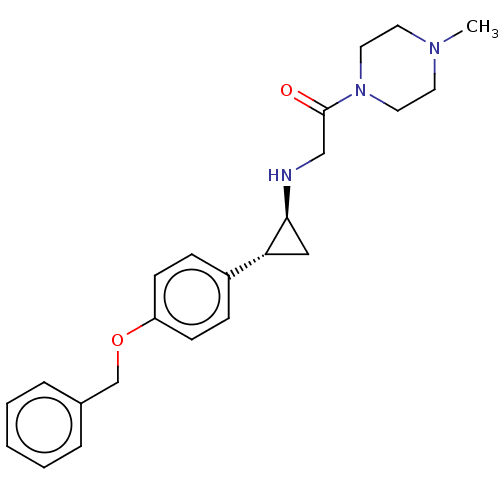
(US8524717, 26)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2ccccc2)cc1 |r| Show InChI InChI=1S/C23H29N3O2/c1-25-11-13-26(14-12-25)23(27)16-24-22-15-21(22)19-7-9-20(10-8-19)28-17-18-5-3-2-4-6-18/h2-10,21-22,24H,11-17H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| US Patent
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50445346

(CHEMBL3104250)Show SMILES CC(=NNC(=O)c1cccc(c1)S(=O)(=O)N1CCOCC1)c1cc(Cl)ccc1O |w:2.2| Show InChI InChI=1S/C19H20ClN3O5S/c1-13(17-12-15(20)5-6-18(17)24)21-22-19(25)14-3-2-4-16(11-14)29(26,27)23-7-9-28-10-8-23/h2-6,11-12,24H,7-10H2,1H3,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of recombinant full-length His6-tagged LSD1 (unknown origin) using dimethylated K4N-terminal H3 peptide as substrate at 10... |
J Med Chem 56: 9496-508 (2014)
Article DOI: 10.1021/jm400870h
BindingDB Entry DOI: 10.7270/Q2Z60QJ7 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50445346

(CHEMBL3104250)Show SMILES CC(=NNC(=O)c1cccc(c1)S(=O)(=O)N1CCOCC1)c1cc(Cl)ccc1O |w:2.2| Show InChI InChI=1S/C19H20ClN3O5S/c1-13(17-12-15(20)5-6-18(17)24)21-22-19(25)14-3-2-4-16(11-14)29(26,27)23-7-9-28-10-8-23/h2-6,11-12,24H,7-10H2,1H3,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of recombinant full-length His6-tagged LSD1 (unknown origin) using dimethylated K4N-terminal H3 peptide as substrate at 30... |
J Med Chem 56: 9496-508 (2014)
Article DOI: 10.1021/jm400870h
BindingDB Entry DOI: 10.7270/Q2Z60QJ7 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50445346

(CHEMBL3104250)Show SMILES CC(=NNC(=O)c1cccc(c1)S(=O)(=O)N1CCOCC1)c1cc(Cl)ccc1O |w:2.2| Show InChI InChI=1S/C19H20ClN3O5S/c1-13(17-12-15(20)5-6-18(17)24)21-22-19(25)14-3-2-4-16(11-14)29(26,27)23-7-9-28-10-8-23/h2-6,11-12,24H,7-10H2,1H3,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of recombinant full-length His6-tagged LSD1 (unknown origin) using dimethylated K4N-terminal H3 peptide as substrate at 3 ... |
J Med Chem 56: 9496-508 (2014)
Article DOI: 10.1021/jm400870h
BindingDB Entry DOI: 10.7270/Q2Z60QJ7 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50346870
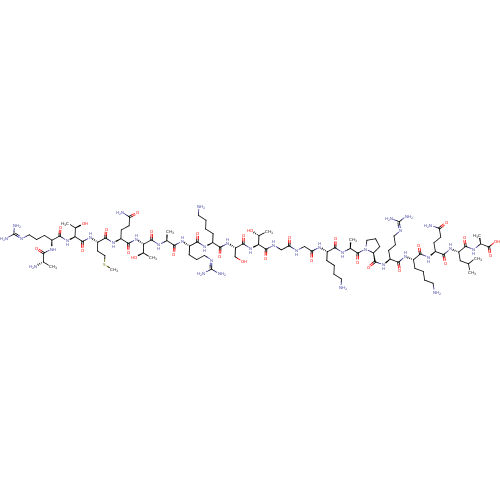
(CHEMBL1797647)Show SMILES [#6]-[#16]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](-[#8])=O |r| Show InChI InChI=1S/C93H169N35O28S/c1-45(2)41-62(83(148)113-49(6)90(155)156)123-79(144)59(28-30-65(98)133)119-75(140)54(22-12-15-34-95)117-77(142)57(25-18-37-107-92(102)103)121-85(150)64-27-20-39-128(64)89(154)48(5)112-74(139)53(21-11-14-33-94)114-68(136)43-109-67(135)42-110-86(151)69(50(7)130)125-84(149)63(44-129)124-78(143)55(23-13-16-35-96)118-76(141)56(24-17-36-106-91(100)101)116-73(138)47(4)111-87(152)70(51(8)131)126-82(147)60(29-31-66(99)134)120-80(145)61(32-40-157-10)122-88(153)71(52(9)132)127-81(146)58(115-72(137)46(3)97)26-19-38-108-93(104)105/h45-64,69-71,129-132H,11-44,94-97H2,1-10H3,(H2,98,133)(H2,99,134)(H,109,135)(H,110,151)(H,111,152)(H,112,139)(H,113,148)(H,114,136)(H,115,137)(H,116,138)(H,117,142)(H,118,141)(H,119,140)(H,120,145)(H,121,150)(H,122,153)(H,123,144)(H,124,143)(H,125,149)(H,126,147)(H,127,146)(H,155,156)(H4,100,101,106)(H4,102,103,107)(H4,104,105,108)/t46-,47-,48-,49-,50+,51+,52+,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human LSD1 expressed in Escherichia coli using methylated H3-K4 peptide as substrate by peroxidase-coupled assay |
ACS Med Chem Lett 5: 29-33 (2014)
Article DOI: 10.1021/ml4002997
BindingDB Entry DOI: 10.7270/Q2HT2QS9 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240124
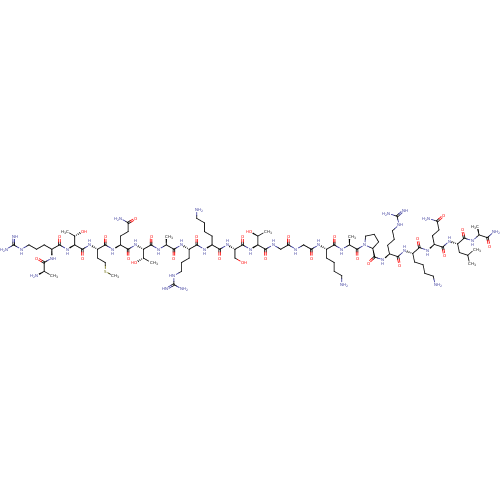
(CHEMBL4090728)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](C)N)[C@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(N)=O |r| Show InChI InChI=1S/C93H170N36O27S/c1-45(2)41-62(84(150)112-47(4)72(100)138)124-80(146)59(28-30-65(98)134)120-76(142)54(22-12-15-34-95)118-78(144)57(25-18-37-108-92(103)104)122-86(152)64-27-20-39-129(64)90(156)49(6)114-75(141)53(21-11-14-33-94)115-68(137)43-110-67(136)42-111-87(153)69(50(7)131)126-85(151)63(44-130)125-79(145)55(23-13-16-35-96)119-77(143)56(24-17-36-107-91(101)102)117-74(140)48(5)113-88(154)70(51(8)132)127-83(149)60(29-31-66(99)135)121-81(147)61(32-40-157-10)123-89(155)71(52(9)133)128-82(148)58(116-73(139)46(3)97)26-19-38-109-93(105)106/h45-64,69-71,130-133H,11-44,94-97H2,1-10H3,(H2,98,134)(H2,99,135)(H2,100,138)(H,110,136)(H,111,153)(H,112,150)(H,113,154)(H,114,141)(H,115,137)(H,116,139)(H,117,140)(H,118,144)(H,119,143)(H,120,142)(H,121,147)(H,122,152)(H,123,155)(H,124,146)(H,125,145)(H,126,151)(H,127,149)(H,128,148)(H4,101,102,107)(H4,103,104,108)(H4,105,106,109)/t46-,47+,48+,49+,50+,51+,52+,53+,54+,55+,56+,57+,58+,59+,60+,61+,62+,63+,64+,69+,70+,71+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using pLys4Met H3 peptide as substrate by peroxidase coupled UV-visible spectrophot... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50346870

(CHEMBL1797647)Show SMILES [#6]-[#16]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](-[#8])=O |r| Show InChI InChI=1S/C93H169N35O28S/c1-45(2)41-62(83(148)113-49(6)90(155)156)123-79(144)59(28-30-65(98)133)119-75(140)54(22-12-15-34-95)117-77(142)57(25-18-37-107-92(102)103)121-85(150)64-27-20-39-128(64)89(154)48(5)112-74(139)53(21-11-14-33-94)114-68(136)43-109-67(135)42-110-86(151)69(50(7)130)125-84(149)63(44-129)124-78(143)55(23-13-16-35-96)118-76(141)56(24-17-36-106-91(100)101)116-73(138)47(4)111-87(152)70(51(8)131)126-82(147)60(29-31-66(99)134)120-80(145)61(32-40-157-10)122-88(153)71(52(9)132)127-81(146)58(115-72(137)46(3)97)26-19-38-108-93(104)105/h45-64,69-71,129-132H,11-44,94-97H2,1-10H3,(H2,98,133)(H2,99,134)(H,109,135)(H,110,151)(H,111,152)(H,112,139)(H,113,148)(H,114,136)(H,115,137)(H,116,138)(H,117,142)(H,118,141)(H,119,140)(H,120,145)(H,121,150)(H,122,153)(H,123,144)(H,124,143)(H,125,149)(H,126,147)(H,127,146)(H,155,156)(H4,100,101,106)(H4,102,103,107)(H4,104,105,108)/t46-,47-,48-,49-,50+,51+,52+,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant KDM1A expressed in Escherichia coli using H3K4me as substrate by peroxidase coupled enzyme assay |
J Med Chem 59: 1308-29 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01758
BindingDB Entry DOI: 10.7270/Q2MP5559 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101297
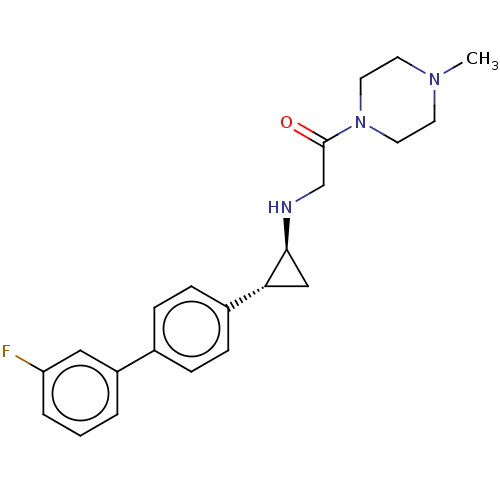
(US8524717, 52)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1cccc(F)c1 |r| Show InChI InChI=1S/C22H26FN3O/c1-25-9-11-26(12-10-25)22(27)15-24-21-14-20(21)17-7-5-16(6-8-17)18-3-2-4-19(23)13-18/h2-8,13,20-21,24H,9-12,14-15H2,1H3/t20-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101257
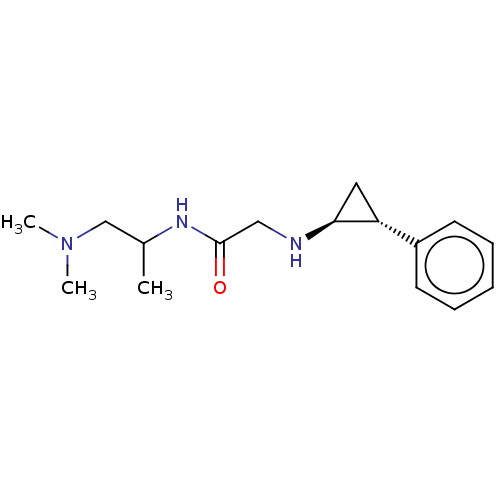
(US8524717, 12)Show SMILES CC(CN(C)C)NC(=O)CN[C@H]1C[C@@H]1c1ccccc1 |r| Show InChI InChI=1S/C16H25N3O/c1-12(11-19(2)3)18-16(20)10-17-15-9-14(15)13-7-5-4-6-8-13/h4-8,12,14-15,17H,9-11H2,1-3H3,(H,18,20)/t12?,14-,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101287
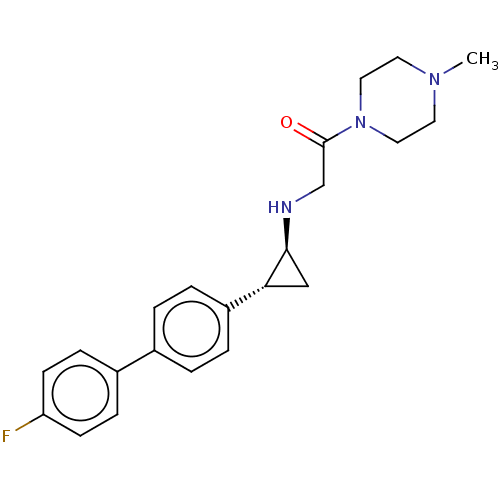
(US8524717, 42)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1ccc(F)cc1 |r| Show InChI InChI=1S/C22H26FN3O/c1-25-10-12-26(13-11-25)22(27)15-24-21-14-20(21)18-4-2-16(3-5-18)17-6-8-19(23)9-7-17/h2-9,20-21,24H,10-15H2,1H3/t20-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101282

(US8524717, 37)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(cc1)-c1cccnc1 |r| Show InChI InChI=1S/C21H26N4O/c1-24-9-11-25(12-10-24)21(26)15-23-20-13-19(20)17-6-4-16(5-7-17)18-3-2-8-22-14-18/h2-8,14,19-20,23H,9-13,15H2,1H3/t19-,20+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50142187

(CHEMBL3758634)Show SMILES FC(F)(F)Oc1ccc(cc1)-c1ncc(OCC2CCNCC2)cc1-c1ccc(cc1)C#N Show InChI InChI=1S/C25H22F3N3O2/c26-25(27,28)33-21-7-5-20(6-8-21)24-23(19-3-1-17(14-29)2-4-19)13-22(15-31-24)32-16-18-9-11-30-12-10-18/h1-8,13,15,18,30H,9-12,16H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 46 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Baylor College of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human GST-tagged LSD1 catalytic domain (172 to 833 residues) using dimethylated H3K4 peptide substrate preincubated for 10 ... |
J Med Chem 59: 253-63 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01361
BindingDB Entry DOI: 10.7270/Q23J3FSW |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101303
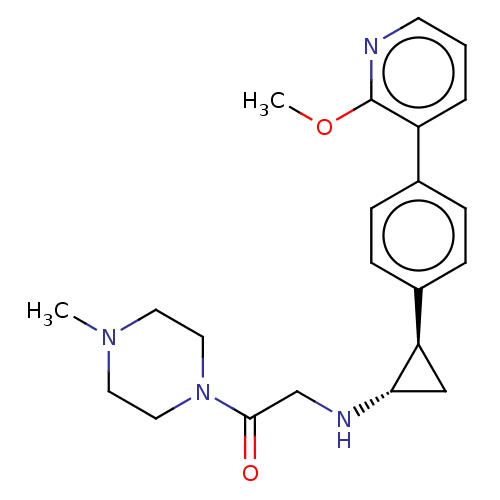
(US8524717, 58)Show SMILES COc1ncccc1-c1ccc(cc1)[C@H]1C[C@@H]1NCC(=O)N1CCN(C)CC1 |r| Show InChI InChI=1S/C22H28N4O2/c1-25-10-12-26(13-11-25)21(27)15-24-20-14-19(20)17-7-5-16(6-8-17)18-4-3-9-23-22(18)28-2/h3-9,19-20,24H,10-15H2,1-2H3/t19-,20+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 47 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM101275
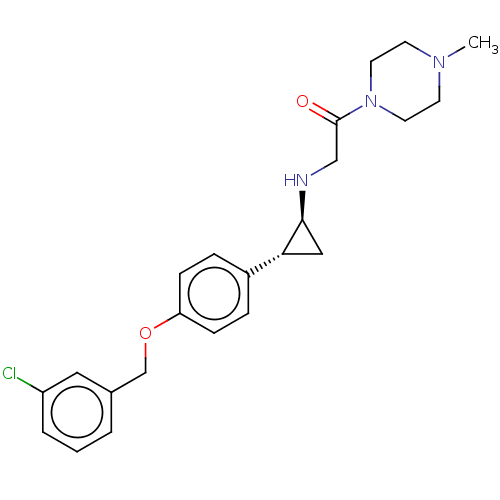
(US8524717, 30)Show SMILES CN1CCN(CC1)C(=O)CN[C@H]1C[C@@H]1c1ccc(OCc2cccc(Cl)c2)cc1 |r| Show InChI InChI=1S/C23H28ClN3O2/c1-26-9-11-27(12-10-26)23(28)15-25-22-14-21(22)18-5-7-20(8-6-18)29-16-17-3-2-4-19(24)13-17/h2-8,13,21-22,25H,9-12,14-16H2,1H3/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 47 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics, S.A.
US Patent
| Assay Description
Biological assay using monoamine oxidase or LSD1. |
US Patent US8524717 (2013)
BindingDB Entry DOI: 10.7270/Q26H4G1D |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM179840
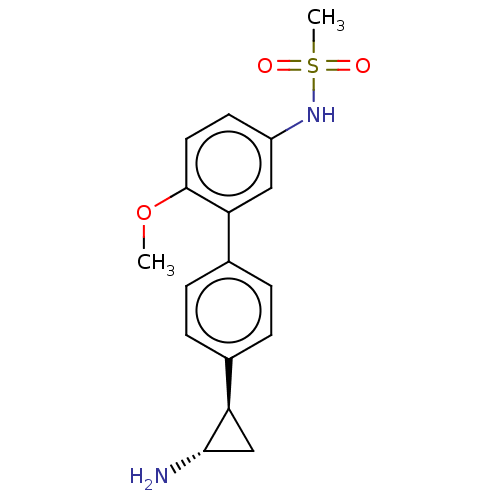
(US9676701, 75 N-(4′-((trans)-2-aminocyclopro...)Show SMILES COc1ccc(NS(C)(=O)=O)cc1-c1ccc(cc1)[C@H]1C[C@@H]1N |r| Show InChI InChI=1S/C17H20N2O3S/c1-22-17-8-7-13(19-23(2,20)21)9-15(17)12-5-3-11(4-6-12)14-10-16(14)18/h3-9,14,16,19H,10,18H2,1-2H3/t14-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 50 | -9.95 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS, S.A.
US Patent
| Assay Description
Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of various concentrations of inhibitor (e.... |
US Patent US9676701 (2017)
BindingDB Entry DOI: 10.7270/Q27P8WJN |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data