

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-lactamase TEM (Escherichia coli) | BDBM178163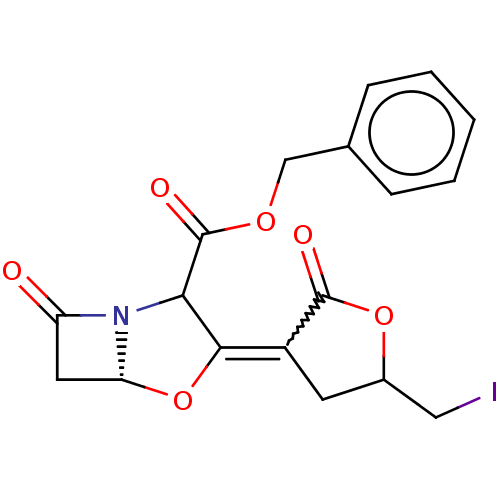 (US9120808, Example 41 | US9120808, Example 42) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178167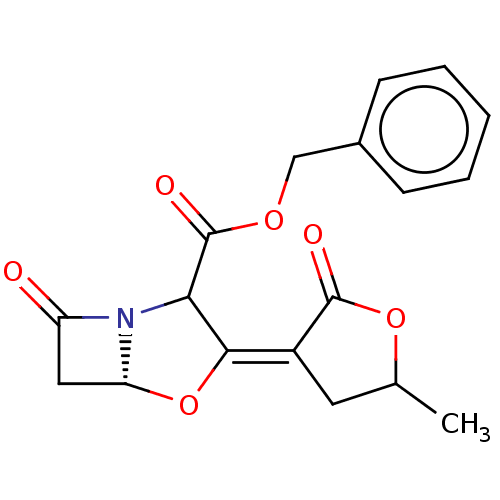 (US9120808, Example 46 | US9120808, Example 47) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50149468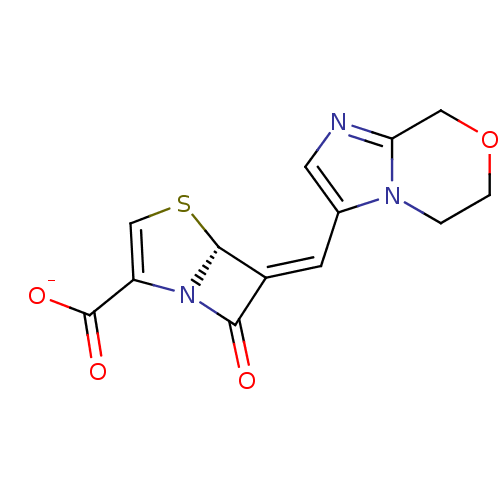 (CHEMBL263746 | Sodium; (R)-6-[1-(5,6-dihydro-8H-im...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description In vitro inhibitory activity against Class A (TEM-1) beta-Lactamases | J Med Chem 47: 3674-88 (2004) Article DOI: 10.1021/jm049903j BindingDB Entry DOI: 10.7270/Q2MK6CCT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191378 (CHEMBL212163 | sodium (R,E)-6-((6,8-dihydro-5H-imi...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM467000 (2-(((2S,5R)-2-carbamoyl-4-methyl-7-oxo-1,6-diazabi...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191390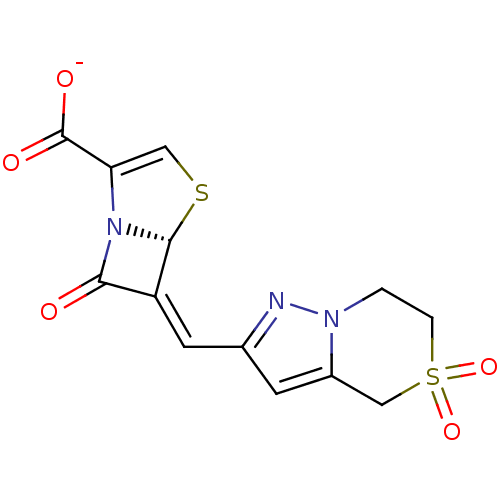 ((5R),(6Z)-6-(5,5-dioxo-4,5,6,7-tetrahydro-5'6-pyra...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191389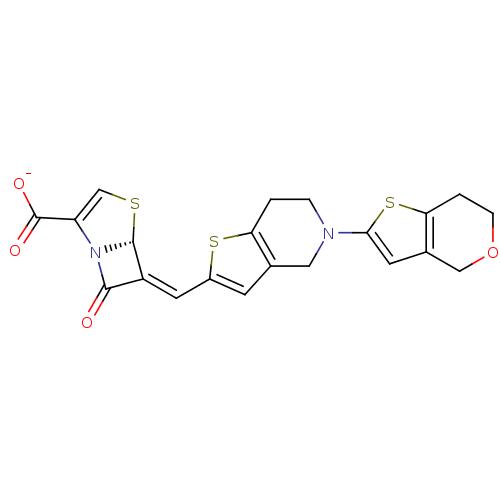 (6-(6,7-dihydro-4H-thieno[3,2-c]pyran-2-ylmethylene...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191379 ((5R)(6Z)-6-(6,7-dihydro-4H-pyrazolo[5,1-c][1,4]-th...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191377 ((5R),(6Z)-7-oxo-6-(4,5,6,7-tetrahydropyrazolo[1,5-...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50149467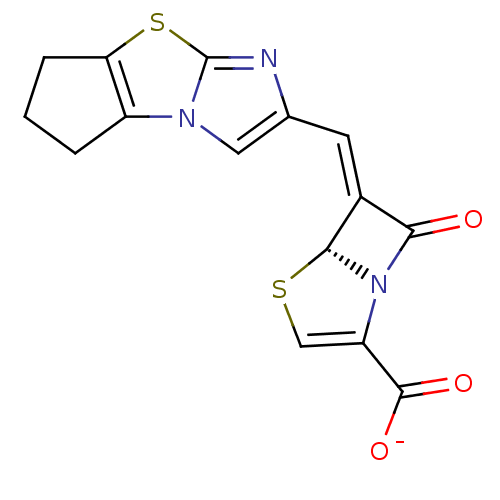 ((5R,6Z)-6-(6,7-dihydro-5H-cyclopenta-[d]imidazo[2,...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 47: 6556-68 (2004) Article DOI: 10.1021/jm049680x BindingDB Entry DOI: 10.7270/Q2542N22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50149466 (CHEMBL124416 | Sodium; (R)-6-[1-(5,6-dihydro-4H-py...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description In vitro inhibitory activity against Class A (TEM-1) beta-Lactamases | J Med Chem 47: 3674-88 (2004) Article DOI: 10.1021/jm049903j BindingDB Entry DOI: 10.7270/Q2MK6CCT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50149467 ((5R,6Z)-6-(6,7-dihydro-5H-cyclopenta-[d]imidazo[2,...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description In vitro inhibitory activity against Class A (TEM-1) beta-Lactamases | J Med Chem 47: 3674-88 (2004) Article DOI: 10.1021/jm049903j BindingDB Entry DOI: 10.7270/Q2MK6CCT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM467003 (US10800778, Comparator 98) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM466965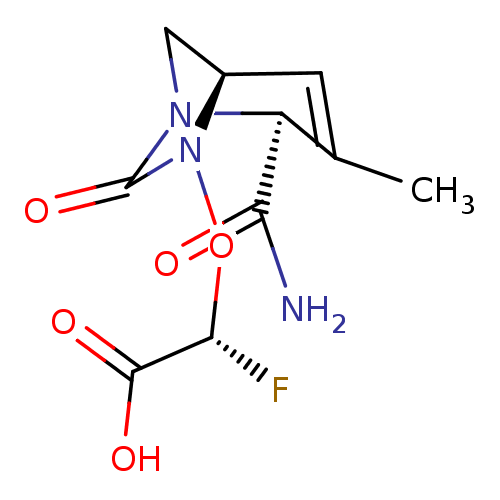 ((2R)-{[(2S,5R)-2-carbamoyl-3-methyl-7-oxo-1,6-diaz...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.35 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178215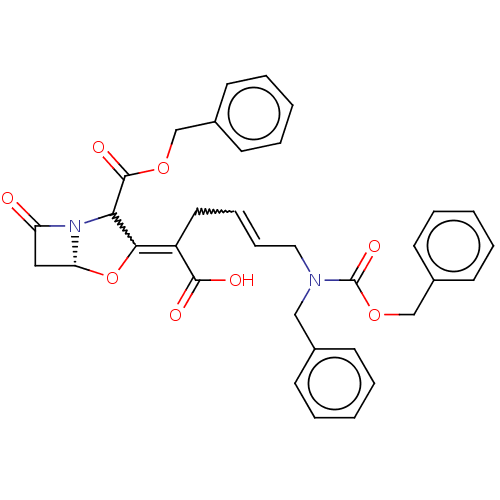 (US9120808, Example 58) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM466967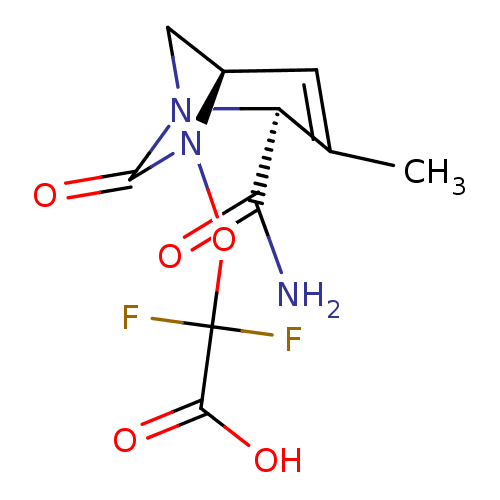 (US10800778, Example 8 | {[(2S,5R)-2-carbamoyl-3-me...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178167 (US9120808, Example 46 | US9120808, Example 47) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178163 (US9120808, Example 41 | US9120808, Example 42) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178170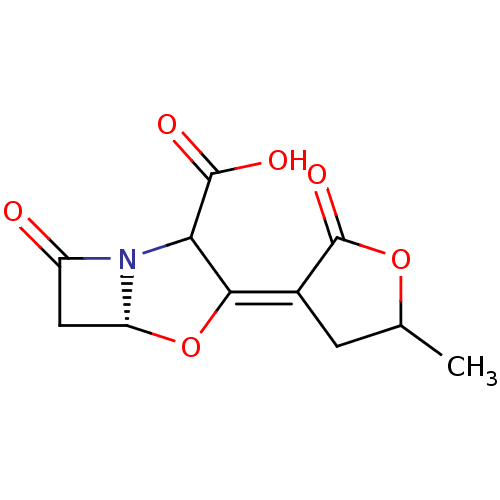 (US9120808, Example 49) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50339145 (CHEMBL1689063 | trans-7-oxo-6-(sulfooxy)-1,6-diaza...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of bacterial Beta-lactamase TEM-1 (24 - 286) (unknown origin) expressed in Escherichia coli Transetta (DE3) | Citation and Details Article DOI: 10.1016/j.bmcl.2021.127956 BindingDB Entry DOI: 10.7270/Q2B85CX1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191386 ((5R)(6Z)-7-oxo-6-(4,5,6,7-tetrahydropyrazolo-[1,5-...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191381 ((5R,6Z)-6-{[5-(4-methoxybenzyl)-4,5,6,7-tetrahydro...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178169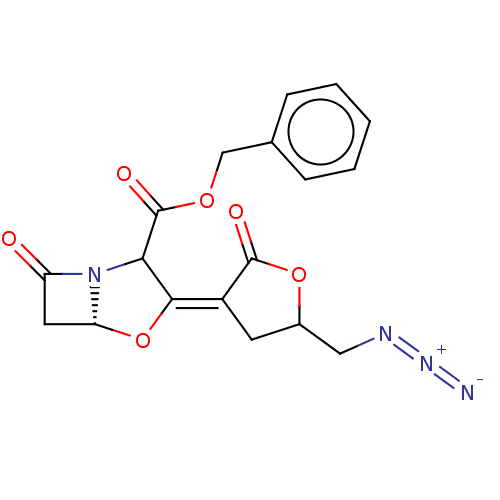 (US9120808, Example 48) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50284790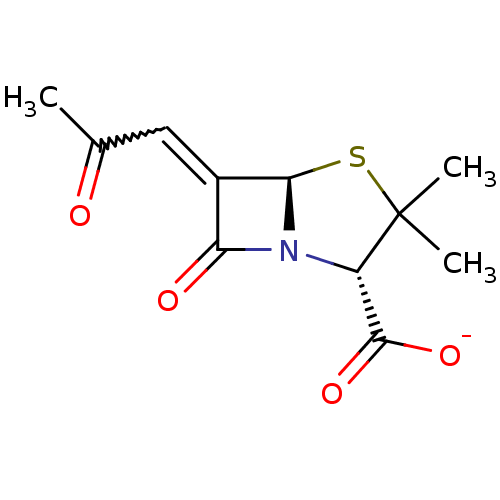 (CHEMBL35987 | Sodium; (2S,5R)-3,3-dimethyl-7-oxo-6...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against beta-Lactamase enzyme derived from Escherichia coli WC3310 TEM-2 | Bioorg Med Chem Lett 5: 1513-1518 (1995) Article DOI: 10.1016/0960-894X(95)00249-S BindingDB Entry DOI: 10.7270/Q2ZW1MDG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50157688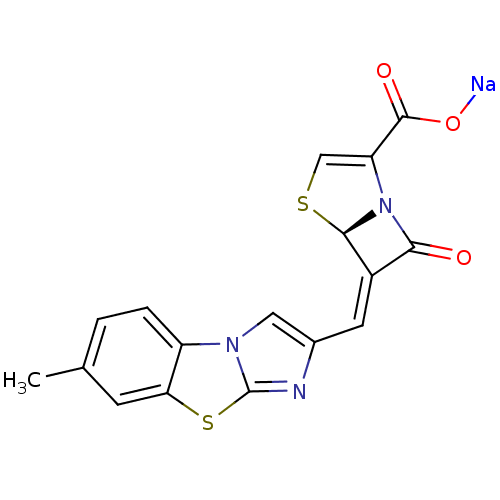 ((5R,6Z)-6-[(7-methylimidazo[2,1-b][1,3]-benzothiaz...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 47: 6556-68 (2004) Article DOI: 10.1021/jm049680x BindingDB Entry DOI: 10.7270/Q2542N22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191383 ((5R),(6Z)-6-(5,6-dihydro-8H-imidazo[2,1-c]-[1,4]th...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50505403 (CHEMBL4533632) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford Curated by ChEMBL | Assay Description Inhibition of recombinant bacterial class A serine beta-lactamase TEM-1 expressed in Escherichia coli assessed as reduction in breakdown of cephalosp... | J Med Chem 62: 8544-8556 (2019) Article DOI: 10.1021/acs.jmedchem.9b00911 BindingDB Entry DOI: 10.7270/Q2P272DC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM466972 ((2R)-2-(((2S,5R)-2-carbamoyl-4-methyl-7-oxo-1,6-di...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178165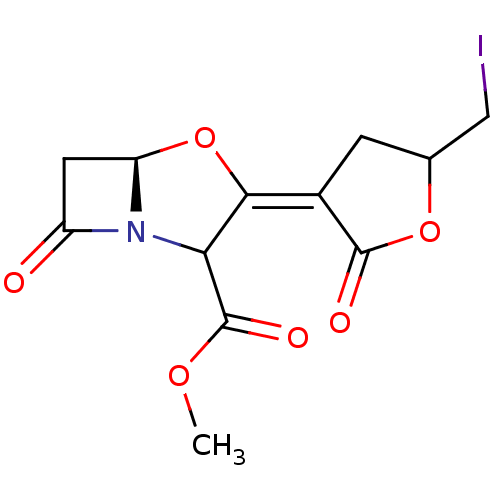 (US9120808, Mixture of Example 43 and Example 44) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM466974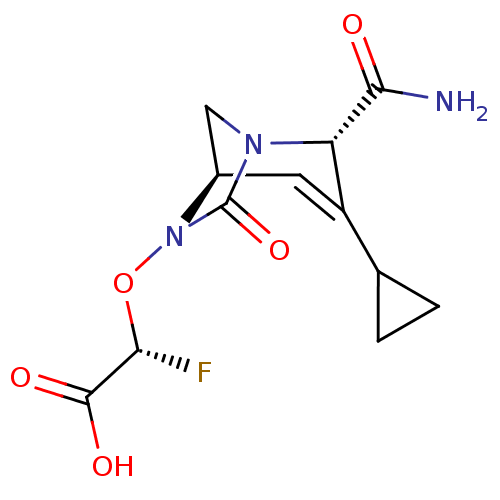 ((2R)-2-(((2S,5R)-2-carbamoyl-3-cyclopropyl-7-oxo-1...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191387 ((5R),(6Z)-6-(7-methyl-5,6,7,8-tetrahydroimidazo[1,...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191385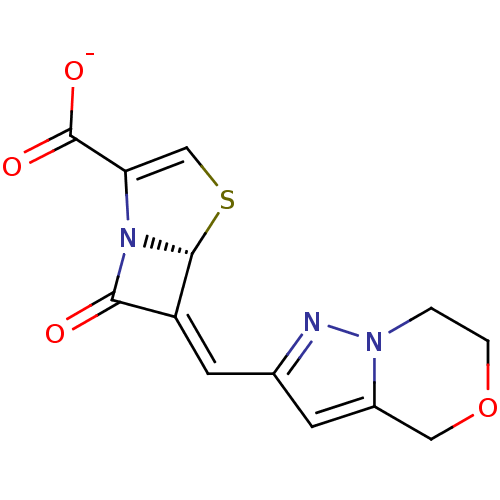 ((5R,6Z)-6-(6,7-dihydro-4H-pyrazolo[5,1-c]-[1,4]oxa...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178171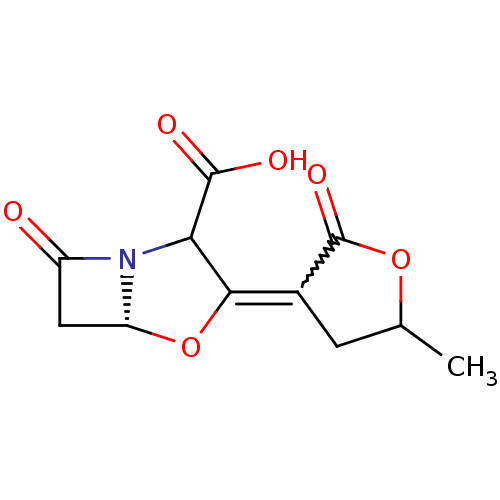 (US9120808, Example 50) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM466966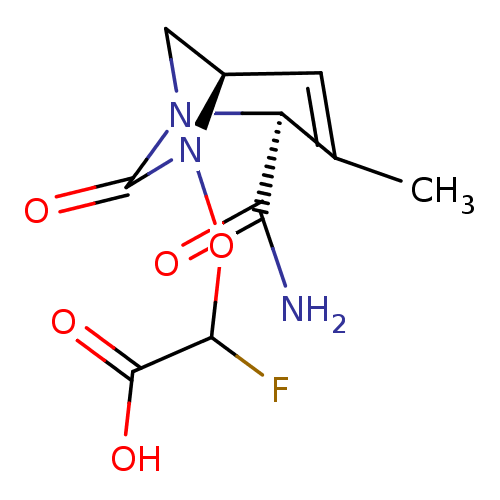 (US10800778, Example 5 | {[(2S,5R)-2-carbamoyl-3-me...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Entasis Therapeutics Limited US Patent | Assay Description A buffer consisting of 0.1 M sodium phosphate (pH 7.0), 10 mM NaHCO3, and 0.005% Triton X-100 was used for all enzymes. The chromogenic substrate nit... | US Patent US10800778 (2020) BindingDB Entry DOI: 10.7270/Q2DF6V9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191380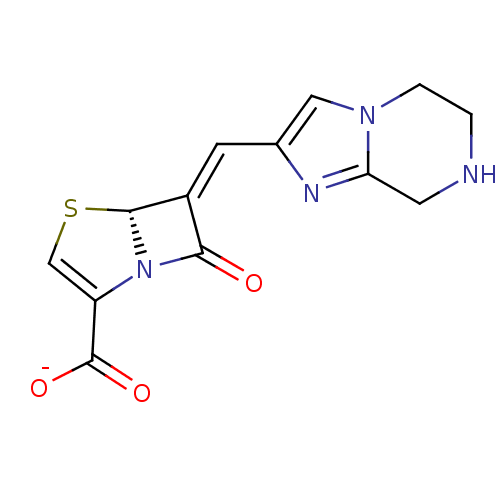 (CHEMBL379440 | sodium (R,E)-7-oxo-6-((5,6,7,8-tetr...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50033680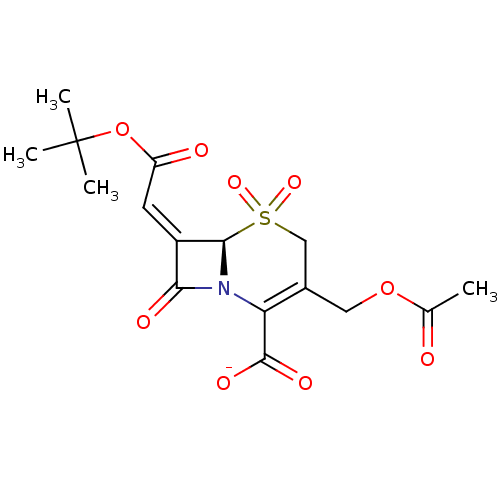 (CHEMBL268919 | Sodium; (R)-3-acetoxymethyl-7-[1-te...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Methodist University Curated by ChEMBL | Assay Description The concentration of compound to inhibit beta-lactamase was measured on E. coli WC3310 | J Med Chem 38: 1022-34 (1995) BindingDB Entry DOI: 10.7270/Q29S1Q29 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50033680 (CHEMBL268919 | Sodium; (R)-3-acetoxymethyl-7-[1-te...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against beta-Lactamase enzyme derived from Escherichia coli WC3310 TEM-2 | Bioorg Med Chem Lett 5: 1513-1518 (1995) Article DOI: 10.1016/0960-894X(95)00249-S BindingDB Entry DOI: 10.7270/Q2ZW1MDG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50157693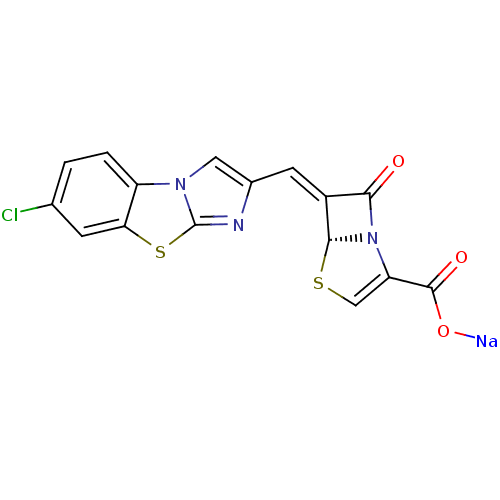 ((5R,6Z)-6-[(7-chloroimidazo[2,1-b][1,3]-benzothiaz...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 47: 6556-68 (2004) Article DOI: 10.1021/jm049680x BindingDB Entry DOI: 10.7270/Q2542N22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191388 ((5R)(6Z)-6-(6,7-5H-dihydropyrazolo[5,1-b]-oxazin-2...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50076683 (CHEMBL6533 | Sodium; (2S,3S,5R,6R)-6-hydroxymethyl...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research Curated by ChEMBL | Assay Description Inhibitory activity of the compound against TEM-1 (class A) beta-lactamase | Bioorg Med Chem Lett 9: 997-1002 (1999) BindingDB Entry DOI: 10.7270/Q2PK0FBJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50076678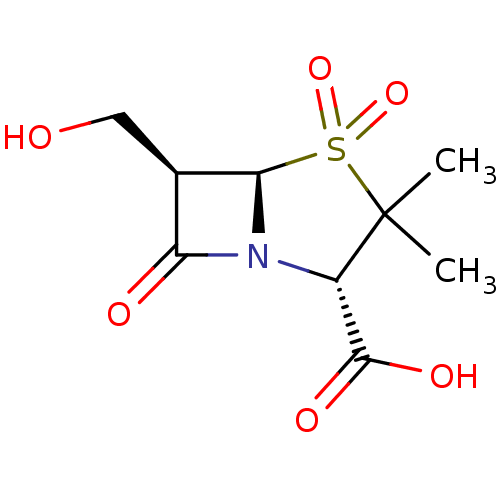 ((2S,5R,6R)-6-Hydroxymethyl-3,3-dimethyl-4,4,7-trio...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research Curated by ChEMBL | Assay Description Inhibitory activity of the compound against TEM-1 (class A) beta-lactamase | Bioorg Med Chem Lett 9: 991-6 (1999) BindingDB Entry DOI: 10.7270/Q2TB163Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50157694 ((5R,6Z)-6-[(7-fluoroimidazo[2,1-b][1,3]-benzothiaz...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 47: 6556-68 (2004) Article DOI: 10.1021/jm049680x BindingDB Entry DOI: 10.7270/Q2542N22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50157687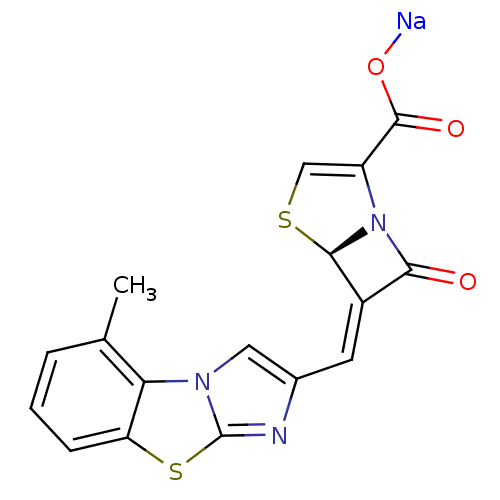 ((5R,6Z)-6-[(5-methylimidazo[2,1-b][1,3]-benzothiaz...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 47: 6556-68 (2004) Article DOI: 10.1021/jm049680x BindingDB Entry DOI: 10.7270/Q2542N22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178201 (US9120808, Example 26) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 9.20 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50157689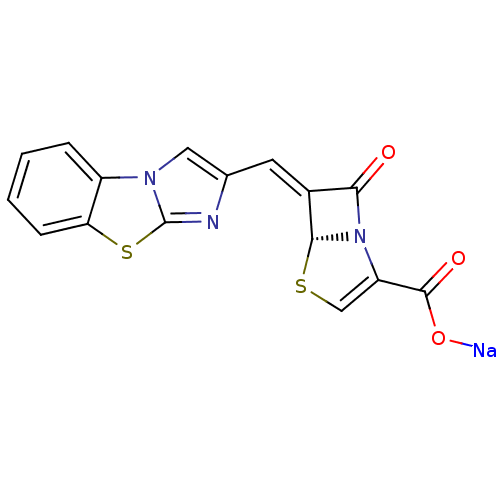 ((5R,6Z)-6-(imidazo[2,1-b][1,3]benzothiazol-2-ylmet...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 47: 6556-68 (2004) Article DOI: 10.1021/jm049680x BindingDB Entry DOI: 10.7270/Q2542N22 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50191382 ((5R,6Z)-7-oxo-6-{[5-(pyridin-3-ylmethyl)-4,5,6,7-t...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 49: 4623-37 (2006) Article DOI: 10.1021/jm060021p BindingDB Entry DOI: 10.7270/Q2TX3F1B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50212634 (CHEMBL309009) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | n/a | n/a | 10.5 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibitory activity against Escherichia coli HA58R Class-III TEM-1 type beta-lactamase enzyme | Citation and Details BindingDB Entry DOI: 10.7270/Q2ZW1P2T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM178214 (US9120808, Example 57) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 10.6 | n/a | n/a | n/a | n/a | 7.0 | 37 |
NABRIVA THERAPEUTICS AG US Patent | Assay Description The inhibitory concentrations (IC50, [uM]) of the beta -lactamase inhibitors against purified TEM-1, SHV-1 and AmpC beta -lactamases are assessed by ... | US Patent US9120808 (2015) BindingDB Entry DOI: 10.7270/Q2Q81BT5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50218932 (CHEMBL396998 | sodium (8R,9R)-10(S)-[1(R)-hydroxye...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Lek Pharmaceuticals d.d. Curated by ChEMBL | Assay Description Inhibition of Escherichia coli TEM1 | J Med Chem 50: 4113-21 (2007) Article DOI: 10.1021/jm0703237 BindingDB Entry DOI: 10.7270/Q2FF3T5F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-lactamase TEM (Escherichia coli) | BDBM50212641 (Brobactam) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | n/a | n/a | 14.3 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Assay Description Inhibitory activity against Escherichia coli HA58R Class-III TEM-1 type beta-lactamase enzyme | Citation and Details BindingDB Entry DOI: 10.7270/Q2ZW1P2T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 316 total ) | Next | Last >> |