 Found 390 hits of ec50 for UniProtKB: P33032
Found 390 hits of ec50 for UniProtKB: P33032 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50121261
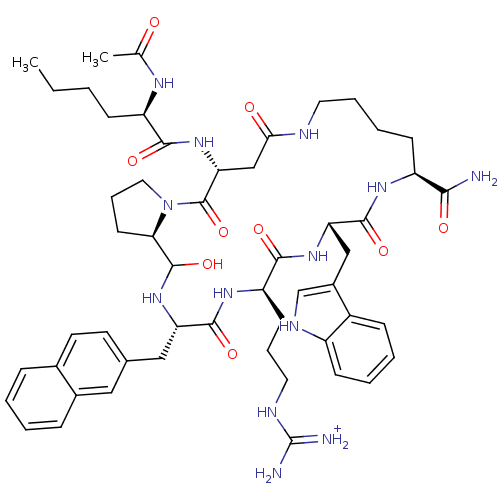
(5-(2-Acetylamino-hexanoylamino)-19-[3-(diaminometh...)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](CCCNC(N)=[NH2+])NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(O)[C@H]2CCCN2C1=O)C(N)=O Show InChI InChI=1S/C53H73N13O9/c1-3-4-16-39(60-31(2)67)47(70)65-43-29-45(68)57-23-10-9-18-38(46(54)69)61-50(73)42(28-35-30-59-37-17-8-7-15-36(35)37)63-48(71)40(19-11-24-58-53(55)56)62-49(72)41(64-51(74)44-20-12-25-66(44)52(43)75)27-32-21-22-33-13-5-6-14-34(33)26-32/h5-8,13-15,17,21-22,26,30,38-44,51,59,64,74H,3-4,9-12,16,18-20,23-25,27-29H2,1-2H3,(H2,54,69)(H,57,68)(H,60,67)(H,61,73)(H,62,72)(H,63,71)(H,65,70)(H4,55,56,58)/p+1/t38-,39+,40+,41-,42-,43+,44+,51?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Binding affinity against human Melanocortin 5 receptor |
J Med Chem 45: 5287-94 (2002)
BindingDB Entry DOI: 10.7270/Q20G3KW8 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211392
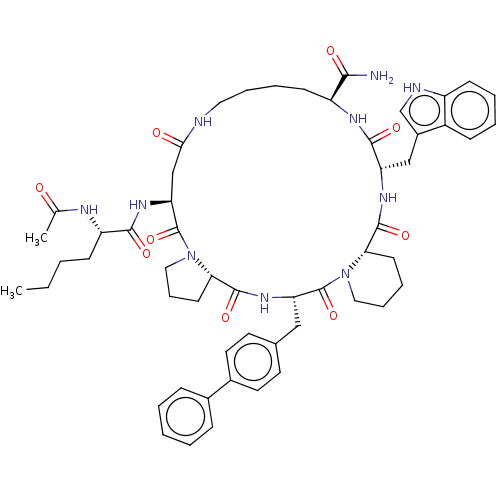
(Ac-Nle4-cyclo(Asp5-Pro6-D-4,4'-Bip7-Pro8-Trp9-Lys1...)Show SMILES [H][C@@]12CCCN1C(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC(=O)[C@]1([H])CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O Show InChI InChI=1S/C54H68N10O9/c1-3-4-17-41(58-33(2)65)49(68)61-44-31-47(66)56-26-11-10-19-40(48(55)67)59-50(69)42(30-37-32-57-39-18-9-8-16-38(37)39)60-51(70)45-20-12-27-63(45)53(72)43(62-52(71)46-21-13-28-64(46)54(44)73)29-34-22-24-36(25-23-34)35-14-6-5-7-15-35/h5-9,14-16,18,22-25,32,40-46,57H,3-4,10-13,17,19-21,26-31H2,1-2H3,(H2,55,67)(H,56,66)(H,58,65)(H,59,69)(H,60,70)(H,61,68)(H,62,71)/t40?,41-,42-,43-,44-,45-,46-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0310 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50121267
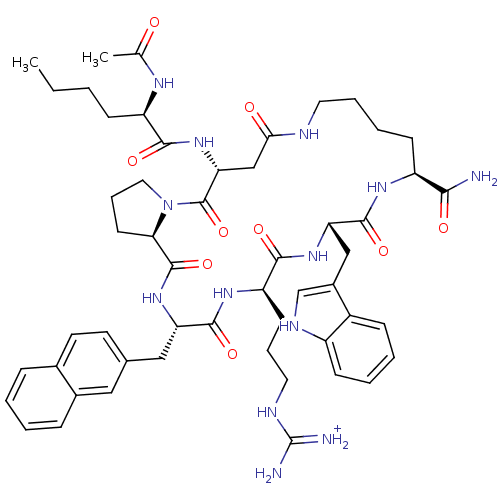
(5-(2-Acetylamino-hexanoylamino)-19-[3-(diaminometh...)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](CCCNC(N)=[NH2+])NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H]2CCCN2C1=O)C(N)=O Show InChI InChI=1S/C53H71N13O9/c1-3-4-16-39(60-31(2)67)47(70)65-43-29-45(68)57-23-10-9-18-38(46(54)69)61-50(73)42(28-35-30-59-37-17-8-7-15-36(35)37)63-48(71)40(19-11-24-58-53(55)56)62-49(72)41(64-51(74)44-20-12-25-66(44)52(43)75)27-32-21-22-33-13-5-6-14-34(33)26-32/h5-8,13-15,17,21-22,26,30,38-44,59H,3-4,9-12,16,18-20,23-25,27-29H2,1-2H3,(H2,54,69)(H,57,68)(H,60,67)(H,61,73)(H,62,72)(H,63,71)(H,64,74)(H,65,70)(H4,55,56,58)/p+1/t38-,39+,40+,41-,42-,43+,44+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Binding affinity against human Melanocortin 5 receptor |
J Med Chem 45: 5287-94 (2002)
BindingDB Entry DOI: 10.7270/Q20G3KW8 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50565811
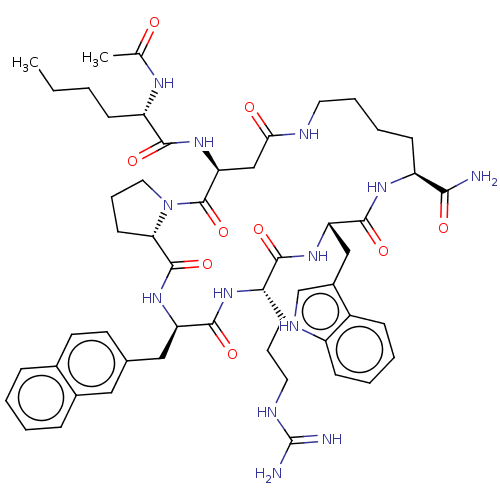
(CHEMBL4792986)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@@H]2CCCN2C1=O)C(N)=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | n/a | n/a | 0.0720 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268805
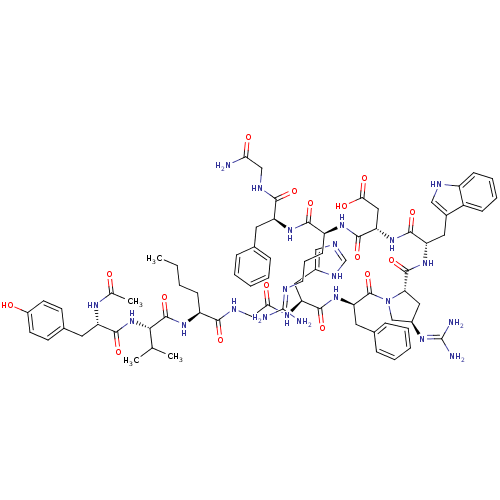
(Ac-Tyr-Val-Nle-Gly-His-DPhe-trans-Xaa-Trp-Asp-Arg-...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N1C[C@@H](C[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O)N=C(N)N |r,wU:8.8,45.46,59.63,96.101,35.35,77.82,wD:4.4,57.117,12.21,63.66,85.90,(3.89,-26.77,;2.57,-27.55,;1.22,-26.79,;-.1,-27.57,;-1.45,-26.81,;-2.78,-27.6,;-2.76,-29.14,;-1.41,-29.89,;-4.09,-29.92,;-4.07,-31.46,;-5.4,-32.25,;-6.73,-31.49,;-5.38,-33.78,;-4.04,-34.54,;-2.72,-33.75,;-2.73,-32.22,;-1.4,-31.43,;-.06,-32.19,;1.27,-31.4,;-.04,-33.74,;-1.37,-34.52,;-6.71,-34.57,;-6.71,-36.11,;-8.02,-36.89,;-5.35,-36.86,;-5.43,-29.17,;-5.44,-27.63,;-6.74,-29.95,;-1.46,-25.28,;-2.8,-24.52,;-.14,-24.49,;-.16,-22.95,;1.17,-22.16,;2.52,-22.92,;1.16,-20.62,;2.48,-19.84,;2.46,-18.3,;1.13,-17.55,;.96,-16.02,;-.56,-15.71,;-1.31,-17.05,;-.27,-18.18,;3.82,-20.6,;3.85,-22.14,;5.15,-19.82,;6.5,-20.57,;6.51,-22.12,;7.85,-22.87,;7.87,-24.42,;9.21,-25.18,;10.53,-24.39,;10.51,-22.85,;9.17,-22.09,;7.86,-19.86,;9.11,-20.76,;7.93,-18.32,;6.9,-17.17,;7.69,-15.84,;9.19,-16.18,;9.34,-17.71,;10.66,-18.5,;10.65,-20.04,;12,-17.74,;13.32,-18.54,;13.31,-20.08,;14.59,-20.94,;16.03,-20.4,;16.98,-21.61,;16.14,-22.89,;16.51,-24.38,;15.41,-25.47,;13.93,-25.05,;13.55,-23.56,;14.65,-22.48,;14.67,-17.78,;14.68,-16.24,;15.99,-18.57,;17.33,-17.82,;17.34,-16.27,;18.68,-15.52,;20.02,-16.29,;18.7,-13.97,;18.66,-18.59,;18.64,-20.14,;20,-17.84,;21.32,-18.63,;21.3,-20.18,;22.62,-20.95,;22.61,-22.5,;23.94,-23.27,;23.91,-24.81,;22.59,-25.56,;25.26,-25.58,;22.66,-17.88,;22.67,-16.34,;23.99,-18.65,;25.33,-17.89,;25.35,-16.35,;26.69,-15.6,;28.02,-16.38,;29.36,-15.63,;29.38,-14.09,;28.05,-13.3,;26.71,-14.06,;26.66,-18.67,;26.64,-20.22,;28,-17.92,;29.32,-18.7,;30.66,-17.95,;31.99,-18.73,;30.68,-16.41,;7.08,-14.43,;7.99,-13.19,;7.38,-11.78,;9.52,-13.37,)| Show InChI InChI=1S/C77H102N22O16/c1-5-6-21-53(93-74(114)65(42(2)3)98-72(112)56(89-43(4)100)30-46-24-26-50(101)27-25-46)66(106)87-39-63(103)91-58(33-48-37-83-41-88-48)70(110)97-60(31-45-18-11-8-12-19-45)75(115)99-40-49(90-77(81)82)34-61(99)73(113)96-57(32-47-36-85-52-22-14-13-20-51(47)52)69(109)95-59(35-64(104)105)71(111)92-54(23-15-28-84-76(79)80)68(108)94-55(67(107)86-38-62(78)102)29-44-16-9-7-10-17-44/h7-14,16-20,22,24-27,36-37,41-42,49,53-61,65,85,101H,5-6,15,21,23,28-35,38-40H2,1-4H3,(H2,78,102)(H,83,88)(H,86,107)(H,87,106)(H,89,100)(H,91,103)(H,92,111)(H,93,114)(H,94,108)(H,95,109)(H,96,113)(H,97,110)(H,98,112)(H,104,105)(H4,79,80,84)(H4,81,82,90)/t49-,53+,54+,55+,56+,57+,58+,59+,60-,61+,65+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268798
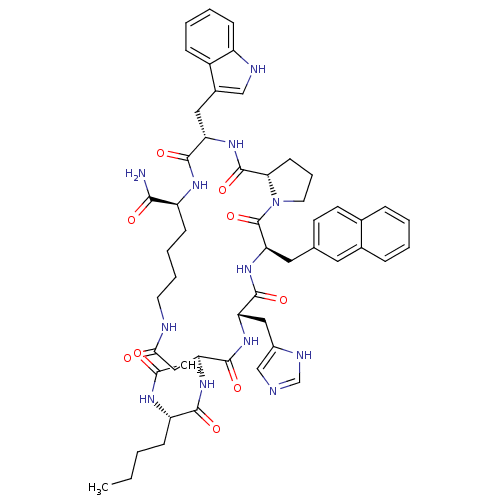
(Ac-Nle-c[Asp-His-DNaI(2')-Pro-Trp-Lys]-NH2 | CHEMB...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C53H66N12O9/c1-3-4-15-40(59-31(2)66)48(69)62-43-27-46(67)56-21-10-9-17-39(47(54)68)60-49(70)41(25-35-28-57-38-16-8-7-14-37(35)38)63-52(73)45-18-11-22-65(45)53(74)44(24-32-19-20-33-12-5-6-13-34(33)23-32)64-50(71)42(61-51(43)72)26-36-29-55-30-58-36/h5-8,12-14,16,19-20,23,28-30,39-45,57H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,54,68)(H,55,58)(H,56,67)(H,59,66)(H,60,70)(H,61,72)(H,62,69)(H,63,73)(H,64,71)/t39-,40-,41-,42-,43-,44+,45-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268803
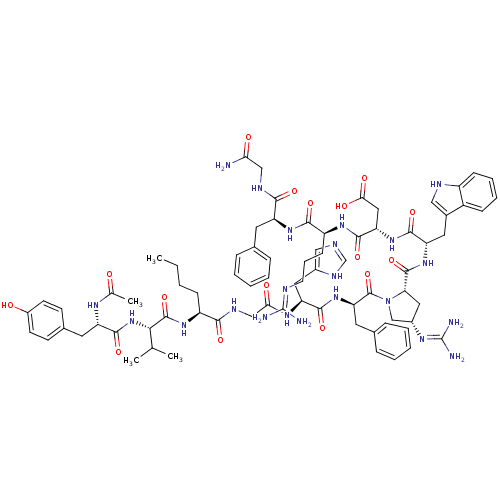
(Ac-Tyr-Val-Nle-Gly-His-DPhe-cis-Xaa-Trp-Asp-Arg-Ph...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N1C[C@H](C[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O)N=C(N)N |r,wU:8.8,45.46,59.63,57.117,96.101,35.35,77.82,wD:4.4,12.21,63.66,85.90,(2.53,-50.05,;1.21,-50.83,;-.13,-50.07,;-1.45,-50.85,;-2.8,-50.09,;-4.13,-50.88,;-4.11,-52.42,;-2.77,-53.17,;-5.44,-53.2,;-5.43,-54.75,;-6.75,-55.53,;-8.09,-54.77,;-6.74,-57.06,;-5.4,-57.82,;-4.07,-57.04,;-4.09,-55.5,;-2.76,-54.71,;-1.41,-55.47,;-.08,-54.68,;-1.39,-57.02,;-2.73,-57.8,;-8.07,-57.85,;-8.06,-59.39,;-9.38,-60.18,;-6.71,-60.14,;-6.78,-52.45,;-6.8,-50.91,;-8.1,-53.23,;-2.82,-48.56,;-4.16,-47.8,;-1.49,-47.77,;-1.51,-46.23,;-.18,-45.44,;1.17,-46.2,;-.2,-43.9,;1.13,-43.12,;1.11,-41.58,;-.23,-40.83,;-.4,-39.3,;-1.91,-38.99,;-2.66,-40.33,;-1.63,-41.47,;2.47,-43.88,;2.49,-45.42,;3.8,-43.1,;5.14,-43.85,;5.16,-45.4,;6.5,-46.15,;6.51,-47.7,;7.85,-48.46,;9.17,-47.68,;9.16,-46.14,;7.81,-45.37,;6.5,-43.14,;7.76,-44.04,;6.57,-41.61,;5.55,-40.45,;6.33,-39.12,;7.84,-39.46,;7.99,-40.99,;9.31,-41.78,;9.3,-43.32,;10.65,-41.02,;11.97,-41.82,;11.95,-43.36,;13.23,-44.22,;14.68,-43.68,;15.63,-44.89,;14.78,-46.18,;15.16,-47.67,;14.06,-48.75,;12.58,-48.33,;12.19,-46.84,;13.3,-45.76,;13.31,-41.06,;13.32,-39.52,;14.64,-41.85,;15.98,-41.1,;15.99,-39.56,;17.33,-38.8,;18.67,-39.57,;17.35,-37.26,;17.3,-41.87,;17.29,-43.42,;18.65,-41.12,;19.96,-41.91,;19.94,-43.46,;21.27,-44.24,;21.26,-45.78,;22.59,-46.55,;22.56,-48.09,;21.24,-48.84,;23.9,-48.86,;21.3,-41.16,;21.31,-39.62,;22.64,-41.93,;23.98,-41.17,;23.99,-39.64,;25.33,-38.88,;26.66,-39.66,;28.01,-38.91,;28.02,-37.37,;26.7,-36.58,;25.36,-37.34,;25.31,-41.95,;25.29,-43.5,;26.65,-41.2,;27.97,-41.99,;29.31,-41.23,;30.64,-42.01,;29.32,-39.69,;5.72,-37.71,;6.64,-36.47,;6.03,-35.06,;8.17,-36.65,)| Show InChI InChI=1S/C77H102N22O16/c1-5-6-21-53(93-74(114)65(42(2)3)98-72(112)56(89-43(4)100)30-46-24-26-50(101)27-25-46)66(106)87-39-63(103)91-58(33-48-37-83-41-88-48)70(110)97-60(31-45-18-11-8-12-19-45)75(115)99-40-49(90-77(81)82)34-61(99)73(113)96-57(32-47-36-85-52-22-14-13-20-51(47)52)69(109)95-59(35-64(104)105)71(111)92-54(23-15-28-84-76(79)80)68(108)94-55(67(107)86-38-62(78)102)29-44-16-9-7-10-17-44/h7-14,16-20,22,24-27,36-37,41-42,49,53-61,65,85,101H,5-6,15,21,23,28-35,38-40H2,1-4H3,(H2,78,102)(H,83,88)(H,86,107)(H,87,106)(H,89,100)(H,91,103)(H,92,111)(H,93,114)(H,94,108)(H,95,109)(H,96,113)(H,97,110)(H,98,112)(H,104,105)(H4,79,80,84)(H4,81,82,90)/t49-,53-,54-,55-,56-,57-,58-,59-,60+,61-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268792
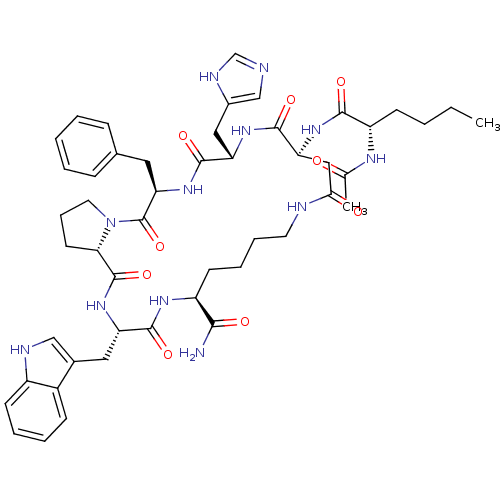
(AC-Nle-c[Asp-His-DPhe-Pro-Trp-Lys]-NH2 | CHEMBL501...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C49H64N12O9/c1-3-4-16-36(55-29(2)62)44(65)58-39-25-42(63)52-20-11-10-18-35(43(50)64)56-45(66)37(23-31-26-53-34-17-9-8-15-33(31)34)59-48(69)41-19-12-21-61(41)49(70)40(22-30-13-6-5-7-14-30)60-46(67)38(57-47(39)68)24-32-27-51-28-54-32/h5-9,13-15,17,26-28,35-41,53H,3-4,10-12,16,18-25H2,1-2H3,(H2,50,64)(H,51,54)(H,52,63)(H,55,62)(H,56,66)(H,57,68)(H,58,65)(H,59,69)(H,60,67)/t35-,36-,37-,38-,39-,40+,41-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268800
(Ac-Nle-c[Asp-His-DNal(2')-cis-4-guanidinyl-Pro-Trp...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H]2C[C@H](CN2C(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)N=C(N)N)C(N)=O |r,wU:46.49,21.81,12.11,4.4,39.45,wD:61.66,25.25,41.77,(17.08,-28.2,;15.72,-27.49,;15.65,-25.95,;14.29,-25.23,;14.22,-23.7,;15.52,-22.87,;15.46,-21.33,;16.76,-20.51,;14.09,-20.62,;12.85,-22.98,;12.79,-21.44,;11.56,-23.81,;10.19,-23.09,;8.89,-23.93,;8.95,-25.47,;10.32,-26.17,;7.66,-26.29,;6.29,-25.58,;4.99,-26.4,;3.62,-25.69,;2.33,-26.52,;.96,-25.8,;.9,-24.26,;-.47,-23.55,;-1.77,-24.38,;-.53,-22.01,;-1.9,-21.3,;-1.97,-19.76,;-.75,-18.81,;-1.29,-17.36,;-2.83,-17.43,;-3.91,-16.34,;-5.39,-16.73,;-5.79,-18.22,;-4.71,-19.3,;-3.24,-18.91,;.77,-21.19,;1.43,-22.26,;1.77,-23.81,;2.13,-21.9,;2.76,-23.47,;4.3,-23.41,;4.72,-21.93,;3.43,-21.07,;3.37,-19.54,;2,-18.82,;4.67,-18.71,;4.6,-17.17,;3.24,-16.46,;1.94,-17.29,;.57,-16.58,;.5,-15.04,;-.85,-14.32,;-.92,-12.79,;.39,-11.96,;1.75,-12.68,;1.81,-14.22,;3.17,-14.92,;6.03,-19.42,;7.33,-18.6,;7.26,-17.05,;8.7,-19.3,;10,-18.48,;9.94,-16.94,;11.15,-15.99,;10.63,-14.55,;9.09,-14.6,;8.66,-16.08,;8.76,-20.84,;10.13,-21.56,;11.43,-20.73,;5.25,-24.61,;6.76,-24.38,;7.52,-24.99,;7.32,-22.97,;-.34,-26.63,;-1.71,-25.91,;-.27,-28.17,)| Show InChI InChI=1S/C54H69N15O9/c1-3-4-14-40(62-30(2)70)48(73)66-43-25-46(71)59-19-10-9-16-39(47(55)72)64-49(74)41(22-34-26-60-38-15-8-7-13-37(34)38)67-52(77)45-24-36(63-54(56)57)28-69(45)53(78)44(21-31-17-18-32-11-5-6-12-33(32)20-31)68-50(75)42(65-51(43)76)23-35-27-58-29-61-35/h5-8,11-13,15,17-18,20,26-27,29,36,39-45,60H,3-4,9-10,14,16,19,21-25,28H2,1-2H3,(H2,55,72)(H,58,61)(H,59,71)(H,62,70)(H,64,74)(H,65,76)(H,66,73)(H,67,77)(H,68,75)(H4,56,57,63)/t36-,39+,40+,41+,42+,43+,44-,45+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268804
(Ac-Tyr-Val-Nle-Gly-His-DPhe-Pro-Trp-Asp-Arg-Phe-Gl...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O |r,wU:8.8,45.46,59.63,96.101,35.35,77.82,wD:4.4,12.21,63.66,85.90,(2.68,-6.73,;1.36,-7.52,;.01,-6.75,;-1.31,-7.53,;-2.65,-6.78,;-3.99,-7.57,;-3.97,-9.11,;-2.62,-9.86,;-5.3,-9.89,;-5.28,-11.43,;-6.61,-12.22,;-7.94,-11.46,;-6.59,-13.75,;-5.25,-14.51,;-3.92,-13.72,;-3.94,-12.19,;-2.61,-11.4,;-1.26,-12.16,;.06,-11.37,;-1.25,-13.71,;-2.58,-14.49,;-7.92,-14.54,;-7.92,-16.08,;-9.23,-16.86,;-6.56,-16.83,;-6.64,-9.14,;-6.65,-7.59,;-7.95,-9.92,;-2.67,-5.24,;-4.01,-4.49,;-1.35,-4.46,;-1.37,-2.92,;-.03,-2.13,;1.32,-2.89,;-.05,-.59,;1.27,.2,;1.26,1.73,;-.08,2.49,;-.25,4.02,;-1.77,4.33,;-2.52,2.99,;-1.48,1.85,;2.62,-.57,;2.64,-2.11,;3.94,.22,;5.29,-.54,;5.31,-2.09,;6.64,-2.84,;6.66,-4.39,;8,-5.15,;9.32,-4.36,;9.3,-2.82,;7.96,-2.06,;6.65,.17,;7.9,-.73,;6.72,1.71,;5.7,2.87,;6.48,4.19,;7.98,3.86,;8.13,2.32,;9.45,1.54,;9.44,-0,;10.8,2.29,;12.11,1.49,;12.1,-.05,;13.38,-.9,;14.82,-.37,;15.78,-1.58,;14.93,-2.86,;15.31,-4.35,;14.21,-5.43,;12.73,-5.02,;12.34,-3.52,;13.44,-2.45,;13.46,2.25,;13.47,3.79,;14.78,1.46,;16.13,2.22,;16.14,3.76,;17.48,4.52,;18.82,3.75,;17.5,6.06,;17.45,1.44,;17.43,-.1,;18.79,2.2,;20.11,1.4,;20.09,-.14,;21.42,-.92,;21.41,-2.46,;22.74,-3.24,;22.71,-4.78,;21.38,-5.53,;24.05,-5.55,;21.45,2.16,;21.46,3.7,;22.79,1.39,;24.13,2.15,;24.14,3.68,;25.48,4.44,;26.81,3.66,;28.16,4.41,;28.17,5.95,;26.84,6.74,;25.51,5.98,;25.46,1.36,;25.44,-.18,;26.8,2.12,;28.12,1.33,;29.46,2.09,;30.79,1.3,;29.47,3.63,)| Show InChI InChI=1S/C76H99N19O16/c1-5-6-22-53(89-74(110)65(43(2)3)94-72(108)56(86-44(4)96)33-47-26-28-50(97)29-27-47)66(102)84-41-63(99)87-58(36-49-39-80-42-85-49)70(106)93-60(34-46-19-11-8-12-20-46)75(111)95-31-16-25-61(95)73(109)92-57(35-48-38-82-52-23-14-13-21-51(48)52)69(105)91-59(37-64(100)101)71(107)88-54(24-15-30-81-76(78)79)68(104)90-55(67(103)83-40-62(77)98)32-45-17-9-7-10-18-45/h7-14,17-21,23,26-29,38-39,42-43,53-61,65,82,97H,5-6,15-16,22,24-25,30-37,40-41H2,1-4H3,(H2,77,98)(H,80,85)(H,83,103)(H,84,102)(H,86,96)(H,87,99)(H,88,107)(H,89,110)(H,90,104)(H,91,105)(H,92,109)(H,93,106)(H,94,108)(H,100,101)(H4,78,79,81)/t53-,54-,55-,56-,57-,58-,59-,60+,61-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268794
(Ac-Nle-c[Asp-His-DPhe-cis-4-guanidinyl-Pro-Trp-Lys...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H]2C[C@@H](CN2C(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)N=C(N)N)C(N)=O |r,wU:46.49,21.76,12.11,41.72,4.4,wD:57.61,25.25,39.45,(20.33,-40.8,;18.97,-40.08,;18.92,-38.54,;17.56,-37.81,;17.51,-36.27,;18.82,-35.46,;18.77,-33.92,;20.08,-33.11,;17.41,-33.19,;16.15,-35.55,;16.1,-34.01,;14.85,-36.36,;13.49,-35.64,;12.18,-36.45,;12.23,-37.99,;13.59,-38.71,;10.92,-38.8,;9.56,-38.08,;8.26,-38.89,;6.9,-38.17,;5.59,-38.98,;4.23,-38.25,;4.18,-36.71,;2.82,-35.99,;1.52,-36.8,;2.77,-34.45,;1.41,-33.72,;1.36,-32.19,;2.59,-31.24,;2.06,-29.79,;.52,-29.85,;-.55,-28.75,;-2.04,-29.13,;-2.45,-30.61,;-1.37,-31.7,;.1,-31.32,;4.08,-33.64,;4.74,-34.72,;5.06,-36.27,;5.44,-34.36,;6.06,-35.94,;7.6,-35.89,;8.02,-34.41,;6.75,-33.55,;6.7,-32.01,;5.34,-31.28,;8,-31.2,;7.95,-29.66,;6.59,-28.93,;6.55,-27.39,;5.19,-26.67,;3.88,-27.48,;3.94,-29.03,;5.29,-29.75,;9.36,-31.92,;10.67,-31.11,;10.62,-29.57,;12.03,-31.83,;13.33,-31.02,;13.29,-29.48,;14.51,-28.54,;14,-27.09,;12.46,-27.13,;12.02,-28.61,;12.08,-33.37,;13.44,-34.1,;14.74,-33.28,;8.54,-37.11,;10.04,-36.73,;10.8,-37.54,;10.65,-35.47,;2.93,-39.07,;1.57,-38.34,;2.98,-40.61,)| Show InChI InChI=1S/C50H67N15O9/c1-3-4-15-36(58-28(2)66)44(69)62-39-23-42(67)55-18-11-10-17-35(43(51)68)60-45(70)37(20-30-24-56-34-16-9-8-14-33(30)34)63-48(73)41-22-32(59-50(52)53)26-65(41)49(74)40(19-29-12-6-5-7-13-29)64-46(71)38(61-47(39)72)21-31-25-54-27-57-31/h5-9,12-14,16,24-25,27,32,35-41,56H,3-4,10-11,15,17-23,26H2,1-2H3,(H2,51,68)(H,54,57)(H,55,67)(H,58,66)(H,60,70)(H,61,72)(H,62,69)(H,63,73)(H,64,71)(H4,52,53,59)/t32-,35-,36-,37-,38-,39-,40+,41+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | <0.100 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50105879
(15-(2-Acetylamino-hexanoylamino)-6-(3-guanidino-pr...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O Show InChI InChI=1S/C54H71N15O9/c1-3-4-15-40(63-31(2)70)48(73)69-45-27-46(71)59-21-10-9-17-39(47(55)72)64-51(76)43(25-35-28-61-38-16-8-7-14-37(35)38)67-49(74)41(18-11-22-60-54(56)57)65-50(75)42(24-32-19-20-33-12-5-6-13-34(33)23-32)66-52(77)44(68-53(45)78)26-36-29-58-30-62-36/h5-8,12-14,16,19-20,23,28-30,39-45,61H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,55,72)(H,58,62)(H,59,71)(H,63,70)(H,64,76)(H,65,75)(H,66,77)(H,67,74)(H,68,78)(H,69,73)(H4,56,57,60)/t39-,40+,41+,42+,43-,44+,45+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Signal transduction efficacy in cAMP assay in CHO cells expressing human melanocortin receptor 5 |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50121268
(21-(2-Acetylamino-hexanoylamino)-7-[3-(diaminometh...)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2c[nH]cn2)NC1=O)C(N)=O Show InChI InChI=1S/C54H71N15O9/c1-3-4-15-40(63-31(2)70)48(73)69-45-27-46(71)59-21-10-9-17-39(47(55)72)64-51(76)43(25-35-28-61-38-16-8-7-14-37(35)38)67-49(74)41(18-11-22-60-54(56)57)65-50(75)42(24-32-19-20-33-12-5-6-13-34(33)23-32)66-52(77)44(68-53(45)78)26-36-29-58-30-62-36/h5-8,12-14,16,19-20,23,28-30,39-45,61H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,55,72)(H,58,62)(H,59,71)(H,63,70)(H,64,76)(H,65,75)(H,66,77)(H,67,74)(H,68,78)(H,69,73)(H4,56,57,60)/t39-,40+,41-,42+,43-,44-,45+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Binding affinity against human Melanocortin 5 receptor |
J Med Chem 45: 5287-94 (2002)
BindingDB Entry DOI: 10.7270/Q20G3KW8 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50121266
(21-(2-Acetylamino-hexanoylamino)-7-[3-(diaminometh...)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](CCCNC(N)=[NH2+])NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H]2C[C@@]3(C)CCCC[C@@]3(C)N2C1=O)C(N)=O Show InChI InChI=1S/C59H81N13O9/c1-5-6-19-43(66-35(2)73)51(76)71-47-32-49(74)63-27-14-11-21-42(50(60)75)67-54(79)46(31-39-34-65-41-20-10-9-18-40(39)41)69-52(77)44(22-15-28-64-57(61)62)68-53(78)45(30-36-23-24-37-16-7-8-17-38(37)29-36)70-55(80)48-33-58(3)25-12-13-26-59(58,4)72(48)56(47)81/h7-10,16-18,20,23-24,29,34,42-48,65H,5-6,11-15,19,21-22,25-28,30-33H2,1-4H3,(H2,60,75)(H,63,74)(H,66,73)(H,67,79)(H,68,78)(H,69,77)(H,70,80)(H,71,76)(H4,61,62,64)/p+1/t42-,43+,44+,45-,46-,47+,48+,58+,59+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Binding affinity against human Melanocortin 5 receptor |
J Med Chem 45: 5287-94 (2002)
BindingDB Entry DOI: 10.7270/Q20G3KW8 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50105883
(3-[25-Acetylamino-4-carbamoyl-13-(3-guanidino-prop...)Show SMILES CC(=O)N[C@@H]1CSSC[C@H](NC(=O)CNC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)[C@@H](CCC(O)=O)NC1=O)C(N)=O Show InChI InChI=1S/C51H63N15O11S2/c1-27(67)60-41-25-79-78-24-40(44(52)71)61-42(68)23-58-45(72)38(19-31-21-57-34-10-5-4-9-33(31)34)65-46(73)35(11-6-16-56-51(53)54)62-48(75)37(18-28-12-13-29-7-2-3-8-30(29)17-28)64-49(76)39(20-32-22-55-26-59-32)66-47(74)36(63-50(41)77)14-15-43(69)70/h2-5,7-10,12-13,17,21-22,26,35-41,57H,6,11,14-16,18-20,23-25H2,1H3,(H2,52,71)(H,55,59)(H,58,72)(H,60,67)(H,61,68)(H,62,75)(H,63,77)(H,64,76)(H,65,73)(H,66,74)(H,69,70)(H4,53,54,56)/t35-,36+,37-,38+,39-,40-,41+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.220 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Increase in intracellular cAMP in CHO cells expressing human melanocortin receptor 5. |
J Med Chem 44: 3665-72 (2001)
BindingDB Entry DOI: 10.7270/Q2RF5T9S |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211369
(Ac-Nle4-cyclo(Asp5-Val6-D-4,4'-Bip7-Pip8-Trp9-Lys1...)Show SMILES [H][C@@]12CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](NC(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O)C(C)C Show InChI InChI=1S/C55H72N10O9/c1-5-6-19-42(59-34(4)66)50(69)61-44-31-47(67)57-27-14-12-21-41(49(56)68)60-51(70)43(30-38-32-58-40-20-11-10-18-39(38)40)62-53(72)46-22-13-15-28-65(46)55(74)45(63-54(73)48(33(2)3)64-52(44)71)29-35-23-25-37(26-24-35)36-16-8-7-9-17-36/h7-11,16-18,20,23-26,32-33,41-46,48,58H,5-6,12-15,19,21-22,27-31H2,1-4H3,(H2,56,68)(H,57,67)(H,59,66)(H,60,70)(H,61,69)(H,62,72)(H,63,73)(H,64,71)/t41?,42-,43-,44-,45-,46-,48-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50017181
(CHEMBL441738)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Effective concentration required for intracellular cAMP accumulation by Melanocortin 5 receptor |
J Med Chem 43: 4998-5002 (2001)
BindingDB Entry DOI: 10.7270/Q2P55P7V |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211395
(Ac-Nle4-cyclo(Asp5-Phg6-D-4,4'-Bip7-Pip8-Trp9-Lys1...)Show SMILES [H][C@@]12CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](NC(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O)c1ccccc1 Show InChI InChI=1S/C58H70N10O9/c1-3-4-22-45(62-36(2)69)53(72)64-47-34-50(70)60-30-15-13-24-44(52(59)71)63-54(73)46(33-41-35-61-43-23-12-11-21-42(41)43)65-56(75)49-25-14-16-31-68(49)58(77)48(32-37-26-28-39(29-27-37)38-17-7-5-8-18-38)66-57(76)51(67-55(47)74)40-19-9-6-10-20-40/h5-12,17-21,23,26-29,35,44-49,51,61H,3-4,13-16,22,24-25,30-34H2,1-2H3,(H2,59,71)(H,60,70)(H,62,69)(H,63,73)(H,64,72)(H,65,75)(H,66,76)(H,67,74)/t44?,45-,46-,47-,48-,49-,51-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.290 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268801
(Ac-Nle-c[Asp-trans-4-guanidinyl-Pro-DNal(2')-Arg-T...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@@H]2C[C@H](CN2C1=O)N=C(N)N)C(N)=O |r,wU:4.4,12.11,67.77,21.81,50.52,wD:25.25,39.41,65.73,(14,-47.54,;12.64,-46.82,;12.59,-45.28,;11.23,-44.55,;11.18,-43.02,;12.48,-42.2,;12.43,-40.66,;13.74,-39.85,;11.08,-39.94,;9.82,-42.29,;9.77,-40.75,;8.51,-43.1,;7.15,-42.38,;5.85,-43.19,;5.9,-44.73,;7.25,-45.46,;4.58,-45.55,;3.23,-44.82,;1.92,-45.64,;.56,-44.9,;-.75,-45.72,;-2.1,-44.99,;-2.16,-43.45,;-3.51,-42.73,;-4.83,-43.54,;-3.56,-41.19,;-4.94,-40.46,;-6.39,-40.96,;-7.65,-40.07,;-8.88,-40.99,;-8.38,-42.46,;-9.11,-43.8,;-8.32,-45.1,;-6.78,-45.06,;-6.05,-43.72,;-6.84,-42.42,;-2.26,-40.37,;-.94,-40.6,;.38,-41.4,;-2.31,-38.83,;-3.67,-38.11,;-3.72,-36.57,;-5.08,-35.85,;-5.13,-34.31,;-6.49,-33.58,;-6.54,-32.04,;-7.8,-34.4,;-1,-38.02,;.36,-38.75,;.41,-40.29,;1.67,-37.93,;1.61,-36.39,;.26,-35.67,;-1.06,-36.48,;-2.41,-35.77,;-2.46,-34.22,;-3.82,-33.49,;-3.87,-31.96,;-2.55,-31.14,;-1.19,-31.87,;-1.15,-33.41,;.21,-34.13,;3.02,-38.66,;4.34,-37.85,;4.28,-36.3,;5.7,-38.58,;6.78,-37.48,;8.27,-37.87,;8.27,-39.41,;5.74,-40.1,;7.1,-40.84,;8.41,-40.03,;9.5,-36.94,;9.3,-35.41,;7.88,-34.82,;10.52,-34.48,;-3.41,-45.81,;-4.77,-45.08,;-3.37,-47.35,)| Show InChI InChI=1S/C54H74N16O9/c1-3-4-15-39(63-30(2)71)47(74)69-43-27-45(72)60-21-10-9-17-38(46(55)73)65-50(77)42(25-34-28-62-37-16-8-7-14-36(34)37)67-48(75)40(18-11-22-61-53(56)57)66-49(76)41(24-31-19-20-32-12-5-6-13-33(32)23-31)68-51(78)44-26-35(64-54(58)59)29-70(44)52(43)79/h5-8,12-14,16,19-20,23,28,35,38-44,62H,3-4,9-11,15,17-18,21-22,24-27,29H2,1-2H3,(H2,55,73)(H,60,72)(H,63,71)(H,65,77)(H,66,76)(H,67,75)(H,68,78)(H,69,74)(H4,56,57,61)(H4,58,59,64)/t35-,38+,39+,40+,41-,42+,43+,44+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211375
(Ac-Nle4-cyclo(Asp5-His6-D-4,4'-Bip7-Pip8-Trp9-Lys1...)Show SMILES [H][C@@]12CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O Show InChI InChI=1S/C56H70N12O9/c1-3-4-17-43(62-34(2)69)51(72)65-46-30-49(70)59-25-12-10-19-42(50(57)71)63-52(73)44(28-38-31-60-41-18-9-8-16-40(38)41)66-55(76)48-20-11-13-26-68(48)56(77)47(27-35-21-23-37(24-22-35)36-14-6-5-7-15-36)67-53(74)45(64-54(46)75)29-39-32-58-33-61-39/h5-9,14-16,18,21-24,31-33,42-48,60H,3-4,10-13,17,19-20,25-30H2,1-2H3,(H2,57,71)(H,58,61)(H,59,70)(H,62,69)(H,63,73)(H,64,75)(H,65,72)(H,66,76)(H,67,74)/t42?,43-,44-,45-,46-,47-,48-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.370 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50017181

(CHEMBL441738)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a |
Departments of Medicinal Chemistry and Pharmacodynamics , University of Florida , Gainesville , Florida 32610 , United States.
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as increase in cAMP production incubated for 2 hrs by AlphaScreen assay |
J Med Chem 61: 3738-3744 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00251
BindingDB Entry DOI: 10.7270/Q2CV4M69 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211374
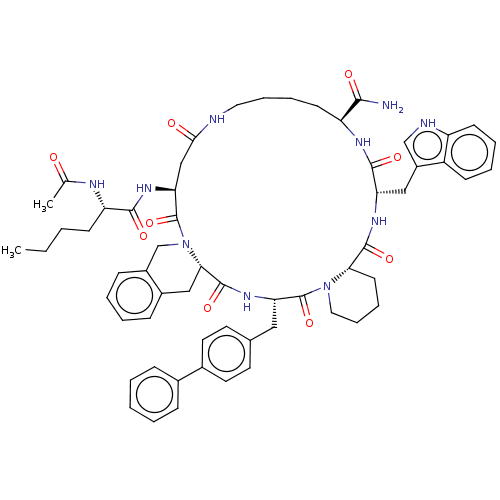
(CHEMBL2371967 | c-Nle4-cyclo(Asp5-Tic6-D-4,4'-Bip7...)Show SMILES [H][C@@]12CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@]1([H])Cc3ccccc3CN1C(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O Show InChI InChI=1S/C60H72N10O9/c1-3-4-21-47(64-37(2)71)55(74)67-50-34-53(72)62-29-14-12-23-46(54(61)73)65-56(75)48(32-43-35-63-45-22-11-10-20-44(43)45)66-57(76)51-24-13-15-30-69(51)59(78)49(31-38-25-27-40(28-26-38)39-16-6-5-7-17-39)68-58(77)52-33-41-18-8-9-19-42(41)36-70(52)60(50)79/h5-11,16-20,22,25-28,35,46-52,63H,3-4,12-15,21,23-24,29-34,36H2,1-2H3,(H2,61,73)(H,62,72)(H,64,71)(H,65,75)(H,66,76)(H,67,74)(H,68,77)/t46?,47-,48-,49-,50-,51-,52-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.450 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM82411
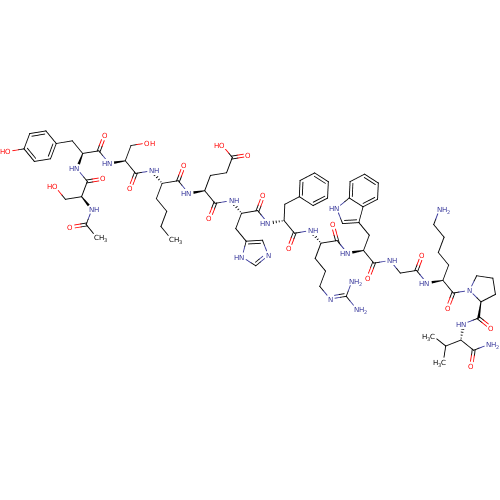
(CAS_75921-69-6 | NDP-MSH)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r,wU:66.68,55.56,45.45,111.116,4.4,14.23,wD:77.79,95.99,107.113,36.36,8.10,26.29,(-7.66,4.36,;-8.98,3.65,;-9.15,2.12,;-10.5,1.37,;-10.5,-.23,;-11.93,-.94,;-13.21,-.15,;-13.17,1.41,;-14.6,-.83,;-14.64,-2.37,;-13.43,-3.18,;-15.78,-.09,;-17.19,-.68,;-17.34,-2.22,;-18.55,.02,;-18.4,1.67,;-17.08,2.44,;-17.08,3.97,;-15.61,4.72,;-14.35,3.82,;-13,4.55,;-14.45,2.26,;-15.78,1.52,;-19.9,-.62,;-21.14,.17,;-21.14,1.73,;-22.53,-.62,;-22.53,-2.12,;-21.33,-2.88,;-23.81,.17,;-25.13,-.41,;-26.27,.45,;-25.2,-1.9,;-9.26,-1,;-9.3,-2.5,;-7.94,-.34,;-6.59,-1.15,;-6.59,-2.71,;-7.94,-3.46,;-7.94,-5,;-9.3,-5.6,;-6.63,-5.81,;-5.24,-.34,;-5.2,1.15,;-3.86,-1.11,;-2.57,-.23,;-2.57,1.26,;-1.19,2.05,;.1,1.47,;1.29,2.58,;.52,3.87,;-.97,3.65,;-1.19,-1,;-1.19,-2.5,;.1,-.3,;1.55,-1.15,;1.55,-2.65,;2.79,-3.4,;2.79,-4.89,;4.22,-5.7,;5.56,-4.89,;5.56,-3.4,;4.22,-2.65,;2.79,-.34,;2.79,1.2,;4.22,-1,;5.33,-.09,;5.29,1.47,;6.63,2.26,;6.53,3.82,;7.81,4.72,;7.81,6.13,;9.13,7.03,;6.46,6.92,;6.85,-.73,;7,-2.37,;8,.13,;9.35,-.41,;9.69,-2.07,;10.95,-2.71,;12.29,-1.9,;13.51,-2.99,;12.83,-4.36,;13.43,-5.81,;12.55,-7.03,;10.95,-6.77,;10.33,-5.43,;11.29,-4.21,;10.69,.41,;10.48,2.16,;12.04,-.41,;13.38,.3,;14.58,-.51,;14.47,-2.03,;15.99,.17,;17.25,-.68,;17.25,-2.26,;18.49,-3.08,;18.45,-4.57,;19.73,-5.55,;19.69,-7.03,;18.66,.02,;18.77,1.52,;20.01,-.68,;20.09,-2.22,;21.72,-2.54,;22.29,-1.11,;21.5,-.09,;21.78,1.47,;20.65,2.84,;23.32,1.64,;23.83,2.97,;23,4.19,;23.6,5.58,;21.5,4.04,;25.41,3.12,;26.03,4.44,;26.27,1.94,)| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.580 | n/a | n/a | n/a | n/a |
University of Michigan Medical Center
Curated by ChEMBL
| Assay Description
effective concentration of peptide at 50% maximal cAMP accumulation on Melanocortin 5 receptor |
J Med Chem 40: 2133-9 (1997)
Article DOI: 10.1021/jm960840h
BindingDB Entry DOI: 10.7270/Q28K79R6 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM82411

(CAS_75921-69-6 | NDP-MSH)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r,wU:66.68,55.56,45.45,111.116,4.4,14.23,wD:77.79,95.99,107.113,36.36,8.10,26.29,(-7.66,4.36,;-8.98,3.65,;-9.15,2.12,;-10.5,1.37,;-10.5,-.23,;-11.93,-.94,;-13.21,-.15,;-13.17,1.41,;-14.6,-.83,;-14.64,-2.37,;-13.43,-3.18,;-15.78,-.09,;-17.19,-.68,;-17.34,-2.22,;-18.55,.02,;-18.4,1.67,;-17.08,2.44,;-17.08,3.97,;-15.61,4.72,;-14.35,3.82,;-13,4.55,;-14.45,2.26,;-15.78,1.52,;-19.9,-.62,;-21.14,.17,;-21.14,1.73,;-22.53,-.62,;-22.53,-2.12,;-21.33,-2.88,;-23.81,.17,;-25.13,-.41,;-26.27,.45,;-25.2,-1.9,;-9.26,-1,;-9.3,-2.5,;-7.94,-.34,;-6.59,-1.15,;-6.59,-2.71,;-7.94,-3.46,;-7.94,-5,;-9.3,-5.6,;-6.63,-5.81,;-5.24,-.34,;-5.2,1.15,;-3.86,-1.11,;-2.57,-.23,;-2.57,1.26,;-1.19,2.05,;.1,1.47,;1.29,2.58,;.52,3.87,;-.97,3.65,;-1.19,-1,;-1.19,-2.5,;.1,-.3,;1.55,-1.15,;1.55,-2.65,;2.79,-3.4,;2.79,-4.89,;4.22,-5.7,;5.56,-4.89,;5.56,-3.4,;4.22,-2.65,;2.79,-.34,;2.79,1.2,;4.22,-1,;5.33,-.09,;5.29,1.47,;6.63,2.26,;6.53,3.82,;7.81,4.72,;7.81,6.13,;9.13,7.03,;6.46,6.92,;6.85,-.73,;7,-2.37,;8,.13,;9.35,-.41,;9.69,-2.07,;10.95,-2.71,;12.29,-1.9,;13.51,-2.99,;12.83,-4.36,;13.43,-5.81,;12.55,-7.03,;10.95,-6.77,;10.33,-5.43,;11.29,-4.21,;10.69,.41,;10.48,2.16,;12.04,-.41,;13.38,.3,;14.58,-.51,;14.47,-2.03,;15.99,.17,;17.25,-.68,;17.25,-2.26,;18.49,-3.08,;18.45,-4.57,;19.73,-5.55,;19.69,-7.03,;18.66,.02,;18.77,1.52,;20.01,-.68,;20.09,-2.22,;21.72,-2.54,;22.29,-1.11,;21.5,-.09,;21.78,1.47,;20.65,2.84,;23.32,1.64,;23.83,2.97,;23,4.19,;23.6,5.58,;21.5,4.04,;25.41,3.12,;26.03,4.44,;26.27,1.94,)| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.580 | n/a | n/a | n/a | n/a |
University of Michigan Medical Center
Curated by ChEMBL
| Assay Description
Effective concentration that was able to generate 50% maximal intracellular cAMP in L-cells transfected with Melanocortin 5 receptor |
J Med Chem 40: 1738-48 (1997)
Article DOI: 10.1021/jm960845e
BindingDB Entry DOI: 10.7270/Q29K4BWK |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50112984
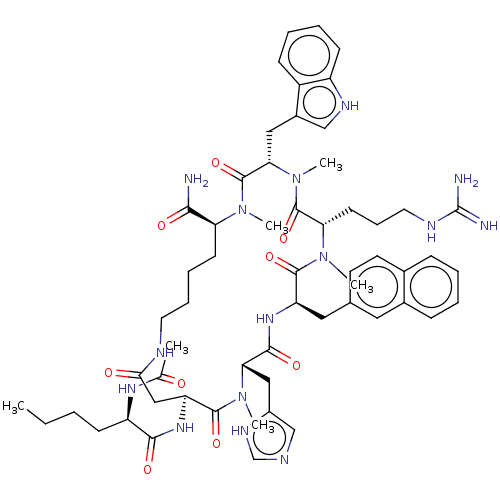
(CHEMBL3601428)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](N(C)C(=O)[C@H](Cc2c[nH]c3ccccc23)N(C)C(=O)[C@H](CCCNC(N)=N)N(C)C(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)N(C)C1=O)C(N)=O |r| Show InChI InChI=1S/C58H79N15O9/c1-7-8-19-43(67-35(2)74)52(77)68-45-31-50(75)63-25-14-13-21-46(51(59)76)70(3)57(82)49(29-39-32-65-42-20-12-11-18-41(39)42)73(6)56(81)47(22-15-26-64-58(60)61)71(4)54(79)44(28-36-23-24-37-16-9-10-17-38(37)27-36)69-53(78)48(72(5)55(45)80)30-40-33-62-34-66-40/h9-12,16-18,20,23-24,27,32-34,43-49,65H,7-8,13-15,19,21-22,25-26,28-31H2,1-6H3,(H2,59,76)(H,62,66)(H,63,75)(H,67,74)(H,68,77)(H,69,78)(H4,60,61,64)/t43-,44-,45+,46+,47+,48+,49+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5 receptor expressed in HEK293 cells assessed as intracellular cAMP accumulation using [3H]-cAMP by luminescence counting |
J Med Chem 58: 6359-67 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00102
BindingDB Entry DOI: 10.7270/Q251410F |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50232416
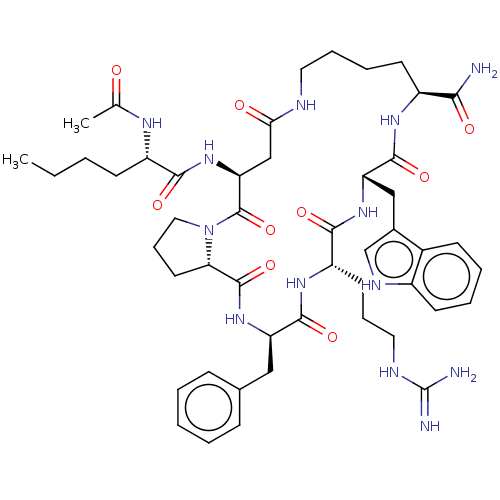
(CHEMBL4061566)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H]2CCCN2C1=O)C(N)=O |r| Show InChI InChI=1S/C49H69N13O9/c1-3-4-17-35(56-29(2)63)43(66)61-39-27-41(64)53-22-11-10-19-34(42(50)65)57-46(69)38(26-31-28-55-33-18-9-8-16-32(31)33)59-44(67)36(20-12-23-54-49(51)52)58-45(68)37(25-30-14-6-5-7-15-30)60-47(70)40-21-13-24-62(40)48(39)71/h5-9,14-16,18,28,34-40,55H,3-4,10-13,17,19-27H2,1-2H3,(H2,50,65)(H,53,64)(H,56,63)(H,57,69)(H,58,68)(H,59,67)(H,60,70)(H,61,66)(H4,51,52,54)/t34-,35-,36-,37+,38-,39-,40-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.930 | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as increase in IBMX-induced cAMP accumulation after 40 mins |
J Med Chem 60: 805-813 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01707
BindingDB Entry DOI: 10.7270/Q2R213MT |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211397
(Ac-Nle4-cyclo(Asp5-Oic6-D-4,4'-Bip7-Pro8-Trp9-Lys1...)Show SMILES [H][C@@]12CC3CCCCC3N1C(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC(=O)[C@]1([H])CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O Show InChI InChI=1S/C58H74N10O9/c1-3-4-19-44(62-35(2)69)53(72)65-47-33-51(70)60-28-13-12-21-43(52(59)71)63-54(73)45(31-40-34-61-42-20-10-9-18-41(40)42)64-55(74)49-23-14-29-67(49)57(76)46(30-36-24-26-38(27-25-36)37-15-6-5-7-16-37)66-56(75)50-32-39-17-8-11-22-48(39)68(50)58(47)77/h5-7,9-10,15-16,18,20,24-27,34,39,43-50,61H,3-4,8,11-14,17,19,21-23,28-33H2,1-2H3,(H2,59,71)(H,60,70)(H,62,69)(H,63,73)(H,64,74)(H,65,72)(H,66,75)/t39?,43?,44-,45-,46-,47-,48?,49-,50-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.990 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM82411

(CAS_75921-69-6 | NDP-MSH)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r,wU:66.68,55.56,45.45,111.116,4.4,14.23,wD:77.79,95.99,107.113,36.36,8.10,26.29,(-7.66,4.36,;-8.98,3.65,;-9.15,2.12,;-10.5,1.37,;-10.5,-.23,;-11.93,-.94,;-13.21,-.15,;-13.17,1.41,;-14.6,-.83,;-14.64,-2.37,;-13.43,-3.18,;-15.78,-.09,;-17.19,-.68,;-17.34,-2.22,;-18.55,.02,;-18.4,1.67,;-17.08,2.44,;-17.08,3.97,;-15.61,4.72,;-14.35,3.82,;-13,4.55,;-14.45,2.26,;-15.78,1.52,;-19.9,-.62,;-21.14,.17,;-21.14,1.73,;-22.53,-.62,;-22.53,-2.12,;-21.33,-2.88,;-23.81,.17,;-25.13,-.41,;-26.27,.45,;-25.2,-1.9,;-9.26,-1,;-9.3,-2.5,;-7.94,-.34,;-6.59,-1.15,;-6.59,-2.71,;-7.94,-3.46,;-7.94,-5,;-9.3,-5.6,;-6.63,-5.81,;-5.24,-.34,;-5.2,1.15,;-3.86,-1.11,;-2.57,-.23,;-2.57,1.26,;-1.19,2.05,;.1,1.47,;1.29,2.58,;.52,3.87,;-.97,3.65,;-1.19,-1,;-1.19,-2.5,;.1,-.3,;1.55,-1.15,;1.55,-2.65,;2.79,-3.4,;2.79,-4.89,;4.22,-5.7,;5.56,-4.89,;5.56,-3.4,;4.22,-2.65,;2.79,-.34,;2.79,1.2,;4.22,-1,;5.33,-.09,;5.29,1.47,;6.63,2.26,;6.53,3.82,;7.81,4.72,;7.81,6.13,;9.13,7.03,;6.46,6.92,;6.85,-.73,;7,-2.37,;8,.13,;9.35,-.41,;9.69,-2.07,;10.95,-2.71,;12.29,-1.9,;13.51,-2.99,;12.83,-4.36,;13.43,-5.81,;12.55,-7.03,;10.95,-6.77,;10.33,-5.43,;11.29,-4.21,;10.69,.41,;10.48,2.16,;12.04,-.41,;13.38,.3,;14.58,-.51,;14.47,-2.03,;15.99,.17,;17.25,-.68,;17.25,-2.26,;18.49,-3.08,;18.45,-4.57,;19.73,-5.55,;19.69,-7.03,;18.66,.02,;18.77,1.52,;20.01,-.68,;20.09,-2.22,;21.72,-2.54,;22.29,-1.11,;21.5,-.09,;21.78,1.47,;20.65,2.84,;23.32,1.64,;23.83,2.97,;23,4.19,;23.6,5.58,;21.5,4.04,;25.41,3.12,;26.03,4.44,;26.27,1.94,)| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Effective concentration of peptide at 50% maximal cAMP generation |
J Med Chem 46: 3728-33 (2003)
Article DOI: 10.1021/jm030111j
BindingDB Entry DOI: 10.7270/Q2125TDT |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50017181

(CHEMBL441738)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Effective concentration for intracellular cAMP accumulation in human melanocortin 5 receptor expressing HEK 293 cells; (N = 4) |
J Med Chem 48: 1839-48 (2005)
Article DOI: 10.1021/jm049579s
BindingDB Entry DOI: 10.7270/Q2XD12F5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50114078
(8-(3-Guanidino-propyl)-2-(3H-imidazol-4-ylmethyl)-...)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)CCCC(=O)NCCCC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC1=O)C(N)=O Show InChI InChI=1S/C47H59N13O7/c48-42(63)35-13-5-6-19-52-40(61)15-7-16-41(62)56-39(24-32-26-51-27-55-32)46(67)59-37(22-28-17-18-29-9-1-2-10-30(29)21-28)44(65)58-36(14-8-20-53-47(49)50)43(64)60-38(45(66)57-35)23-31-25-54-34-12-4-3-11-33(31)34/h1-4,9-12,17-18,21,25-27,35-39,54H,5-8,13-16,19-20,22-24H2,(H2,48,63)(H,51,55)(H,52,61)(H,56,62)(H,57,66)(H,58,65)(H,59,67)(H,60,64)(H4,49,50,53)/t35-,36+,37-,38-,39+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Effective concentration against human melanocortin 5 receptor (hMC5R) in HEK293 cells |
J Med Chem 45: 2644-50 (2002)
BindingDB Entry DOI: 10.7270/Q2RB75B3 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211379
(Ac-Nle4-cyclo(Asp5-Chg6-D-4,4'-Bip7-Pip8-Trp9-Lys1...)Show SMILES [H][C@@]12CCCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](NC(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O)C1CCCCC1 Show InChI InChI=1S/C58H76N10O9/c1-3-4-22-45(62-36(2)69)53(72)64-47-34-50(70)60-30-15-13-24-44(52(59)71)63-54(73)46(33-41-35-61-43-23-12-11-21-42(41)43)65-56(75)49-25-14-16-31-68(49)58(77)48(32-37-26-28-39(29-27-37)38-17-7-5-8-18-38)66-57(76)51(67-55(47)74)40-19-9-6-10-20-40/h5,7-8,11-12,17-18,21,23,26-29,35,40,44-49,51,61H,3-4,6,9-10,13-16,19-20,22,24-25,30-34H2,1-2H3,(H2,59,71)(H,60,70)(H,62,69)(H,63,73)(H,64,72)(H,65,75)(H,66,76)(H,67,74)/t44?,45-,46-,47-,48-,49-,51-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50250586
(CHEMBL4093140)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)CN[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H113N21O18/c1-5-6-19-54(93-75(115)62(42-101)97-73(113)59(34-47-24-27-51(103)28-25-47)95-74(114)61(41-100)89-45(4)102)69(109)90-49(26-29-65(105)106)39-86-57(36-50-38-83-43-88-50)71(111)94-58(33-46-16-8-7-9-17-46)72(112)92-55(22-14-31-84-78(81)82)70(110)96-60(35-48-37-85-53-20-11-10-18-52(48)53)68(108)87-40-64(104)91-56(21-12-13-30-79)77(117)99-32-15-23-63(99)76(116)98-66(44(2)3)67(80)107/h7-11,16-18,20,24-25,27-28,37-38,43-44,49,54-63,66,85-86,100-101,103H,5-6,12-15,19,21-23,26,29-36,39-42,79H2,1-4H3,(H2,80,107)(H,83,88)(H,87,108)(H,89,102)(H,90,109)(H,91,104)(H,92,112)(H,93,115)(H,94,111)(H,95,114)(H,96,110)(H,97,113)(H,98,116)(H,105,106)(H4,81,82,84)/t49-,54-,55-,56-,57-,58+,59-,60-,61-,62-,63-,66-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
The University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as induction of intracellular cAMP accumulation after 3 mins |
J Med Chem 60: 9320-9329 (2017)
Article DOI: 10.1021/acs.jmedchem.7b01295
BindingDB Entry DOI: 10.7270/Q2XG9TJB |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50017181

(CHEMBL441738)Show SMILES CCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(N)=O |r| Show InChI InChI=1S/C78H111N21O19/c1-5-6-19-52(91-75(116)61(41-101)97-72(113)57(34-46-24-26-49(103)27-25-46)94-74(115)60(40-100)88-44(4)102)68(109)92-54(28-29-64(105)106)70(111)96-59(36-48-38-83-42-87-48)73(114)93-56(33-45-16-8-7-9-17-45)71(112)90-53(22-14-31-84-78(81)82)69(110)95-58(35-47-37-85-51-20-11-10-18-50(47)51)67(108)86-39-63(104)89-55(21-12-13-30-79)77(118)99-32-15-23-62(99)76(117)98-65(43(2)3)66(80)107/h7-11,16-18,20,24-27,37-38,42-43,52-62,65,85,100-101,103H,5-6,12-15,19,21-23,28-36,39-41,79H2,1-4H3,(H2,80,107)(H,83,87)(H,86,108)(H,88,102)(H,89,104)(H,90,112)(H,91,116)(H,92,109)(H,93,114)(H,94,115)(H,95,110)(H,96,111)(H,97,113)(H,98,117)(H,105,106)(H4,81,82,84)/t52-,53-,54-,55-,56+,57-,58-,59-,60-,61-,62-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a |
CECB, Department of Chemistry, University of Copenhagen , Universitetsparken 5, 2100 Copenhagen, Denmark.
Curated by ChEMBL
| Assay Description
Agonist activity at EYFP-fused human MC5R expressed in HEK293 cells after 16 to 20 hrs by CRE-driven reporter assay |
J Med Chem 60: 8716-8730 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00353
BindingDB Entry DOI: 10.7270/Q2VQ3535 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50112905
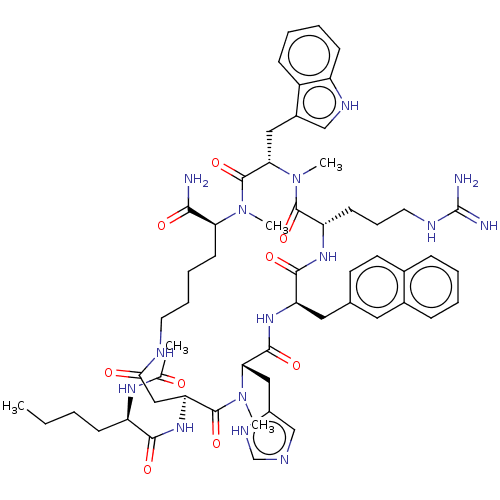
(CHEMBL3600916)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](N(C)C(=O)[C@H](Cc2c[nH]c3ccccc23)N(C)C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2cnc[nH]2)N(C)C1=O)C(N)=O |r| Show InChI InChI=1S/C57H77N15O9/c1-6-7-18-42(66-34(2)73)51(76)69-45-30-49(74)62-24-13-12-21-46(50(58)75)70(3)56(81)48(28-38-31-64-41-19-11-10-17-40(38)41)72(5)54(79)43(20-14-25-63-57(59)60)67-52(77)44(27-35-22-23-36-15-8-9-16-37(36)26-35)68-53(78)47(71(4)55(45)80)29-39-32-61-33-65-39/h8-11,15-17,19,22-23,26,31-33,42-48,64H,6-7,12-14,18,20-21,24-25,27-30H2,1-5H3,(H2,58,75)(H,61,65)(H,62,74)(H,66,73)(H,67,77)(H,68,78)(H,69,76)(H4,59,60,63)/t42-,43+,44-,45+,46+,47+,48+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5 receptor expressed in HEK293 cells assessed as intracellular cAMP accumulation using [3H]-cAMP by luminescence counting |
J Med Chem 58: 6359-67 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00102
BindingDB Entry DOI: 10.7270/Q251410F |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50121268

(21-(2-Acetylamino-hexanoylamino)-7-[3-(diaminometh...)Show SMILES CCCC[C@@H](NC(C)=O)C(=O)N[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2c[nH]cn2)NC1=O)C(N)=O Show InChI InChI=1S/C54H71N15O9/c1-3-4-15-40(63-31(2)70)48(73)69-45-27-46(71)59-21-10-9-17-39(47(55)72)64-51(76)43(25-35-28-61-38-16-8-7-14-37(35)38)67-49(74)41(18-11-22-60-54(56)57)65-50(75)42(24-32-19-20-33-12-5-6-13-34(33)23-32)66-52(77)44(68-53(45)78)26-36-29-58-30-62-36/h5-8,12-14,16,19-20,23,28-30,39-45,61H,3-4,9-11,15,17-18,21-22,24-27H2,1-2H3,(H2,55,72)(H,58,62)(H,59,71)(H,63,70)(H,64,76)(H,65,75)(H,66,77)(H,67,74)(H,68,78)(H,69,73)(H4,56,57,60)/t39-,40+,41-,42+,43-,44-,45+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50184359
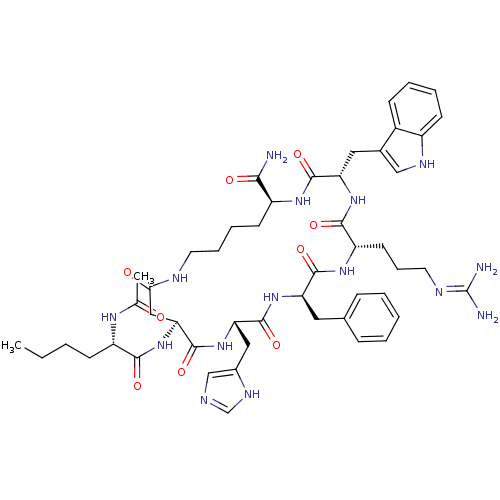
((3S,6S,9R,12S,15S,23S)-15-((S)-2-acetylamino-hexan...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O |r,wU:21.75,50.52,12.11,4.4,39.41,wD:25.25,61.64,(19.68,-11.7,;18.32,-10.97,;18.27,-9.43,;16.91,-8.71,;16.86,-7.17,;18.17,-6.36,;18.11,-4.82,;19.42,-4,;16.76,-4.09,;15.5,-6.44,;15.45,-4.9,;14.19,-7.26,;12.83,-6.53,;11.53,-7.35,;11.58,-8.88,;12.94,-9.61,;10.27,-9.7,;8.91,-8.97,;7.6,-9.79,;6.25,-9.06,;4.94,-9.87,;3.58,-9.15,;3.53,-7.61,;2.17,-6.88,;.86,-7.7,;2.12,-5.35,;.76,-4.62,;.71,-3.08,;1.93,-2.14,;1.41,-.69,;-.14,-.74,;-1.21,.35,;-2.69,-.02,;-3.1,-1.51,;-2.03,-2.6,;-.56,-2.22,;3.43,-4.53,;4.79,-5.26,;4.84,-6.8,;6.98,-5.22,;8.76,-4.36,;10.12,-5.08,;10.17,-6.62,;8.86,-7.43,;7.5,-6.71,;6.19,-7.52,;8.83,-5.93,;6.09,-4.44,;6.04,-2.9,;4.68,-2.18,;7.35,-2.09,;7.3,-.55,;5.94,.17,;5.89,1.71,;4.53,2.44,;3.23,1.62,;3.28,.07,;4.64,-.64,;8.71,-2.82,;10.02,-2,;9.96,-.46,;11.37,-2.73,;12.68,-1.91,;12.64,-.37,;13.86,.56,;13.35,2.01,;11.81,1.97,;11.37,.5,;11.42,-4.27,;12.78,-4.99,;14.09,-4.18,;2.27,-9.96,;.91,-9.24,;2.32,-11.5,)| Show InChI InChI=1S/C50H69N15O9/c1-3-4-16-36(59-29(2)66)44(69)65-41-25-42(67)55-20-11-10-18-35(43(51)68)60-47(72)39(23-31-26-57-34-17-9-8-15-33(31)34)63-45(70)37(19-12-21-56-50(52)53)61-46(71)38(22-30-13-6-5-7-14-30)62-48(73)40(64-49(41)74)24-32-27-54-28-58-32/h5-9,13-15,17,26-28,35-41,57H,3-4,10-12,16,18-25H2,1-2H3,(H2,51,68)(H,54,58)(H,55,67)(H,59,66)(H,60,72)(H,61,71)(H,62,73)(H,63,70)(H,64,74)(H,65,69)(H4,52,53,56)/t35-,36-,37-,38+,39-,40-,41-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
University of Michigan Medical Center
Curated by ChEMBL
| Assay Description
Effective concentration that was able to generate 50% maximal intracellular cAMP in L-cells transfected with Melanocortin 5 receptor |
J Med Chem 40: 1738-48 (1997)
Article DOI: 10.1021/jm960845e
BindingDB Entry DOI: 10.7270/Q29K4BWK |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268795
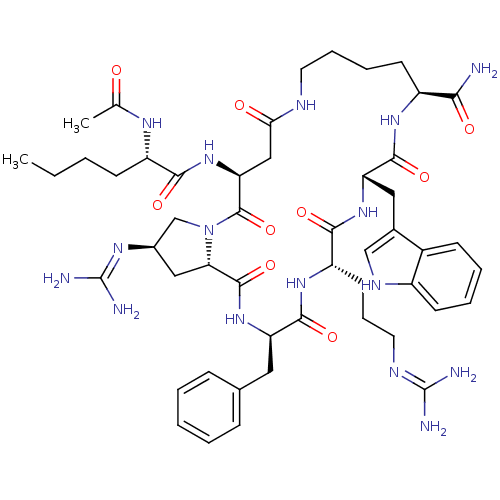
(Ac-Nle-c[Asp-trans-4-guanidinyl-Pro-DPhe-Arg-Trp-L...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@@H]2C[C@H](CN2C1=O)N=C(N)N)C(N)=O |r,wU:4.4,50.52,63.72,12.11,21.76,wD:25.25,39.41,61.68,(21.05,-10.24,;19.69,-9.52,;19.64,-7.97,;18.28,-7.25,;18.23,-5.71,;19.54,-4.89,;19.48,-3.35,;20.79,-2.54,;18.13,-2.63,;16.87,-4.98,;16.82,-3.44,;15.56,-5.8,;14.2,-5.07,;12.89,-5.89,;12.94,-7.43,;14.3,-8.15,;11.63,-8.24,;10.27,-7.52,;8.96,-8.33,;7.6,-7.6,;6.29,-8.41,;4.93,-7.69,;4.88,-6.15,;3.52,-5.42,;2.21,-6.24,;3.47,-3.88,;2.1,-3.15,;.64,-3.65,;-.62,-2.76,;-1.85,-3.68,;-1.35,-5.15,;-2.08,-6.49,;-1.28,-7.8,;.26,-7.76,;.99,-6.41,;.19,-5.12,;4.78,-3.07,;6.1,-3.3,;7.43,-4.09,;4.73,-1.52,;3.37,-.8,;3.32,.74,;1.96,1.47,;1.91,3.01,;.54,3.73,;.5,5.28,;-.76,2.92,;6.03,-.71,;7.4,-1.43,;7.45,-2.98,;8.71,-.62,;8.66,.92,;7.3,1.65,;7.25,3.18,;5.89,3.91,;4.58,3.1,;4.63,1.55,;5.98,.83,;10.06,-1.35,;11.38,-.54,;11.33,1.01,;12.75,-1.27,;13.83,-.17,;15.32,-.56,;15.32,-2.1,;12.78,-2.79,;14.15,-3.53,;15.45,-2.72,;16.54,.37,;16.34,1.9,;14.92,2.5,;17.57,2.84,;3.63,-8.5,;2.26,-7.78,;3.67,-10.04,)| Show InChI InChI=1S/C50H72N16O9/c1-3-4-16-35(59-28(2)67)43(70)65-39-25-41(68)56-20-11-10-18-34(42(51)69)61-46(73)38(23-30-26-58-33-17-9-8-15-32(30)33)63-44(71)36(19-12-21-57-49(52)53)62-45(72)37(22-29-13-6-5-7-14-29)64-47(74)40-24-31(60-50(54)55)27-66(40)48(39)75/h5-9,13-15,17,26,31,34-40,58H,3-4,10-12,16,18-25,27H2,1-2H3,(H2,51,69)(H,56,68)(H,59,67)(H,61,73)(H,62,72)(H,63,71)(H,64,74)(H,65,70)(H4,52,53,57)(H4,54,55,60)/t31-,34+,35+,36+,37-,38+,39+,40+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211383
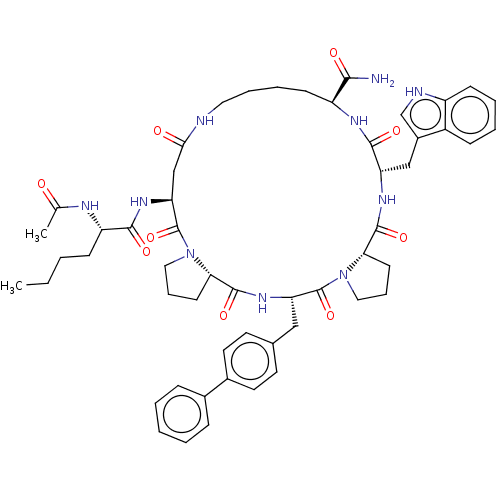
(Ac-Nle4-cyclo(Asp5-Pro6-D-4,4'-Bip7-Pip8-Trp9-Lys1...)Show SMILES [H][C@@]12CCCN1C(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@]1([H])CCCN1C(=O)[C@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC2=O)C(N)=O)NC(=O)[C@H](CCCC)NC(C)=O Show InChI InChI=1S/C55H70N10O9/c1-3-4-18-42(59-34(2)66)50(69)62-45-32-48(67)57-27-12-10-20-41(49(56)68)60-51(70)43(31-38-33-58-40-19-9-8-17-39(38)40)61-52(71)46-21-11-13-28-64(46)54(73)44(63-53(72)47-22-14-29-65(47)55(45)74)30-35-23-25-37(26-24-35)36-15-6-5-7-16-36/h5-9,15-17,19,23-26,33,41-47,58H,3-4,10-14,18,20-22,27-32H2,1-2H3,(H2,56,68)(H,57,67)(H,59,66)(H,60,70)(H,61,71)(H,62,69)(H,63,72)/t41?,42-,43-,44-,45-,46-,47-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50027084
(Melatonan)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01848
BindingDB Entry DOI: 10.7270/Q2J67N1M |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50134943
(CHEMBL2370966 | H-Tyr-Val-Nle-Gly-Aib-Phe-Arg-D-Na...)Show SMILES [#6]-[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c1ccc(-[#8])cc1)-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-c1ccc2ccccc2c1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C75H104N20O15/c1-4-5-22-52(90-73(110)63(43(2)3)95-64(101)51(77)36-46-26-29-50(96)30-27-46)65(102)86-42-61(98)87-55(31-32-76)69(106)92-57(38-45-18-10-7-11-19-45)70(107)88-54(24-15-34-84-75(81)82)68(105)93-58(39-47-25-28-48-20-12-13-21-49(48)35-47)71(108)94-59(40-62(99)100)72(109)89-53(23-14-33-83-74(79)80)67(104)91-56(66(103)85-41-60(78)97)37-44-16-8-6-9-17-44/h6-13,16-21,25-30,35,43,51-59,63,96H,4-5,14-15,22-24,31-34,36-42,76-77H2,1-3H3,(H2,78,97)(H,85,103)(H,86,102)(H,87,98)(H,88,107)(H,89,109)(H,90,110)(H,91,104)(H,92,106)(H,93,105)(H,94,108)(H,95,101)(H,99,100)(H4,79,80,83)(H4,81,82,84)/t51-,52-,53-,54-,55-,56-,57?,58?,59-,63-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Intracellular cAMP accumulation in human Melanocortin 5 receptor functional assay. |
J Med Chem 46: 4965-73 (2003)
Article DOI: 10.1021/jm030119t
BindingDB Entry DOI: 10.7270/Q2SJ1K0P |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268806
(Ac-Tyr-Val-Nle-Gly-His-DPhe-Arg-Trp-Asp-Arg-Phe-Gl...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O |r,wU:12.21,56.58,35.35,81.85,45.46,4.4,100.104,wD:67.69,8.8,89.93,(6.82,-32.01,;5.49,-32.78,;5.49,-34.31,;4.16,-35.09,;4.16,-36.62,;2.82,-37.38,;1.49,-36.61,;1.49,-35.08,;.16,-37.38,;-1.17,-36.62,;-2.5,-37.4,;-2.5,-38.94,;-3.83,-36.63,;-3.83,-35.09,;-2.49,-34.32,;-1.16,-35.09,;.18,-34.32,;.18,-32.78,;1.51,-32,;-1.15,-32.01,;-2.49,-32.78,;-5.18,-37.37,;-6.51,-36.6,;-7.85,-37.37,;-6.51,-35.06,;.16,-38.93,;1.49,-39.69,;-1.18,-39.7,;5.49,-37.39,;5.49,-38.94,;6.83,-36.63,;8.16,-37.4,;9.49,-36.63,;9.49,-35.09,;10.83,-37.4,;12.17,-36.64,;12.17,-35.1,;13.5,-34.33,;14.91,-34.96,;15.95,-33.81,;15.17,-32.48,;13.66,-32.79,;13.5,-37.41,;13.5,-38.95,;14.83,-36.63,;16.17,-37.4,;16.17,-38.95,;17.5,-39.71,;18.84,-38.95,;20.17,-39.72,;20.17,-41.26,;18.83,-42.03,;17.5,-41.26,;17.5,-36.63,;17.5,-35.1,;18.83,-37.4,;20.18,-36.65,;20.18,-35.11,;21.51,-34.34,;21.51,-32.81,;22.84,-32.03,;22.85,-30.49,;21.51,-29.71,;24.18,-29.72,;21.51,-37.42,;21.51,-38.97,;22.84,-36.64,;24.17,-37.41,;24.17,-38.95,;25.5,-39.72,;26.91,-39.1,;27.94,-40.24,;27.17,-41.58,;27.64,-43.04,;26.61,-44.18,;25.1,-43.86,;24.63,-42.39,;25.66,-41.25,;25.5,-36.64,;25.5,-35.1,;26.84,-37.41,;28.17,-36.64,;28.17,-35.11,;29.5,-34.34,;30.83,-35.11,;29.5,-32.8,;29.5,-37.42,;29.5,-38.96,;30.84,-36.64,;32.17,-37.41,;32.17,-38.96,;33.49,-39.72,;33.49,-41.27,;34.82,-42.04,;34.83,-43.58,;33.49,-44.35,;36.16,-44.35,;33.49,-36.64,;33.49,-35.11,;34.83,-37.41,;36.16,-36.64,;36.16,-35.1,;37.5,-34.34,;38.83,-35.11,;40.16,-34.34,;40.17,-32.8,;38.84,-32.03,;37.5,-32.79,;37.5,-37.42,;37.5,-38.95,;38.83,-36.64,;40.16,-37.41,;41.48,-36.64,;42.83,-37.42,;41.48,-35.11,)| Show InChI InChI=1S/C77H104N22O16/c1-5-6-22-53(94-75(115)65(43(2)3)99-74(114)57(90-44(4)100)34-47-26-28-50(101)29-27-47)66(106)88-41-63(103)91-60(36-49-39-83-42-89-49)72(112)96-58(33-46-19-11-8-12-20-46)70(110)92-55(25-16-31-85-77(81)82)69(109)97-59(35-48-38-86-52-23-14-13-21-51(48)52)71(111)98-61(37-64(104)105)73(113)93-54(24-15-30-84-76(79)80)68(108)95-56(67(107)87-40-62(78)102)32-45-17-9-7-10-18-45/h7-14,17-21,23,26-29,38-39,42-43,53-61,65,86,101H,5-6,15-16,22,24-25,30-37,40-41H2,1-4H3,(H2,78,102)(H,83,89)(H,87,107)(H,88,106)(H,90,100)(H,91,103)(H,92,110)(H,93,113)(H,94,115)(H,95,108)(H,96,112)(H,97,109)(H,98,111)(H,99,114)(H,104,105)(H4,79,80,84)(H4,81,82,85)/t53-,54-,55-,56-,57-,58+,59-,60-,61-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a |
The University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as induction of intracellular cAMP accumulation after 3 mins |
J Med Chem 60: 9320-9329 (2017)
Article DOI: 10.1021/acs.jmedchem.7b01295
BindingDB Entry DOI: 10.7270/Q2XG9TJB |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50268806

(Ac-Tyr-Val-Nle-Gly-His-DPhe-Arg-Trp-Asp-Arg-Phe-Gl...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O |r,wU:12.21,56.58,35.35,81.85,45.46,4.4,100.104,wD:67.69,8.8,89.93,(6.82,-32.01,;5.49,-32.78,;5.49,-34.31,;4.16,-35.09,;4.16,-36.62,;2.82,-37.38,;1.49,-36.61,;1.49,-35.08,;.16,-37.38,;-1.17,-36.62,;-2.5,-37.4,;-2.5,-38.94,;-3.83,-36.63,;-3.83,-35.09,;-2.49,-34.32,;-1.16,-35.09,;.18,-34.32,;.18,-32.78,;1.51,-32,;-1.15,-32.01,;-2.49,-32.78,;-5.18,-37.37,;-6.51,-36.6,;-7.85,-37.37,;-6.51,-35.06,;.16,-38.93,;1.49,-39.69,;-1.18,-39.7,;5.49,-37.39,;5.49,-38.94,;6.83,-36.63,;8.16,-37.4,;9.49,-36.63,;9.49,-35.09,;10.83,-37.4,;12.17,-36.64,;12.17,-35.1,;13.5,-34.33,;14.91,-34.96,;15.95,-33.81,;15.17,-32.48,;13.66,-32.79,;13.5,-37.41,;13.5,-38.95,;14.83,-36.63,;16.17,-37.4,;16.17,-38.95,;17.5,-39.71,;18.84,-38.95,;20.17,-39.72,;20.17,-41.26,;18.83,-42.03,;17.5,-41.26,;17.5,-36.63,;17.5,-35.1,;18.83,-37.4,;20.18,-36.65,;20.18,-35.11,;21.51,-34.34,;21.51,-32.81,;22.84,-32.03,;22.85,-30.49,;21.51,-29.71,;24.18,-29.72,;21.51,-37.42,;21.51,-38.97,;22.84,-36.64,;24.17,-37.41,;24.17,-38.95,;25.5,-39.72,;26.91,-39.1,;27.94,-40.24,;27.17,-41.58,;27.64,-43.04,;26.61,-44.18,;25.1,-43.86,;24.63,-42.39,;25.66,-41.25,;25.5,-36.64,;25.5,-35.1,;26.84,-37.41,;28.17,-36.64,;28.17,-35.11,;29.5,-34.34,;30.83,-35.11,;29.5,-32.8,;29.5,-37.42,;29.5,-38.96,;30.84,-36.64,;32.17,-37.41,;32.17,-38.96,;33.49,-39.72,;33.49,-41.27,;34.82,-42.04,;34.83,-43.58,;33.49,-44.35,;36.16,-44.35,;33.49,-36.64,;33.49,-35.11,;34.83,-37.41,;36.16,-36.64,;36.16,-35.1,;37.5,-34.34,;38.83,-35.11,;40.16,-34.34,;40.17,-32.8,;38.84,-32.03,;37.5,-32.79,;37.5,-37.42,;37.5,-38.95,;38.83,-36.64,;40.16,-37.41,;41.48,-36.64,;42.83,-37.42,;41.48,-35.11,)| Show InChI InChI=1S/C77H104N22O16/c1-5-6-22-53(94-75(115)65(43(2)3)99-74(114)57(90-44(4)100)34-47-26-28-50(101)29-27-47)66(106)88-41-63(103)91-60(36-49-39-83-42-89-49)72(112)96-58(33-46-19-11-8-12-20-46)70(110)92-55(25-16-31-85-77(81)82)69(109)97-59(35-48-38-86-52-23-14-13-21-51(48)52)71(111)98-61(37-64(104)105)73(113)93-54(24-15-30-84-76(79)80)68(108)95-56(67(107)87-40-62(78)102)32-45-17-9-7-10-18-45/h7-14,17-21,23,26-29,38-39,42-43,53-61,65,86,101H,5-6,15-16,22,24-25,30-37,40-41H2,1-4H3,(H2,78,102)(H,83,89)(H,87,107)(H,88,106)(H,90,100)(H,91,103)(H,92,110)(H,93,113)(H,94,115)(H,95,108)(H,96,112)(H,97,109)(H,98,111)(H,99,114)(H,104,105)(H4,79,80,84)(H4,81,82,85)/t53-,54-,55-,56-,57-,58+,59-,60-,61-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as stimulation of intracellular cAMP level |
J Med Chem 52: 3627-35 (2009)
Article DOI: 10.1021/jm801300c
BindingDB Entry DOI: 10.7270/Q2D21ZJ5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50163146
(Ac-Tyr-Val-Nle-Gly-His-D-Phe-Arg-Trp-Asp-Arg-Phe-G...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)ON Show InChI InChI=1S/C77H104N22O17/c1-5-6-22-53(94-75(115)65(43(2)3)99-74(114)57(90-44(4)100)34-47-26-28-50(101)29-27-47)66(106)87-40-62(102)91-60(36-49-39-83-42-89-49)72(112)96-58(33-46-19-11-8-12-20-46)70(110)92-55(25-16-31-85-77(80)81)69(109)97-59(35-48-38-86-52-23-14-13-21-51(48)52)71(111)98-61(37-63(103)104)73(113)93-54(24-15-30-84-76(78)79)68(108)95-56(32-45-17-9-7-10-18-45)67(107)88-41-64(105)116-82/h7-14,17-21,23,26-29,38-39,42-43,53-61,65,86,101H,5-6,15-16,22,24-25,30-37,40-41,82H2,1-4H3,(H,83,89)(H,87,106)(H,88,107)(H,90,100)(H,91,102)(H,92,110)(H,93,113)(H,94,115)(H,95,108)(H,96,112)(H,97,109)(H,98,111)(H,99,114)(H,103,104)(H4,78,79,84)(H4,80,81,85)/t53-,54-,55-,56-,57-,58-,59-,60-,61-,65-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Effective concentration for intracellular cAMP accumulation in human melanocortin 5 receptor expressing HEK 293 cells; (N = 4) |
J Med Chem 48: 1839-48 (2005)
Article DOI: 10.1021/jm049579s
BindingDB Entry DOI: 10.7270/Q2XD12F5 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50453139
(CHEMBL4205548)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2c[nH]cn2)NC(=O)C2(CCCCC2)NC(=O)CC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC1=O)C(N)=O |r| Show InChI InChI=1S/C44H57N13O7/c45-37(59)31-15-16-36(58)57-44(17-7-2-8-18-44)42(64)56-35(22-28-24-48-25-51-28)41(63)54-33(20-26-10-3-1-4-11-26)39(61)53-32(14-9-19-49-43(46)47)38(60)55-34(40(62)52-31)21-27-23-50-30-13-6-5-12-29(27)30/h1,3-6,10-13,23-25,31-35,50H,2,7-9,14-22H2,(H2,45,59)(H,48,51)(H,52,62)(H,53,61)(H,54,63)(H,55,60)(H,56,64)(H,57,58)(H4,46,47,49)/t31-,32-,33+,34-,35-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a |
University of Naples "Federico II"
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as induction of intracellular cAMP accumulation after 3 mins |
J Med Chem 61: 4263-4269 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00488
BindingDB Entry DOI: 10.7270/Q22J6FF7 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50453129
(CHEMBL4216242)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2c[nH]cn2)NC(=O)C2(CCC2)NC(=O)CC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC1=O)C(N)=O |r| Show InChI InChI=1S/C42H53N13O7/c43-35(57)29-13-14-34(56)55-42(15-7-16-42)40(62)54-33(20-26-22-46-23-49-26)39(61)52-31(18-24-8-2-1-3-9-24)37(59)51-30(12-6-17-47-41(44)45)36(58)53-32(38(60)50-29)19-25-21-48-28-11-5-4-10-27(25)28/h1-5,8-11,21-23,29-33,48H,6-7,12-20H2,(H2,43,57)(H,46,49)(H,50,60)(H,51,59)(H,52,61)(H,53,58)(H,54,62)(H,55,56)(H4,44,45,47)/t29-,30-,31+,32-,33-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
University of Naples "Federico II"
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in HEK293 cells assessed as induction of intracellular cAMP accumulation after 3 mins |
J Med Chem 61: 4263-4269 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00488
BindingDB Entry DOI: 10.7270/Q22J6FF7 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50121260
(3-(2-Acetylamino-hexanoylamino)-17-[3-(diaminometh...)Show SMILES [H][C@@]12Cc3ccccc3CN1C(=O)[C@@H](CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc1c[nH]c3ccccc13)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@H](Cc1ccc3ccccc3c1)NC2=O)C(N)=O)NC(=O)[C@@H](CCCC)NC(C)=O |r| Show InChI InChI=1S/C58H73N13O9/c1-3-4-19-44(65-34(2)72)52(75)70-48-31-50(73)62-25-12-11-21-43(51(59)74)66-55(78)47(29-40-32-64-42-20-10-9-18-41(40)42)68-53(76)45(22-13-26-63-58(60)61)67-54(77)46(28-35-23-24-36-14-5-6-15-37(36)27-35)69-56(79)49-30-38-16-7-8-17-39(38)33-71(49)57(48)80/h5-10,14-18,20,23-24,27,32,43-49,64H,3-4,11-13,19,21-22,25-26,28-31,33H2,1-2H3,(H2,59,74)(H,62,73)(H,65,72)(H,66,78)(H,67,77)(H,68,76)(H,69,79)(H,70,75)(H4,60,61,63)/t43-,44+,45+,46-,47-,48+,49-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Activity of cyclic alpha MSH analogues at human melanocortin receptor Melanocortin 5 receptor |
J Med Chem 45: 5287-94 (2002)
BindingDB Entry DOI: 10.7270/Q20G3KW8 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | CHEMBL5282523
| PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
UniChem
| | n/a | n/a | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50211377
(Ac-Nle4-cyclo(Asp5-His6-D-4,4'-Bip-Pro8-Trp9-Lys10...)Show SMILES CCCC[C@H](NC(C)=O)C(=O)C[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2cc3ccccc3[nH]2)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](Cc2ccc(cc2)-c2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O Show InChI InChI=1S/C56H69N11O9/c1-3-4-16-43(61-34(2)68)49(69)28-39-29-50(70)59-24-11-10-18-44(51(57)71)63-53(73)45(30-40-27-38-15-8-9-17-42(38)62-40)65-55(75)48-19-12-25-67(48)56(76)47(26-35-20-22-37(23-21-35)36-13-6-5-7-14-36)66-54(74)46(64-52(39)72)31-41-32-58-33-60-41/h5-9,13-15,17,20-23,27,32-33,39,43-48,62H,3-4,10-12,16,18-19,24-26,28-31H2,1-2H3,(H2,57,71)(H,58,60)(H,59,70)(H,61,68)(H,63,73)(H,64,72)(H,65,75)(H,66,74)/t39-,43-,44-,45-,46-,47+,48-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.5 | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human MC5R expressed in CHO cells assessed as cAMP accumulation |
J Med Chem 50: 2520-6 (2007)
Article DOI: 10.1021/jm0614275
BindingDB Entry DOI: 10.7270/Q2JQ10QM |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50134954
(CHEMBL439128 | H-Tyr-Val-Nle-c[Cys-His-Phe-Arg-Trp...)Show SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(C)C)C(=O)N[C@@H]1CSSC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)CNC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O Show InChI InChI=1S/C77H105N23O14S2/c1-4-5-21-53(94-75(114)64(43(2)3)100-65(104)51(78)31-46-25-27-49(101)28-26-46)70(109)98-60-40-115-116-41-61(74(113)93-55(24-15-30-86-77(82)83)68(107)95-56(66(105)88-38-62(79)102)32-44-16-8-6-9-17-44)99-72(111)58(34-47-36-87-52-22-13-12-20-50(47)52)97-69(108)54(23-14-29-85-76(80)81)92-71(110)57(33-45-18-10-7-11-19-45)96-73(112)59(35-48-37-84-42-90-48)91-63(103)39-89-67(60)106/h6-13,16-20,22,25-28,36-37,42-43,51,53-61,64,87,101H,4-5,14-15,21,23-24,29-35,38-41,78H2,1-3H3,(H2,79,102)(H,84,90)(H,88,105)(H,89,106)(H,91,103)(H,92,110)(H,93,113)(H,94,114)(H,95,107)(H,96,112)(H,97,108)(H,98,109)(H,99,111)(H,100,104)(H4,80,81,85)(H4,82,83,86)/t51-,53-,54-,55-,56-,57+,58-,59-,60+,61-,64-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Intracellular cAMP accumulation in human Melanocortin 5 receptor functional assay. |
J Med Chem 46: 4965-73 (2003)
Article DOI: 10.1021/jm030119t
BindingDB Entry DOI: 10.7270/Q2SJ1K0P |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 5
(Homo sapiens (Human)) | BDBM50027084

(Melatonan)Show SMILES CCCC[C@H](NC(C)=O)C(=O)N[C@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnc[nH]2)NC1=O)C(N)=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.30 | n/a | n/a | n/a | n/a |
Universit£ di Napoli Federico II
Curated by ChEMBL
| Assay Description
Agonist activity at human melanocortin 5 receptor expressed in HEK293 cells assessed as intracellular cAMP accumulation incubated for 3 mins in prese... |
J Med Chem 58: 9773-8 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01285
BindingDB Entry DOI: 10.7270/Q24Q7Z0B |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

















































