

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histone-lysine N-methyltransferase, H3 lysine-79 specific (Homo sapiens (Human)) | BDBM121392 (US8722877, 17) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | US Patent | n/a | n/a | <1 | n/a | n/a | n/a | n/a | 8.0 | 25 |
Epizyme, Inc. US Patent | Assay Description Compound was serially diluted 3 fold in DMSO for 10 points and 1 μl was plated in a 384 well microtiter plate. Positive control (100% inhibition... | US Patent US8722877 (2014) BindingDB Entry DOI: 10.7270/Q2X63KKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone-lysine N-methyltransferase, H3 lysine-79 specific (Homo sapiens (Human)) | BDBM121391 (US8722877, 9) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 6 | n/a | n/a | n/a | n/a | 8.0 | 25 |
Epizyme, Inc. US Patent | Assay Description Compound was serially diluted 3 fold in DMSO for 10 points and 1 μl was plated in a 384 well microtiter plate. Positive control (100% inhibition... | US Patent US8722877 (2014) BindingDB Entry DOI: 10.7270/Q2X63KKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone-lysine N-methyltransferase, H3 lysine-79 specific (Homo sapiens (Human)) | BDBM121393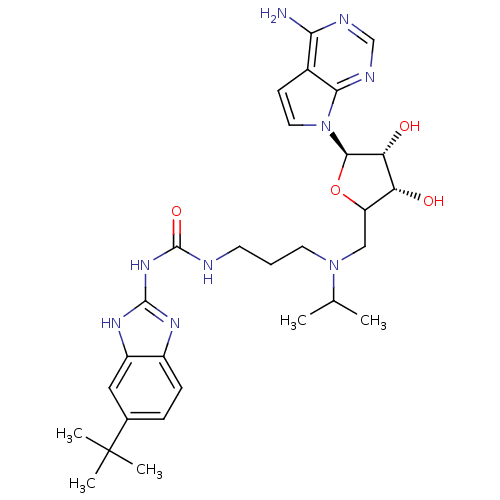 (US8722877, 18) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | <10 | n/a | n/a | n/a | n/a | 8.0 | 25 |
Epizyme, Inc. US Patent | Assay Description Compound was serially diluted 3 fold in DMSO for 10 points and 1 μl was plated in a 384 well microtiter plate. Positive control (100% inhibition... | US Patent US8722877 (2014) BindingDB Entry DOI: 10.7270/Q2X63KKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone-lysine N-methyltransferase, H3 lysine-79 specific (Homo sapiens (Human)) | BDBM121394 (US8722877, 20) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | <1.00E+3 | n/a | n/a | n/a | n/a | 8.0 | 25 |
Epizyme, Inc. US Patent | Assay Description Compound was serially diluted 3 fold in DMSO for 10 points and 1 μl was plated in a 384 well microtiter plate. Positive control (100% inhibition... | US Patent US8722877 (2014) BindingDB Entry DOI: 10.7270/Q2X63KKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone-lysine N-methyltransferase, H3 lysine-79 specific (Homo sapiens (Human)) | BDBM121395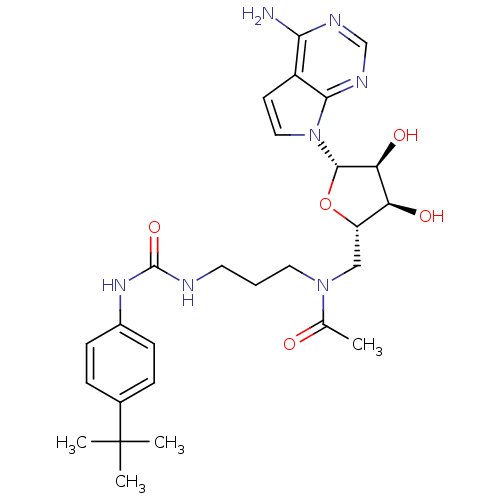 (US8722877, 21) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | US Patent | n/a | n/a | <1.00E+3 | n/a | n/a | n/a | n/a | 8.0 | 25 |
Epizyme, Inc. US Patent | Assay Description Compound was serially diluted 3 fold in DMSO for 10 points and 1 μl was plated in a 384 well microtiter plate. Positive control (100% inhibition... | US Patent US8722877 (2014) BindingDB Entry DOI: 10.7270/Q2X63KKV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||