

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036257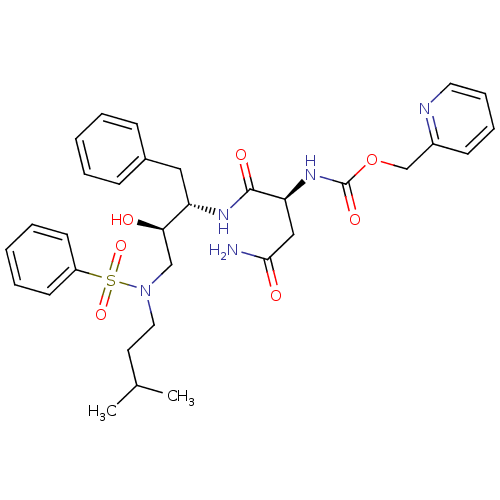 (((S)-1-{(1S,2R)-3-[Benzenesulfonyl-(3-methyl-butyl...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036261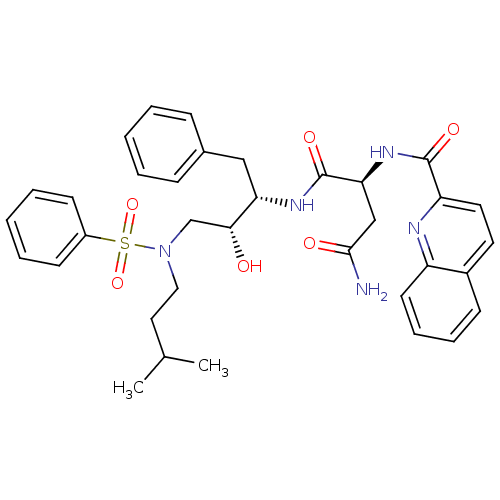 ((S)-N*1*-{(1S,2R)-3-[Benzenesulfonyl-(3-methyl-but...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined in vitro | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036251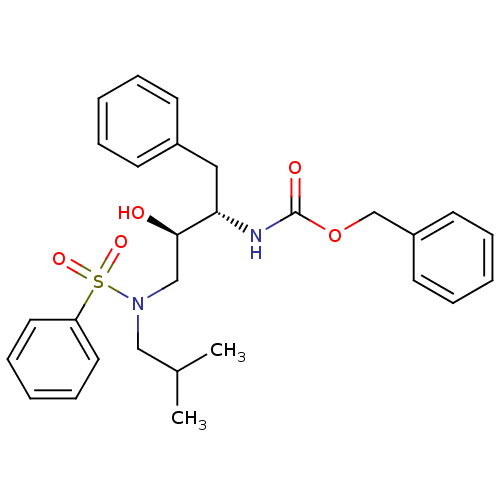 (CHEMBL141265 | [(1S,2R)-3-(Benzenesulfonyl-isobuty...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined in vitro | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036252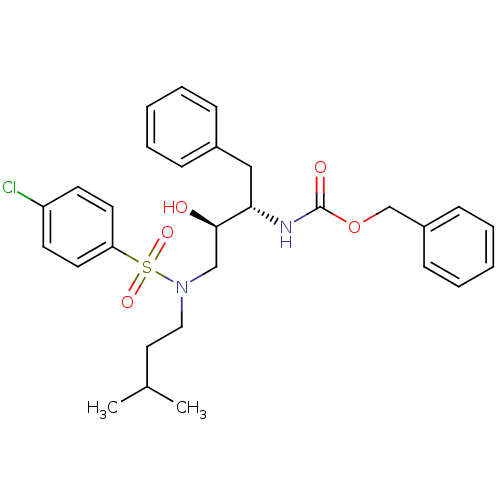 (CHEMBL347671 | {(1S,2R)-1-Benzyl-3-[(4-chloro-benz...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036262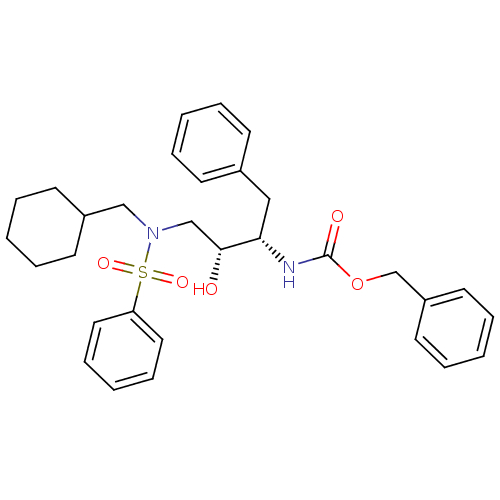 (CHEMBL348282 | [(1S,2R)-3-(Benzenesulfonyl-cyclohe...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036253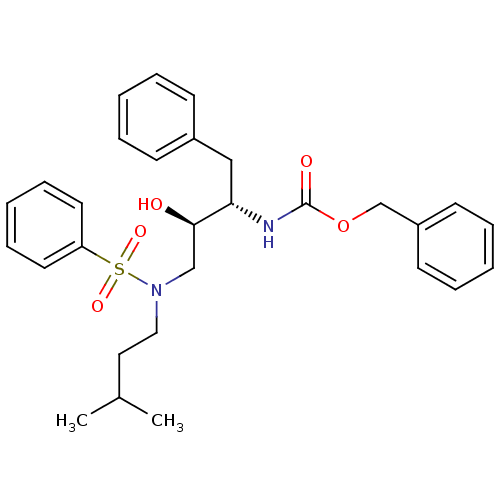 (CHEMBL347431 | {(1S,2R)-3-[Benzenesulfonyl-(3-meth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined in vitro. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036259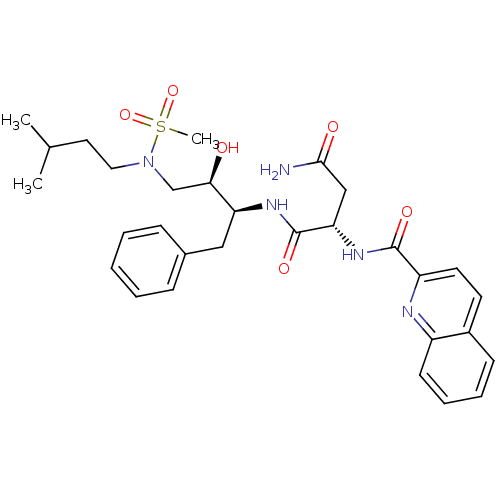 ((S)-N*1*-{(1S,2R)-1-Benzyl-2-hydroxy-3-[methanesul...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined in vitro | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036258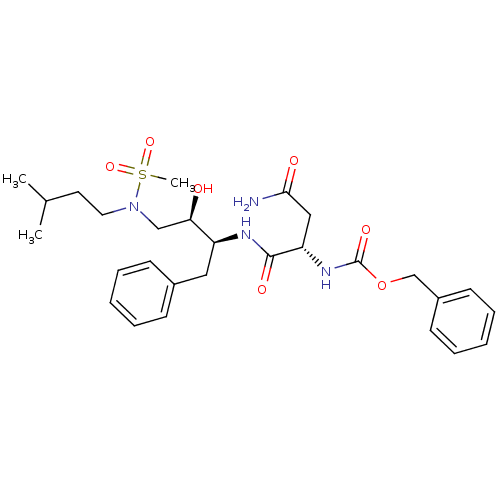 (((S)-1-{(1S,2R)-1-Benzyl-2-hydroxy-3-[methanesulfo...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 57 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036260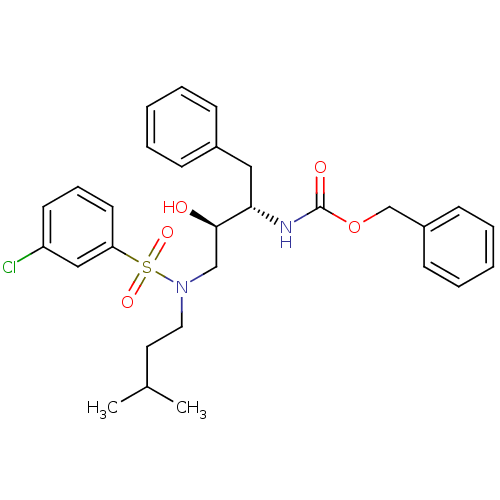 (CHEMBL156424 | {(1S,2R)-1-Benzyl-3-[(3-chloro-benz...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 61 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036255 (CHEMBL154197 | [(1S,2R)-3-(Benzenesulfonyl-benzyl-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 125 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036254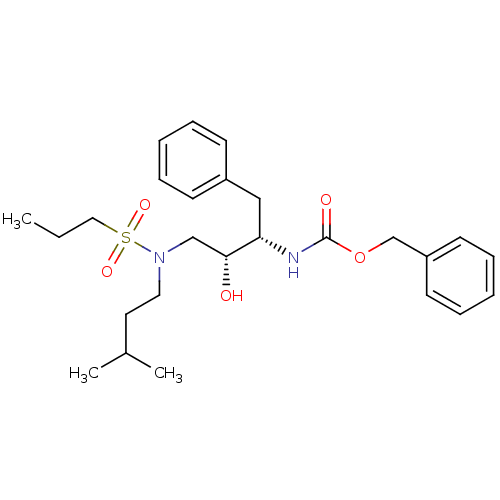 (CHEMBL155051 | {(1S,2R)-1-Benzyl-2-hydroxy-3-[(3-m...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 128 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036264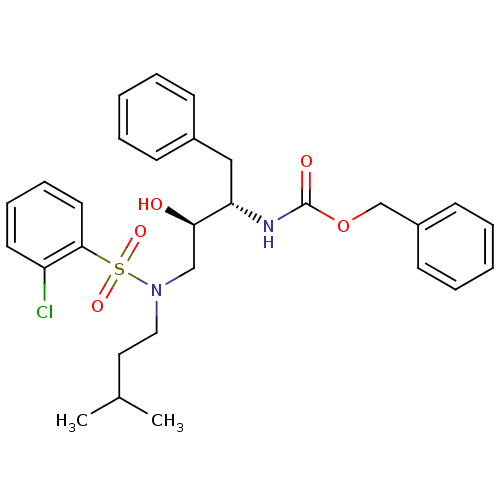 (CHEMBL154600 | {(1S,2R)-1-Benzyl-3-[(2-chloro-benz...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 227 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036263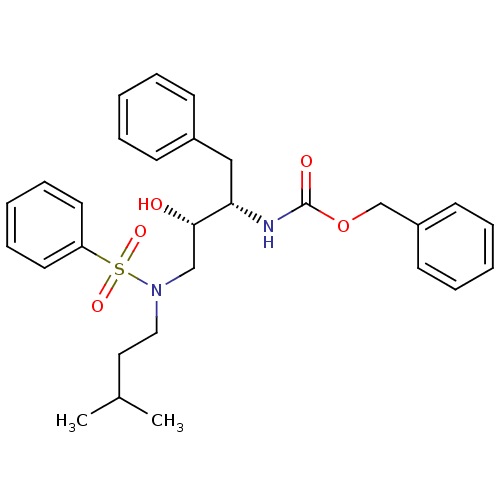 (CHEMBL154638 | {(1S,2S)-3-[Benzenesulfonyl-(3-meth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 295 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036256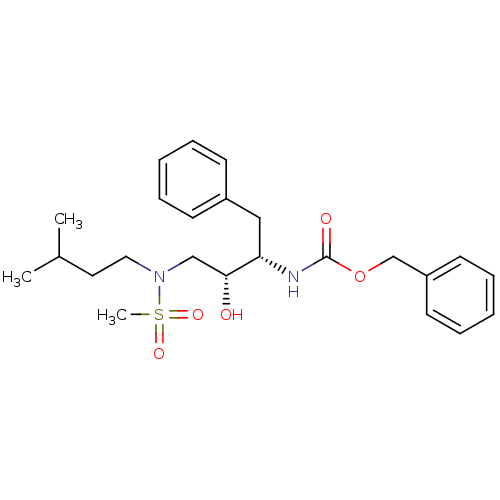 (CHEMBL154798 | {(1S,2R)-1-Benzyl-2-hydroxy-3-[meth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.87E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Binding affinity of the compound for HIV-1 protease was determined | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036261 ((S)-N*1*-{(1S,2R)-3-[Benzenesulfonyl-(3-methyl-but...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036257 (((S)-1-{(1S,2R)-3-[Benzenesulfonyl-(3-methyl-butyl...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036251 (CHEMBL141265 | [(1S,2R)-3-(Benzenesulfonyl-isobuty...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036252 (CHEMBL347671 | {(1S,2R)-1-Benzyl-3-[(4-chloro-benz...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036262 (CHEMBL348282 | [(1S,2R)-3-(Benzenesulfonyl-cyclohe...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036253 (CHEMBL347431 | {(1S,2R)-3-[Benzenesulfonyl-(3-meth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036259 ((S)-N*1*-{(1S,2R)-1-Benzyl-2-hydroxy-3-[methanesul...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036260 (CHEMBL156424 | {(1S,2R)-1-Benzyl-3-[(3-chloro-benz...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 92 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036258 (((S)-1-{(1S,2R)-1-Benzyl-2-hydroxy-3-[methanesulfo...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036255 (CHEMBL154197 | [(1S,2R)-3-(Benzenesulfonyl-benzyl-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036254 (CHEMBL155051 | {(1S,2R)-1-Benzyl-2-hydroxy-3-[(3-m...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036264 (CHEMBL154600 | {(1S,2R)-1-Benzyl-3-[(2-chloro-benz...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 380 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036263 (CHEMBL154638 | {(1S,2S)-3-[Benzenesulfonyl-(3-meth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036256 (CHEMBL154798 | {(1S,2R)-1-Benzyl-2-hydroxy-3-[meth...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50036265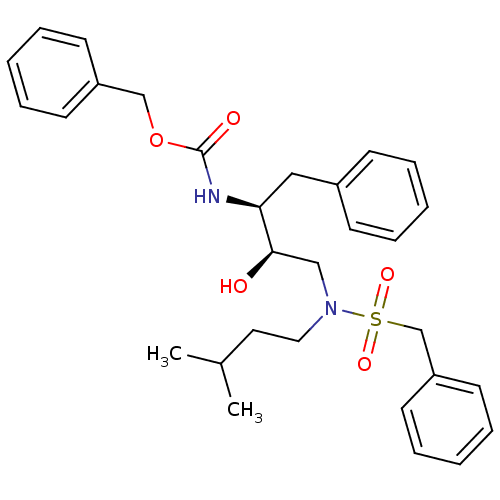 (CHEMBL155260 | {(1S,2R)-1-Benzyl-2-hydroxy-3-[(3-m...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research Curated by ChEMBL | Assay Description Ability of the compound to inhibit HIV-1 protease was determined using spectrofluorometric assay. | J Med Chem 38: 581-4 (1995) BindingDB Entry DOI: 10.7270/Q2SF2V61 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||