

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070535 (1-[(1R,2R)-2-(6-Chloro-3-ethoxy-2-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070536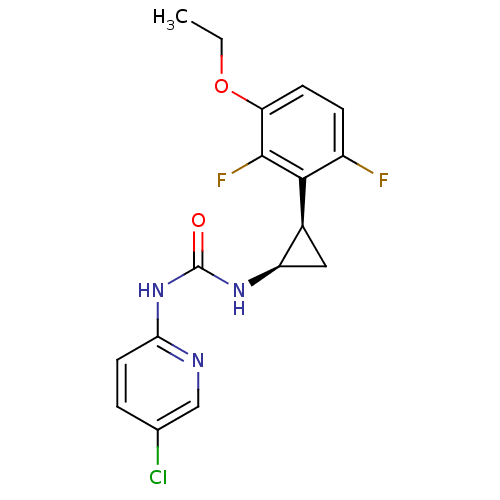 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070533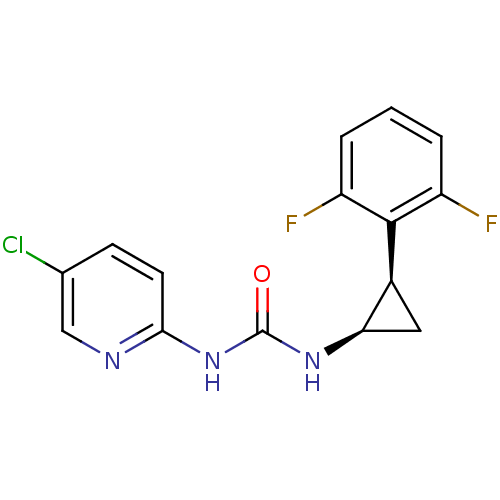 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(2,6-difluo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070531 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(2-fluoro-3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Compound was tested for its inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070537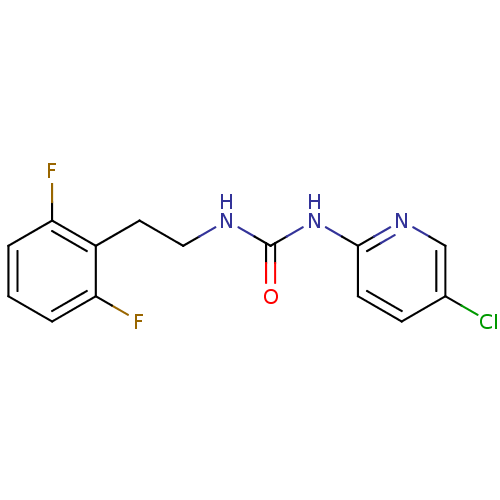 (1-(5-Chloro-pyridin-2-yl)-3-[2-(2,6-difluoro-pheny...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2717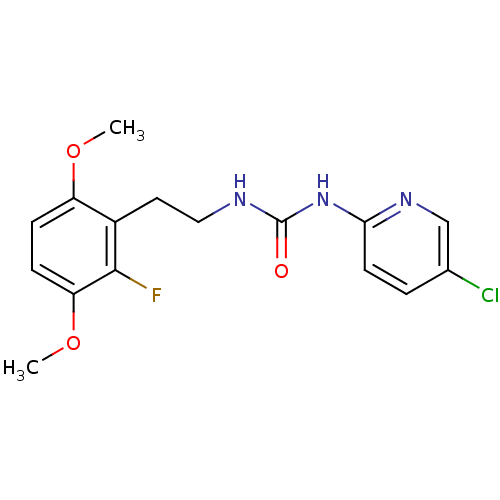 (1-(5-chloropyridin-2-yl)-3-[2-(2-fluoro-3,6-dimeth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2860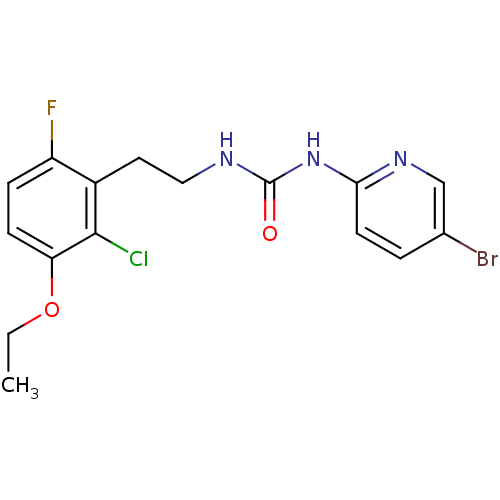 (1-(5-bromopyridin-2-yl)-3-[2-(2-chloro-3-ethoxy-6-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070530 (1-[(1R,2R)-2-(2-Chloro-3-ethoxy-6-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070532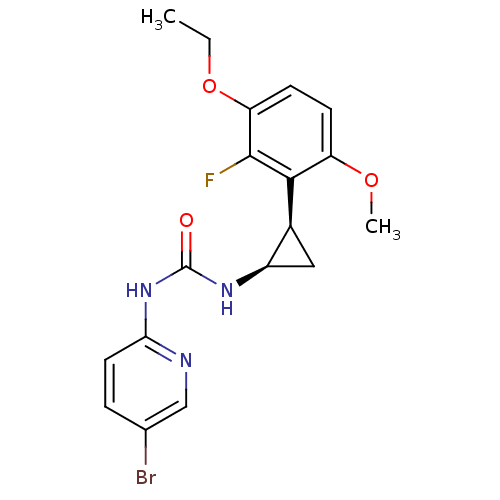 (1-(5-Bromo-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070534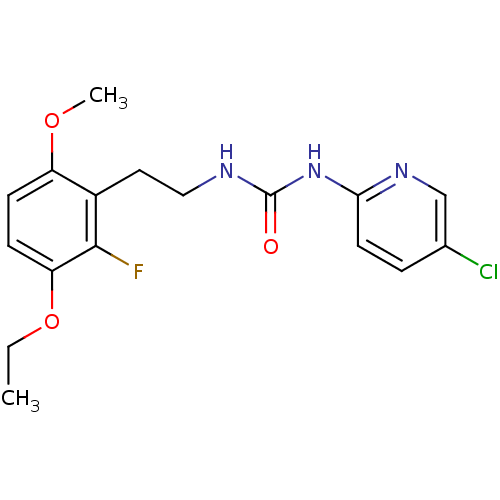 (1-(5-Chloro-pyridin-2-yl)-3-[2-(3-ethoxy-2-fluoro-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070538 (1-[(1R,2R)-2-(2-Chloro-3-ethoxy-6-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1866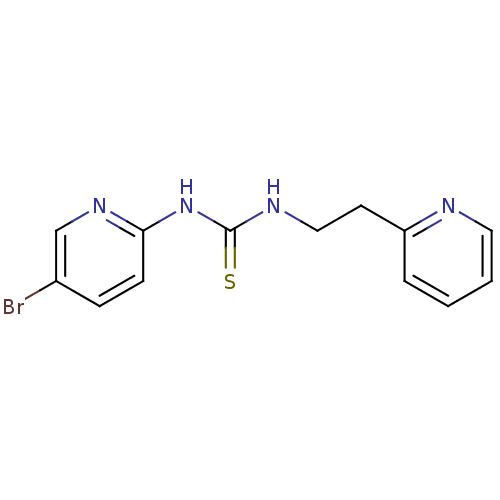 (3-(5-bromopyridin-2-yl)-1-[2-(pyridin-2-yl)ethyl]t...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070536 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070535 (1-[(1R,2R)-2-(6-Chloro-3-ethoxy-2-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2716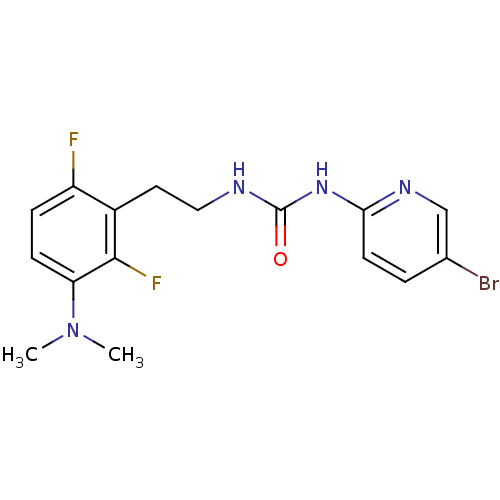 (1-(5-bromopyridin-2-yl)-3-{2-[3-(dimethylamino)-2,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070533 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(2,6-difluo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070538 (1-[(1R,2R)-2-(2-Chloro-3-ethoxy-6-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070535 (1-[(1R,2R)-2-(6-Chloro-3-ethoxy-2-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070530 (1-[(1R,2R)-2-(2-Chloro-3-ethoxy-6-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070530 (1-[(1R,2R)-2-(2-Chloro-3-ethoxy-6-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070538 (1-[(1R,2R)-2-(2-Chloro-3-ethoxy-6-fluoro-phenyl)-c...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070536 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070532 (1-(5-Bromo-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1317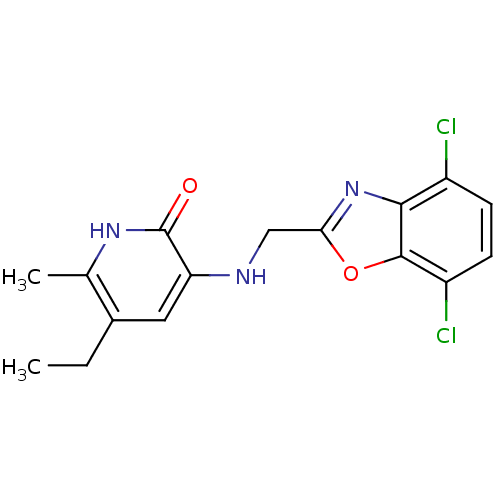 (3-[[(4,7-Dichlorobenzoxazol-2-yl)-methyl]amino]-5-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2720 (1-(5-chloropyridin-2-yl)-3-[2-(3-acetyl-2-fluoro-6...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070533 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(2,6-difluo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2717 (1-(5-chloropyridin-2-yl)-3-[2-(2-fluoro-3,6-dimeth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2717 (1-(5-chloropyridin-2-yl)-3-[2-(2-fluoro-3,6-dimeth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434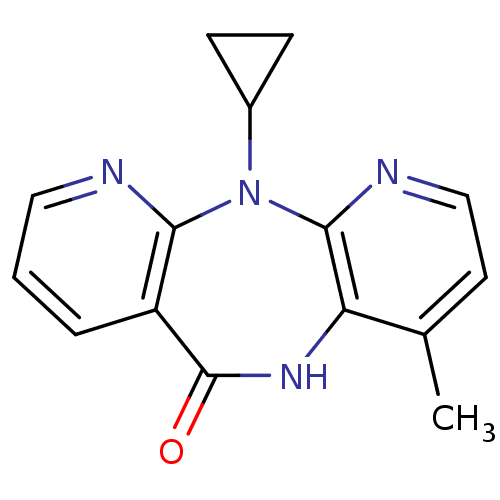 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070539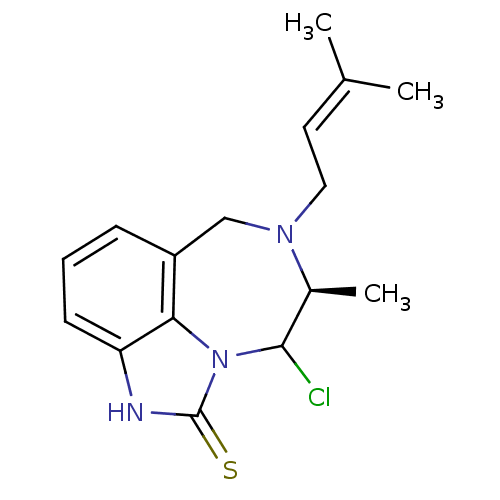 ((S)-9-Chloro-8-methyl-7-(3-methyl-but-2-enyl)-6,7,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Compound was tested for its inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070532 (1-(5-Bromo-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2860 (1-(5-bromopyridin-2-yl)-3-[2-(2-chloro-3-ethoxy-6-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070531 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(2-fluoro-3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070531 (1-(5-Chloro-pyridin-2-yl)-3-[(1R,2R)-2-(2-fluoro-3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070537 (1-(5-Chloro-pyridin-2-yl)-3-[2-(2,6-difluoro-pheny...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Compound was tested for its inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070537 (1-(5-Chloro-pyridin-2-yl)-3-[2-(2,6-difluoro-pheny...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070534 (1-(5-Chloro-pyridin-2-yl)-3-[2-(3-ethoxy-2-fluoro-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1866 (3-(5-bromopyridin-2-yl)-1-[2-(pyridin-2-yl)ethyl]t...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1894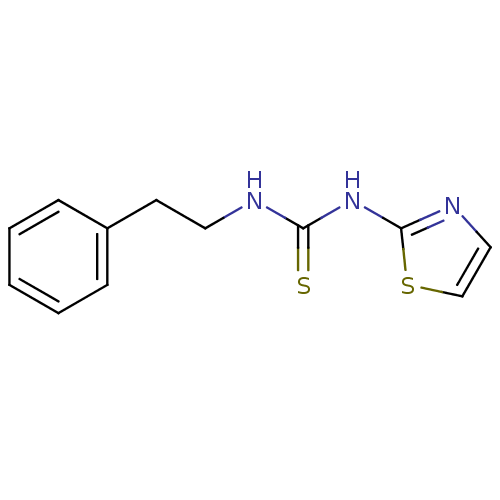 (1-(2-phenylethyl)-3-1,3-thiazol-2-ylthiourea | CHE...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Antiviral activity was determined against HIV- 1 reverse transcriptase | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2720 (1-(5-chloropyridin-2-yl)-3-[2-(3-acetyl-2-fluoro-6...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2716 (1-(5-bromopyridin-2-yl)-3-{2-[3-(dimethylamino)-2,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1317 (3-[[(4,7-Dichlorobenzoxazol-2-yl)-methyl]amino]-5-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2860 (1-(5-bromopyridin-2-yl)-3-[2-(2-chloro-3-ethoxy-6-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070534 (1-(5-Chloro-pyridin-2-yl)-3-[2-(3-ethoxy-2-fluoro-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2716 (1-(5-bromopyridin-2-yl)-3-{2-[3-(dimethylamino)-2,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | >2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1866 (3-(5-bromopyridin-2-yl)-1-[2-(pyridin-2-yl)ethyl]t...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2720 (1-(5-chloropyridin-2-yl)-3-[2-(3-acetyl-2-fluoro-6...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070539 ((S)-9-Chloro-8-methyl-7-(3-methyl-but-2-enyl)-6,7,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Tyr 181 to Cys 181 (clone 90) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1434 (11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido[3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on recombinant HIV- 1 reverse transcriptase which has a mutation Leu 100 to Ile 100 (clone 118) | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1317 (3-[[(4,7-Dichlorobenzoxazol-2-yl)-methyl]amino]-5-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB Curated by ChEMBL | Assay Description Inhibitory effect on wild type HIV- 1 reverse transcriptase using rCdG as template and dGTP as substrate. | Bioorg Med Chem Lett 8: 1511-6 (1999) BindingDB Entry DOI: 10.7270/Q2RR1XDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 52 total ) | Next | Last >> |