 Found 91 hits Enz. Inhib. hit(s) with all data for entry = 50032994
Found 91 hits Enz. Inhib. hit(s) with all data for entry = 50032994 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337654
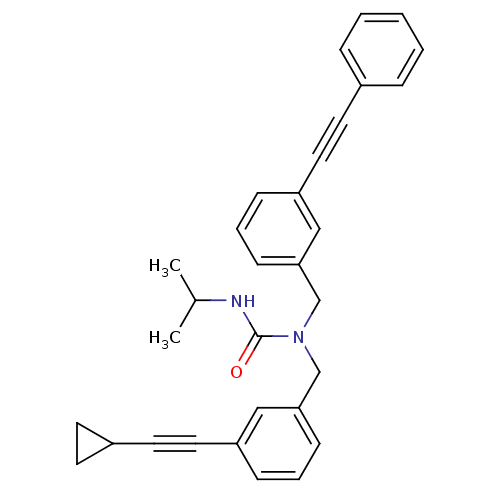
(1-(3-(cyclopropylethynyl)benzyl)-3-isopropyl-1-(3-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#CC1CC1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C31H30N2O/c1-24(2)32-31(34)33(23-30-13-7-11-28(21-30)19-17-26-14-15-26)22-29-12-6-10-27(20-29)18-16-25-8-4-3-5-9-25/h3-13,20-21,24,26H,14-15,22-23H2,1-2H3,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337659
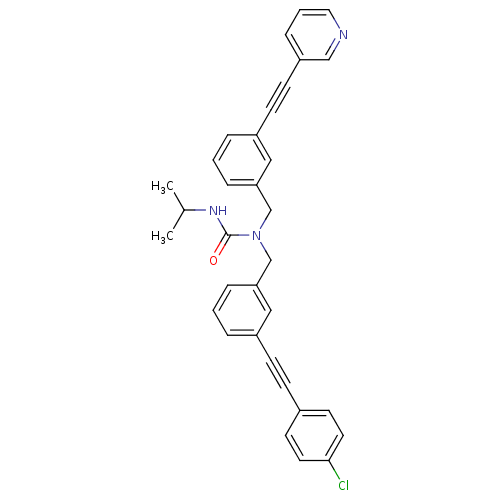
(1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-isopropyl-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccc(Cl)cc1)Cc1cccc(c1)C#Cc1cccnc1 Show InChI InChI=1S/C33H28ClN3O/c1-25(2)36-33(38)37(24-31-9-4-7-28(21-31)13-14-29-10-5-19-35-22-29)23-30-8-3-6-27(20-30)12-11-26-15-17-32(34)18-16-26/h3-10,15-22,25H,23-24H2,1-2H3,(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50227631
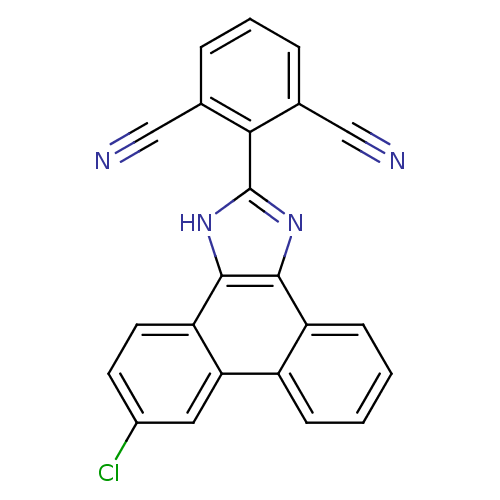
(2-(6-chloro-1H-phenanthro[9,10-d]imidazol-2-yl)iso...)Show SMILES Clc1ccc2c3[nH]c(nc3c3ccccc3c2c1)-c1c(cccc1C#N)C#N Show InChI InChI=1S/C23H11ClN4/c24-15-8-9-18-19(10-15)16-6-1-2-7-17(16)21-22(18)28-23(27-21)20-13(11-25)4-3-5-14(20)12-26/h1-10H,(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337658
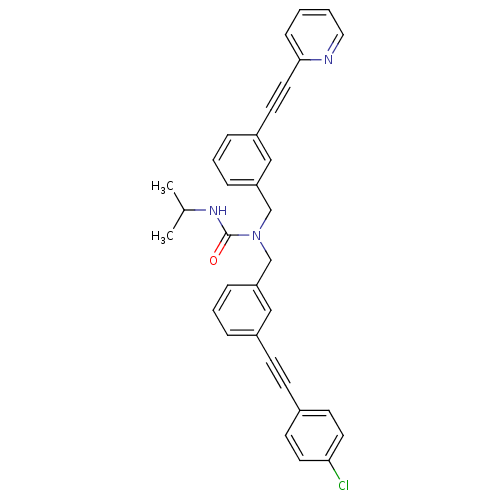
(1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-isopropyl-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccc(Cl)cc1)Cc1cccc(c1)C#Cc1ccccn1 Show InChI InChI=1S/C33H28ClN3O/c1-25(2)36-33(38)37(24-30-10-6-8-28(22-30)16-19-32-11-3-4-20-35-32)23-29-9-5-7-27(21-29)13-12-26-14-17-31(34)18-15-26/h3-11,14-15,17-18,20-22,25H,23-24H2,1-2H3,(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337661
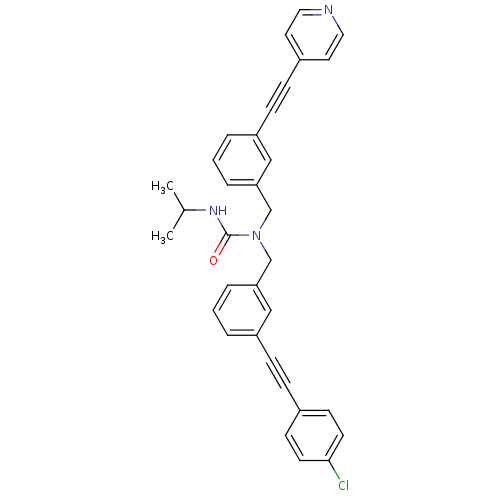
(1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-isopropyl-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccncc1)Cc1cccc(c1)C#Cc1ccc(Cl)cc1 Show InChI InChI=1S/C33H28ClN3O/c1-25(2)36-33(38)37(24-31-8-4-6-29(22-31)12-10-27-17-19-35-20-18-27)23-30-7-3-5-28(21-30)11-9-26-13-15-32(34)16-14-26/h3-8,13-22,25H,23-24H2,1-2H3,(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337657
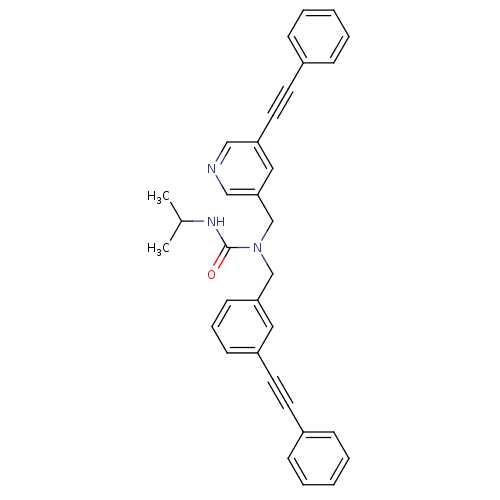
(3-isopropyl-1-(3-(phenylethynyl)benzyl)-1-((5-(phe...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cncc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C33H29N3O/c1-26(2)35-33(37)36(24-31-15-9-14-29(20-31)18-16-27-10-5-3-6-11-27)25-32-21-30(22-34-23-32)19-17-28-12-7-4-8-13-28/h3-15,20-23,26H,24-25H2,1-2H3,(H,35,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337653

(1-(biphenyl-3-ylmethyl)-3-isopropyl-1-(3-(phenylet...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cccc(c1)-c1ccccc1 Show InChI InChI=1S/C32H30N2O/c1-25(2)33-32(35)34(24-29-15-10-18-31(22-29)30-16-7-4-8-17-30)23-28-14-9-13-27(21-28)20-19-26-11-5-3-6-12-26/h3-18,21-22,25H,23-24H2,1-2H3,(H,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337660
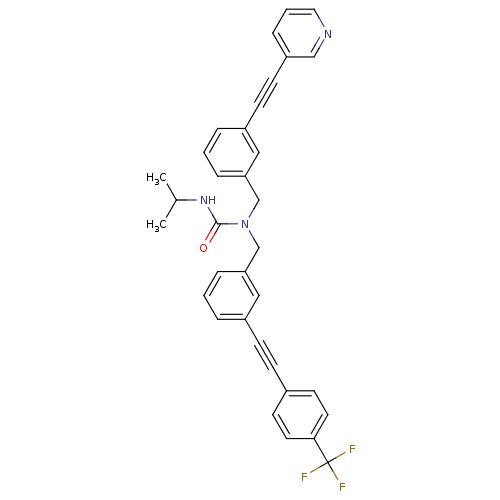
(3-isopropyl-1-(3-(pyridin-3-ylethynyl)benzyl)-1-(3...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccc(cc1)C(F)(F)F)Cc1cccc(c1)C#Cc1cccnc1 Show InChI InChI=1S/C34H28F3N3O/c1-25(2)39-33(41)40(24-31-9-4-7-28(21-31)13-14-29-10-5-19-38-22-29)23-30-8-3-6-27(20-30)12-11-26-15-17-32(18-16-26)34(35,36)37/h3-10,15-22,25H,23-24H2,1-2H3,(H,39,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337656
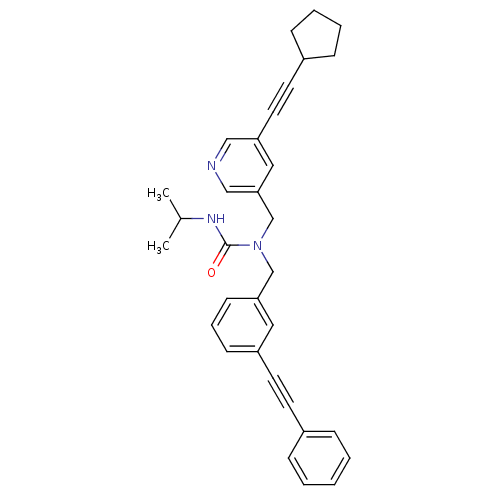
(1-((5-(cyclopentylethynyl)pyridin-3-yl)methyl)-3-i...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cncc(c1)C#CC1CCCC1 Show InChI InChI=1S/C32H33N3O/c1-25(2)34-32(36)35(24-31-20-29(21-33-22-31)18-16-27-11-6-7-12-27)23-30-14-8-13-28(19-30)17-15-26-9-4-3-5-10-26/h3-5,8-10,13-14,19-22,25,27H,6-7,11-12,23-24H2,1-2H3,(H,34,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337662
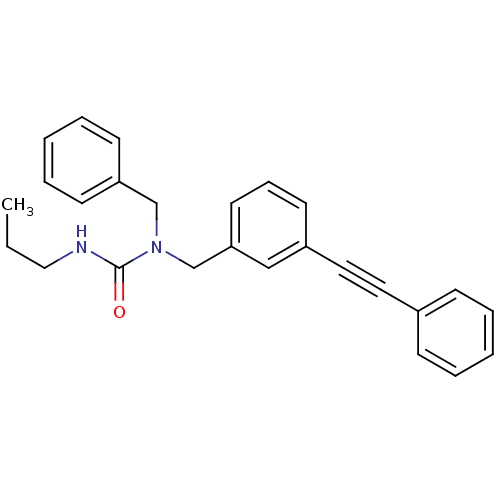
(1-benzyl-1-(3-(phenylethynyl)benzyl)-3-propylurea ...)Show SMILES CCCNC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C26H26N2O/c1-2-18-27-26(29)28(20-24-12-7-4-8-13-24)21-25-15-9-14-23(19-25)17-16-22-10-5-3-6-11-22/h3-15,19H,2,18,20-21H2,1H3,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337641
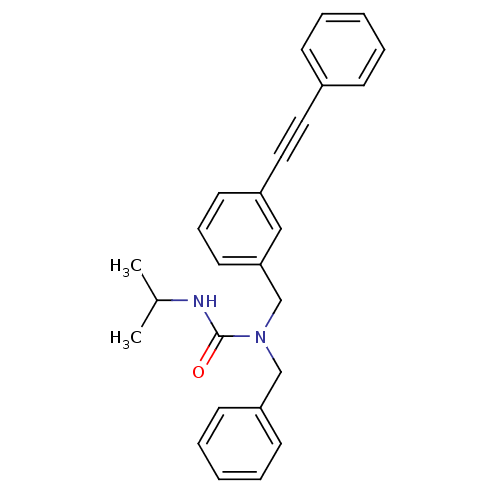
(1-benzyl-3-isopropyl-1-(3-(phenylethynyl)benzyl)ur...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C26H26N2O/c1-21(2)27-26(29)28(19-24-12-7-4-8-13-24)20-25-15-9-14-23(18-25)17-16-22-10-5-3-6-11-22/h3-15,18,21H,19-20H2,1-2H3,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337646
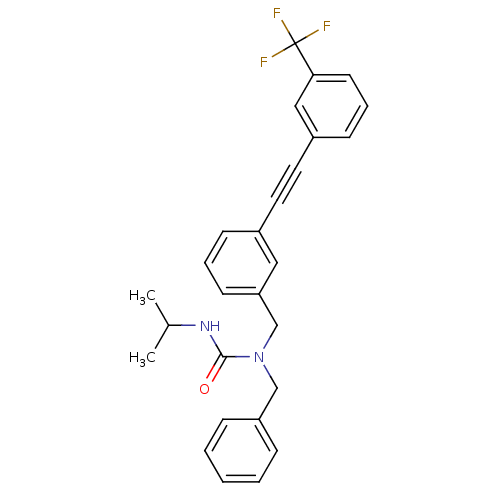
(1-benzyl-3-isopropyl-1-(3-((3-(trifluoromethyl)phe...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1cccc(c1)C(F)(F)F Show InChI InChI=1S/C27H25F3N2O/c1-20(2)31-26(33)32(18-23-8-4-3-5-9-23)19-24-12-6-10-21(16-24)14-15-22-11-7-13-25(17-22)27(28,29)30/h3-13,16-17,20H,18-19H2,1-2H3,(H,31,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337663

(1-benzyl-3-cyclobutyl-1-(3-(phenylethynyl)benzyl)u...)Show SMILES O=C(NC1CCC1)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C27H26N2O/c30-27(28-26-15-8-16-26)29(20-24-11-5-2-6-12-24)21-25-14-7-13-23(19-25)18-17-22-9-3-1-4-10-22/h1-7,9-14,19,26H,8,15-16,20-21H2,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337647

(1-benzyl-3-isopropyl-1-(3-((4-(trifluoromethyl)phe...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C27H25F3N2O/c1-20(2)31-26(33)32(18-23-7-4-3-5-8-23)19-24-10-6-9-22(17-24)12-11-21-13-15-25(16-14-21)27(28,29)30/h3-10,13-17,20H,18-19H2,1-2H3,(H,31,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337644
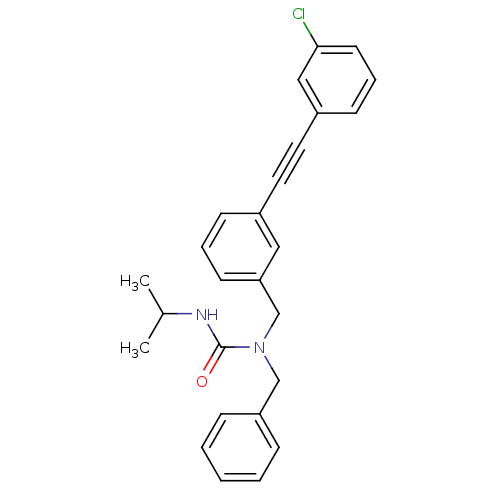
(1-benzyl-1-(3-((3-chlorophenyl)ethynyl)benzyl)-3-i...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1cccc(Cl)c1 Show InChI InChI=1S/C26H25ClN2O/c1-20(2)28-26(30)29(18-23-8-4-3-5-9-23)19-24-12-6-10-21(16-24)14-15-22-11-7-13-25(27)17-22/h3-13,16-17,20H,18-19H2,1-2H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337652

(3-isopropyl-1-(3-methylbenzyl)-1-(3-(phenylethynyl...)Show SMILES CC(C)NC(=O)N(Cc1cccc(C)c1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C27H28N2O/c1-21(2)28-27(30)29(19-25-13-7-9-22(3)17-25)20-26-14-8-12-24(18-26)16-15-23-10-5-4-6-11-23/h4-14,17-18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337655

(1-((5-(cyclopropylethynyl)pyridin-3-yl)methyl)-3-i...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cncc(c1)C#CC1CC1 Show InChI InChI=1S/C30H29N3O/c1-23(2)32-30(34)33(22-29-18-27(19-31-20-29)16-14-25-11-12-25)21-28-10-6-9-26(17-28)15-13-24-7-4-3-5-8-24/h3-10,17-20,23,25H,11-12,21-22H2,1-2H3,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337651
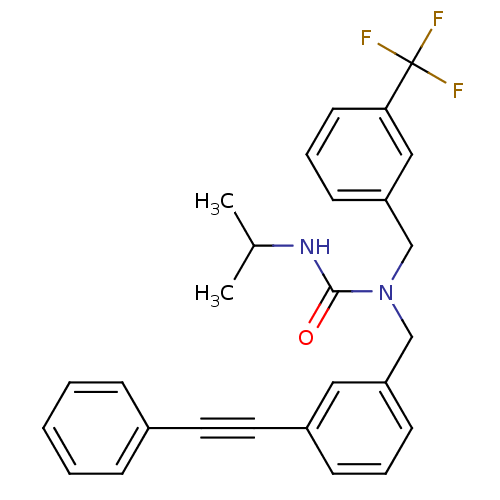
(3-isopropyl-1-(3-(phenylethynyl)benzyl)-1-(3-(trif...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cccc(c1)C(F)(F)F Show InChI InChI=1S/C27H25F3N2O/c1-20(2)31-26(33)32(19-24-12-7-13-25(17-24)27(28,29)30)18-23-11-6-10-22(16-23)15-14-21-8-4-3-5-9-21/h3-13,16-17,20H,18-19H2,1-2H3,(H,31,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337645
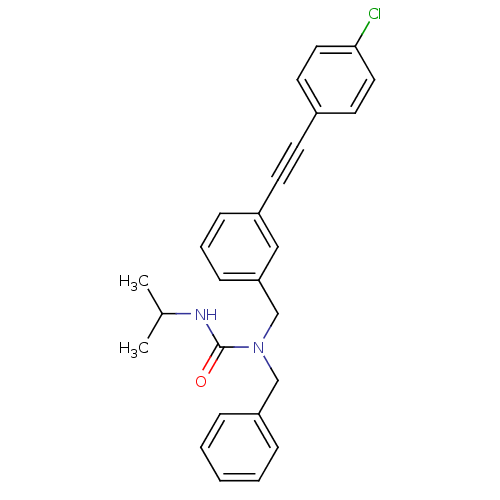
(1-benzyl-1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-i...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccc(Cl)cc1 Show InChI InChI=1S/C26H25ClN2O/c1-20(2)28-26(30)29(18-23-7-4-3-5-8-23)19-24-10-6-9-22(17-24)12-11-21-13-15-25(27)16-14-21/h3-10,13-17,20H,18-19H2,1-2H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337642
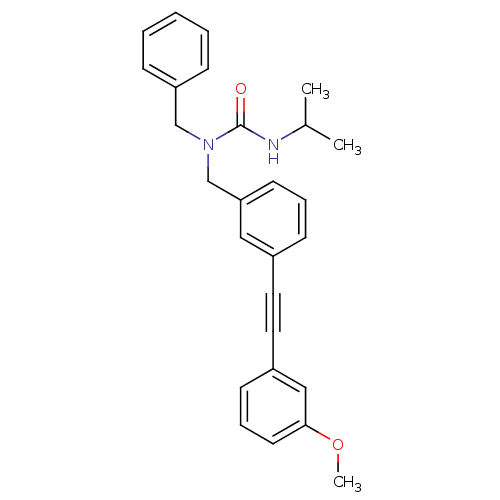
(1-benzyl-3-isopropyl-1-(3-((3-methoxyphenyl)ethyny...)Show SMILES COc1cccc(c1)C#Cc1cccc(CN(Cc2ccccc2)C(=O)NC(C)C)c1 Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(19-24-9-5-4-6-10-24)20-25-13-7-11-22(17-25)15-16-23-12-8-14-26(18-23)31-3/h4-14,17-18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337664

(1-benzyl-3-(pentan-3-yl)-1-(3-(phenylethynyl)benzy...)Show SMILES CCC(CC)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C28H30N2O/c1-3-27(4-2)29-28(31)30(21-25-14-9-6-10-15-25)22-26-17-11-16-24(20-26)19-18-23-12-7-5-8-13-23/h5-17,20,27H,3-4,21-22H2,1-2H3,(H,29,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337665
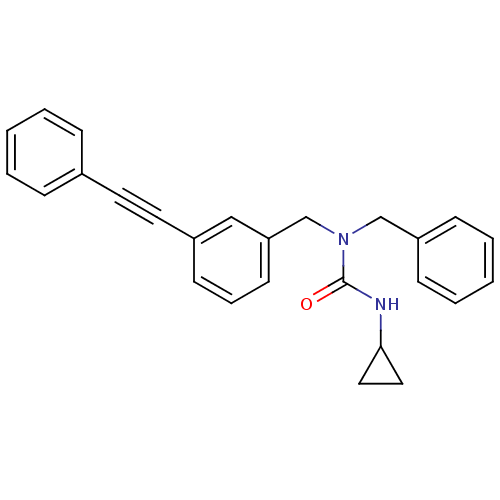
(1-benzyl-3-cyclopropyl-1-(3-(phenylethynyl)benzyl)...)Show SMILES O=C(NC1CC1)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C26H24N2O/c29-26(27-25-16-17-25)28(19-23-10-5-2-6-11-23)20-24-13-7-12-22(18-24)15-14-21-8-3-1-4-9-21/h1-13,18,25H,16-17,19-20H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337670
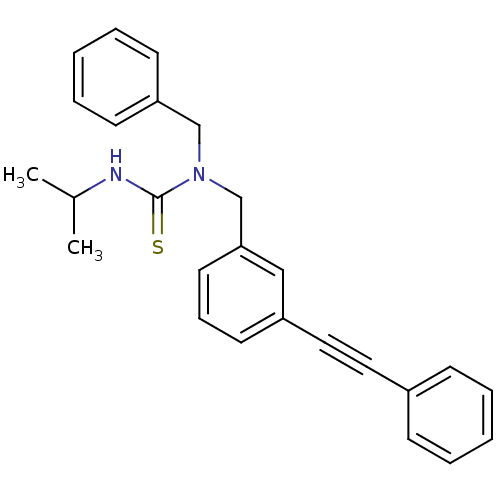
(1-benzyl-3-isopropyl-1-(3-(phenylethynyl)benzyl)th...)Show SMILES CC(C)NC(=S)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C26H26N2S/c1-21(2)27-26(29)28(19-24-12-7-4-8-13-24)20-25-15-9-14-23(18-25)17-16-22-10-5-3-6-11-22/h3-15,18,21H,19-20H2,1-2H3,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337650
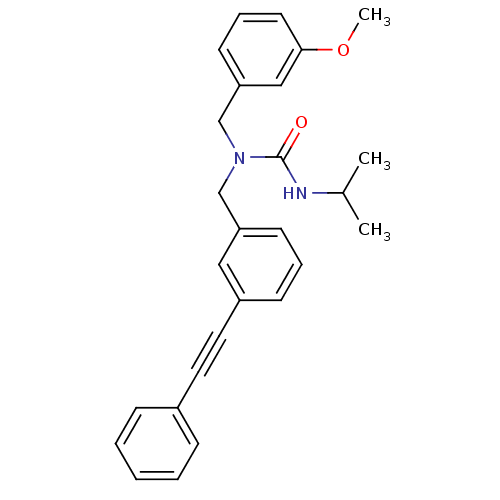
(3-isopropyl-1-(3-methoxybenzyl)-1-(3-(phenylethyny...)Show SMILES COc1cccc(CN(Cc2cccc(c2)C#Cc2ccccc2)C(=O)NC(C)C)c1 Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(20-25-13-8-14-26(18-25)31-3)19-24-12-7-11-23(17-24)16-15-22-9-5-4-6-10-22/h4-14,17-18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337666

(1-benzyl-3-(cyclopropylmethyl)-1-(3-(phenylethynyl...)Show SMILES O=C(NCC1CC1)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C27H26N2O/c30-27(28-19-24-16-17-24)29(20-25-10-5-2-6-11-25)21-26-13-7-12-23(18-26)15-14-22-8-3-1-4-9-22/h1-13,18,24H,16-17,19-21H2,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337643

(1-benzyl-3-isopropyl-1-(3-((4-methoxyphenyl)ethyny...)Show SMILES COc1ccc(cc1)C#Cc1cccc(CN(Cc2ccccc2)C(=O)NC(C)C)c1 Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(19-24-8-5-4-6-9-24)20-25-11-7-10-23(18-25)13-12-22-14-16-26(31-3)17-15-22/h4-11,14-18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 72 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337671
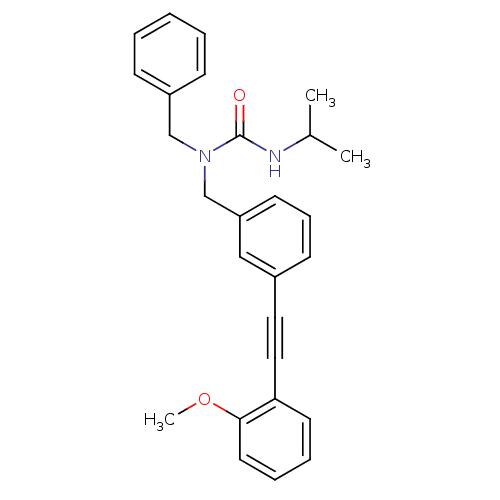
(1-benzyl-3-isopropyl-1-(3-((2-methoxyphenyl)ethyny...)Show SMILES COc1ccccc1C#Cc1cccc(CN(Cc2ccccc2)C(=O)NC(C)C)c1 Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(19-23-10-5-4-6-11-23)20-24-13-9-12-22(18-24)16-17-25-14-7-8-15-26(25)31-3/h4-15,18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337649
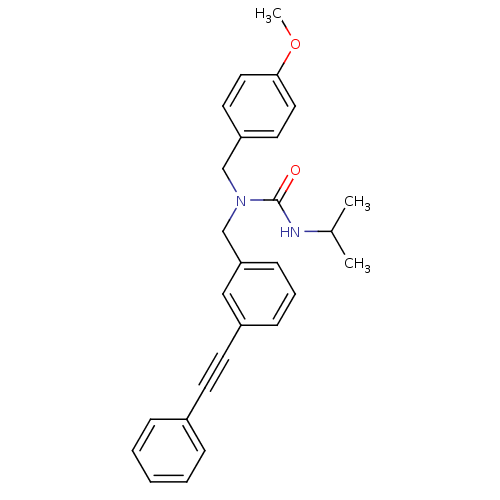
(3-isopropyl-1-(4-methoxybenzyl)-1-(3-(phenylethyny...)Show SMILES COc1ccc(CN(Cc2cccc(c2)C#Cc2ccccc2)C(=O)NC(C)C)cc1 Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(19-24-14-16-26(31-3)17-15-24)20-25-11-7-10-23(18-25)13-12-22-8-5-4-6-9-22/h4-11,14-18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337654

(1-(3-(cyclopropylethynyl)benzyl)-3-isopropyl-1-(3-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#CC1CC1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C31H30N2O/c1-24(2)32-31(34)33(23-30-13-7-11-28(21-30)19-17-26-14-15-26)22-29-12-6-10-27(20-29)18-16-25-8-4-3-5-9-25/h3-13,20-21,24,26H,14-15,22-23H2,1-2H3,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337668
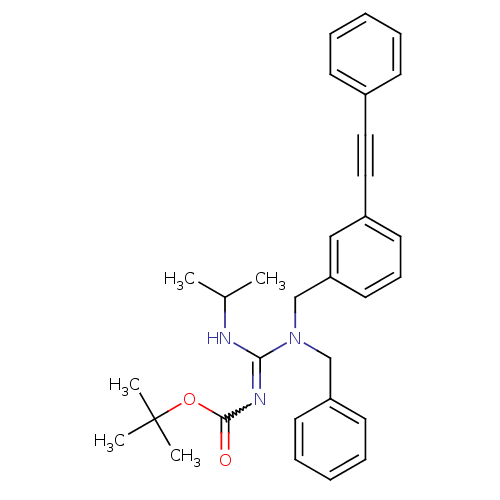
(CHEMBL1683226 | tert-butyl(benzyl(3-(phenylethynyl...)Show SMILES CC(C)NC(=NC(=O)OC(C)(C)C)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 |w:5.5| Show InChI InChI=1S/C31H35N3O2/c1-24(2)32-29(33-30(35)36-31(3,4)5)34(22-27-15-10-7-11-16-27)23-28-18-12-17-26(21-28)20-19-25-13-8-6-9-14-25/h6-18,21,24H,22-23H2,1-5H3,(H,32,33,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337659

(1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-isopropyl-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccc(Cl)cc1)Cc1cccc(c1)C#Cc1cccnc1 Show InChI InChI=1S/C33H28ClN3O/c1-25(2)36-33(38)37(24-31-9-4-7-28(21-31)13-14-29-10-5-19-35-22-29)23-30-8-3-6-27(20-30)12-11-26-15-17-32(34)18-16-26/h3-10,15-22,25H,23-24H2,1-2H3,(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337669

(1-benzyl-2-cyano-3-isopropyl-1-(3-(phenylethynyl)b...)Show SMILES CC(C)NC(=NC#N)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 |w:5.5| Show InChI InChI=1S/C27H26N4/c1-22(2)30-27(29-21-28)31(19-25-12-7-4-8-13-25)20-26-15-9-14-24(18-26)17-16-23-10-5-3-6-11-23/h3-15,18,22H,19-20H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337660

(3-isopropyl-1-(3-(pyridin-3-ylethynyl)benzyl)-1-(3...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccc(cc1)C(F)(F)F)Cc1cccc(c1)C#Cc1cccnc1 Show InChI InChI=1S/C34H28F3N3O/c1-25(2)39-33(41)40(24-31-9-4-7-28(21-31)13-14-29-10-5-19-38-22-29)23-30-8-3-6-27(20-30)12-11-26-15-17-32(18-16-26)34(35,36)37/h3-10,15-22,25H,23-24H2,1-2H3,(H,39,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337661

(1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-isopropyl-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccncc1)Cc1cccc(c1)C#Cc1ccc(Cl)cc1 Show InChI InChI=1S/C33H28ClN3O/c1-25(2)36-33(38)37(24-31-8-4-6-29(22-31)12-10-27-17-19-35-20-18-27)23-30-7-3-5-28(21-30)11-9-26-13-15-32(34)16-14-26/h3-8,13-22,25H,23-24H2,1-2H3,(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337657

(3-isopropyl-1-(3-(phenylethynyl)benzyl)-1-((5-(phe...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cncc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C33H29N3O/c1-26(2)35-33(37)36(24-31-15-9-14-29(20-31)18-16-27-10-5-3-6-11-27)25-32-21-30(22-34-23-32)19-17-28-12-7-4-8-13-28/h3-15,20-23,26H,24-25H2,1-2H3,(H,35,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 360 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50227631

(2-(6-chloro-1H-phenanthro[9,10-d]imidazol-2-yl)iso...)Show SMILES Clc1ccc2c3[nH]c(nc3c3ccccc3c2c1)-c1c(cccc1C#N)C#N Show InChI InChI=1S/C23H11ClN4/c24-15-8-9-18-19(10-15)16-6-1-2-7-17(16)21-22(18)28-23(27-21)20-13(11-25)4-3-5-14(20)12-26/h1-10H,(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337658

(1-(3-((4-chlorophenyl)ethynyl)benzyl)-3-isopropyl-...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccc(Cl)cc1)Cc1cccc(c1)C#Cc1ccccn1 Show InChI InChI=1S/C33H28ClN3O/c1-25(2)36-33(38)37(24-30-10-6-8-28(22-30)16-19-32-11-3-4-20-35-32)23-29-9-5-7-27(21-29)13-12-26-14-17-31(34)18-15-26/h3-11,14-15,17-18,20-22,25H,23-24H2,1-2H3,(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337656

(1-((5-(cyclopentylethynyl)pyridin-3-yl)methyl)-3-i...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cncc(c1)C#CC1CCCC1 Show InChI InChI=1S/C32H33N3O/c1-25(2)34-32(36)35(24-31-20-29(21-33-22-31)18-16-27-11-6-7-12-27)23-30-14-8-13-28(19-30)17-15-26-9-4-3-5-10-26/h3-5,8-10,13-14,19-22,25,27H,6-7,11-12,23-24H2,1-2H3,(H,34,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 490 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337655

(1-((5-(cyclopropylethynyl)pyridin-3-yl)methyl)-3-i...)Show SMILES CC(C)NC(=O)N(Cc1cccc(c1)C#Cc1ccccc1)Cc1cncc(c1)C#CC1CC1 Show InChI InChI=1S/C30H29N3O/c1-23(2)32-30(34)33(22-29-18-27(19-31-20-29)16-14-25-11-12-25)21-28-10-6-9-26(17-28)15-13-24-7-4-3-5-8-24/h3-10,17-20,23,25H,11-12,21-22H2,1-2H3,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 490 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337648

(3-isopropyl-1-(2-methoxybenzyl)-1-(3-(phenylethyny...)Show SMILES COc1ccccc1CN(Cc1cccc(c1)C#Cc1ccccc1)C(=O)NC(C)C Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(20-25-14-7-8-15-26(25)31-3)19-24-13-9-12-23(18-24)17-16-22-10-5-4-6-11-22/h4-15,18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 580 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337638
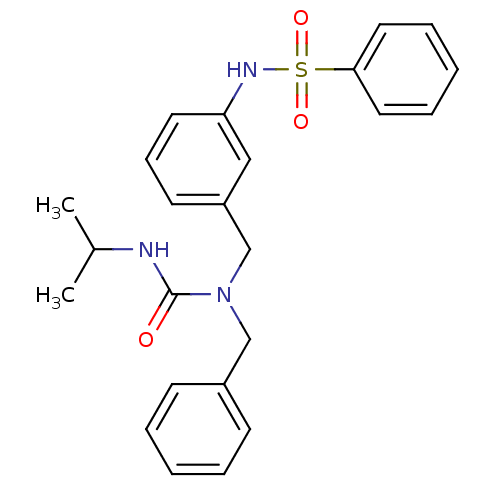
(CHEMBL1683194 | N-(3-((1-benzyl-3-isopropylureido)...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(NS(=O)(=O)c2ccccc2)c1 Show InChI InChI=1S/C24H27N3O3S/c1-19(2)25-24(28)27(17-20-10-5-3-6-11-20)18-21-12-9-13-22(16-21)26-31(29,30)23-14-7-4-8-15-23/h3-16,19,26H,17-18H2,1-2H3,(H,25,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 660 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337641

(1-benzyl-3-isopropyl-1-(3-(phenylethynyl)benzyl)ur...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C26H26N2O/c1-21(2)27-26(29)28(19-24-12-7-4-8-13-24)20-25-15-9-14-23(18-25)17-16-22-10-5-3-6-11-22/h3-15,18,21H,19-20H2,1-2H3,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 910 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337647

(1-benzyl-3-isopropyl-1-(3-((4-(trifluoromethyl)phe...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C27H25F3N2O/c1-20(2)31-26(33)32(18-23-7-4-3-5-8-23)19-24-10-6-9-22(17-24)12-11-21-13-15-25(16-14-21)27(28,29)30/h3-10,13-17,20H,18-19H2,1-2H3,(H,31,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 990 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337639
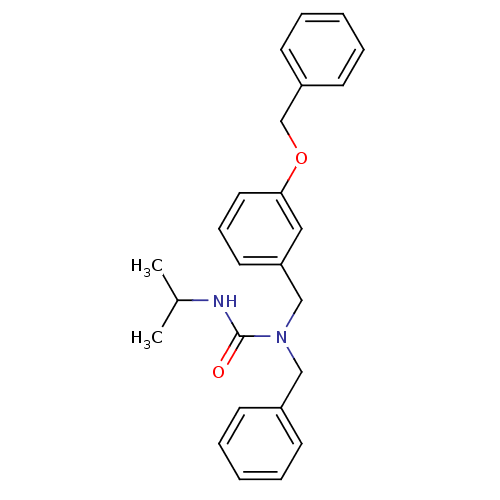
(1-benzyl-1-(3-(benzyloxy)benzyl)-3-isopropylurea |...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(OCc2ccccc2)c1 Show InChI InChI=1S/C25H28N2O2/c1-20(2)26-25(28)27(17-21-10-5-3-6-11-21)18-23-14-9-15-24(16-23)29-19-22-12-7-4-8-13-22/h3-16,20H,17-19H2,1-2H3,(H,26,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337642

(1-benzyl-3-isopropyl-1-(3-((3-methoxyphenyl)ethyny...)Show SMILES COc1cccc(c1)C#Cc1cccc(CN(Cc2ccccc2)C(=O)NC(C)C)c1 Show InChI InChI=1S/C27H28N2O2/c1-21(2)28-27(30)29(19-24-9-5-4-6-10-24)20-25-13-7-11-22(17-25)15-16-23-12-8-14-26(18-23)31-3/h4-14,17-18,21H,19-20H2,1-3H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50227631

(2-(6-chloro-1H-phenanthro[9,10-d]imidazol-2-yl)iso...)Show SMILES Clc1ccc2c3[nH]c(nc3c3ccccc3c2c1)-c1c(cccc1C#N)C#N Show InChI InChI=1S/C23H11ClN4/c24-15-8-9-18-19(10-15)16-6-1-2-7-17(16)21-22(18)28-23(27-21)20-13(11-25)4-3-5-14(20)12-26/h1-10H,(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in LPS-stimulated human whole blood assessed as inhibition of PGE2 production |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337664

(1-benzyl-3-(pentan-3-yl)-1-(3-(phenylethynyl)benzy...)Show SMILES CCC(CC)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 Show InChI InChI=1S/C28H30N2O/c1-3-27(4-2)29-28(31)30(21-25-14-9-6-10-15-25)22-26-17-11-16-24(20-26)19-18-23-12-7-5-8-13-23/h5-17,20,27H,3-4,21-22H2,1-2H3,(H,29,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337669

(1-benzyl-2-cyano-3-isopropyl-1-(3-(phenylethynyl)b...)Show SMILES CC(C)NC(=NC#N)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1ccccc1 |w:5.5| Show InChI InChI=1S/C27H26N4/c1-22(2)30-27(29-21-28)31(19-25-12-7-4-8-13-25)20-26-15-9-14-24(18-26)17-16-23-10-5-3-6-11-23/h3-15,18,22H,19-20H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337635
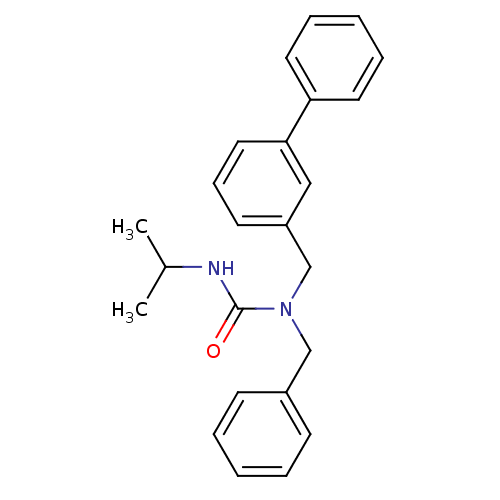
(1-benzyl-1-(biphenyl-3-ylmethyl)-3-isopropylurea |...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)-c1ccccc1 Show InChI InChI=1S/C24H26N2O/c1-19(2)25-24(27)26(17-20-10-5-3-6-11-20)18-21-12-9-15-23(16-21)22-13-7-4-8-14-22/h3-16,19H,17-18H2,1-2H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |
Prostaglandin E synthase
(Homo sapiens (Human)) | BDBM50337646

(1-benzyl-3-isopropyl-1-(3-((3-(trifluoromethyl)phe...)Show SMILES CC(C)NC(=O)N(Cc1ccccc1)Cc1cccc(c1)C#Cc1cccc(c1)C(F)(F)F Show InChI InChI=1S/C27H25F3N2O/c1-20(2)31-26(33)32(18-23-8-4-3-5-9-23)19-24-12-6-10-21(16-24)14-15-22-11-7-13-25(17-22)27(28,29)30/h3-13,16-17,20H,18-19H2,1-2H3,(H,31,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Center for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of mPGES1 in IL-1beta treated human A549 cell microsome assessed as inhibition of PGE2 production after 1 min in presence of 50% FBS |
Bioorg Med Chem Lett 21: 1488-92 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.006
BindingDB Entry DOI: 10.7270/Q2XS5VNP |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data