 Found 29 hits Enz. Inhib. hit(s) with all data for entry = 50044819
Found 29 hits Enz. Inhib. hit(s) with all data for entry = 50044819 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50031501
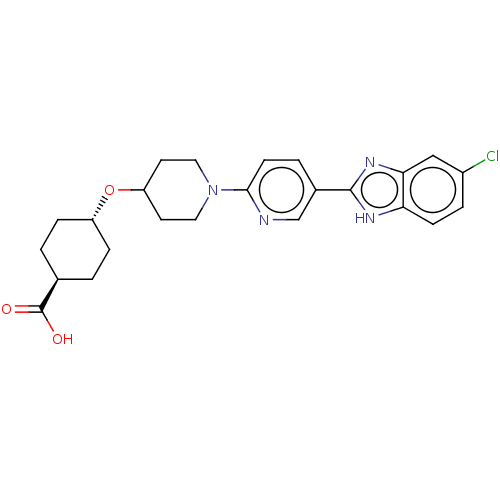
(CHEMBL3342773)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human A2A receptor |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50031501

(CHEMBL3342773)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled MK499 from human ERG |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50031503

(CHEMBL3342775)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,wD:3.2,(29.05,-9.85,;29.05,-8.31,;30.39,-7.55,;27.72,-7.54,;27.72,-6,;26.39,-5.22,;25.06,-5.98,;25.06,-7.53,;26.38,-8.3,;23.73,-5.21,;22.39,-5.98,;22.39,-7.52,;21.07,-8.29,;19.73,-7.52,;19.72,-5.98,;21.06,-5.21,;18.4,-8.29,;17.06,-7.53,;15.73,-8.3,;15.73,-9.84,;17.06,-10.61,;18.4,-9.84,;14.4,-10.61,;12.99,-9.98,;11.96,-11.13,;10.43,-11.13,;9.66,-12.46,;10.44,-13.79,;11.97,-13.78,;12.73,-12.46,;14.23,-12.14,;8.12,-12.46,;7.35,-11.13,;7.35,-13.79,;6.58,-12.45,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human A2A receptor |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50031502
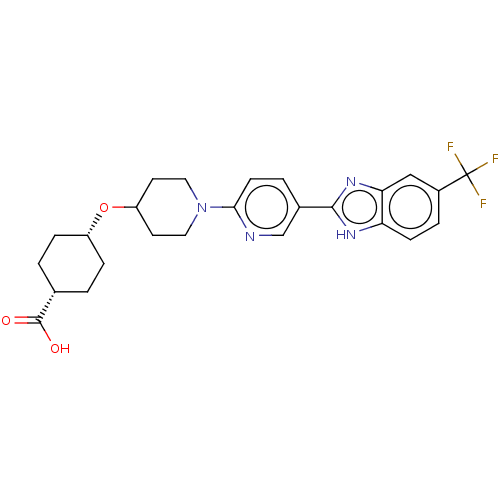
(CHEMBL3342774)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,3.2,(22.37,-7.8,;22.37,-6.26,;23.71,-5.5,;21.04,-5.48,;21.04,-3.94,;19.71,-3.16,;18.38,-3.93,;18.37,-5.48,;19.7,-6.25,;17.05,-3.16,;15.71,-3.93,;15.71,-5.47,;14.38,-6.24,;13.05,-5.47,;13.04,-3.93,;14.38,-3.16,;11.71,-6.24,;10.38,-5.47,;9.05,-6.25,;9.05,-7.79,;10.38,-8.56,;11.72,-7.79,;7.71,-8.55,;6.31,-7.93,;5.28,-9.07,;3.75,-9.07,;2.98,-10.4,;3.76,-11.73,;5.28,-11.73,;6.05,-10.4,;7.55,-10.08,;1.44,-10.4,;.67,-9.07,;.67,-11.74,;-.1,-10.4,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled MK499 from human ERG |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50031502

(CHEMBL3342774)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,3.2,(22.37,-7.8,;22.37,-6.26,;23.71,-5.5,;21.04,-5.48,;21.04,-3.94,;19.71,-3.16,;18.38,-3.93,;18.37,-5.48,;19.7,-6.25,;17.05,-3.16,;15.71,-3.93,;15.71,-5.47,;14.38,-6.24,;13.05,-5.47,;13.04,-3.93,;14.38,-3.16,;11.71,-6.24,;10.38,-5.47,;9.05,-6.25,;9.05,-7.79,;10.38,-8.56,;11.72,-7.79,;7.71,-8.55,;6.31,-7.93,;5.28,-9.07,;3.75,-9.07,;2.98,-10.4,;3.76,-11.73,;5.28,-11.73,;6.05,-10.4,;7.55,-10.08,;1.44,-10.4,;.67,-9.07,;.67,-11.74,;-.1,-10.4,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human A2A receptor |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50031505
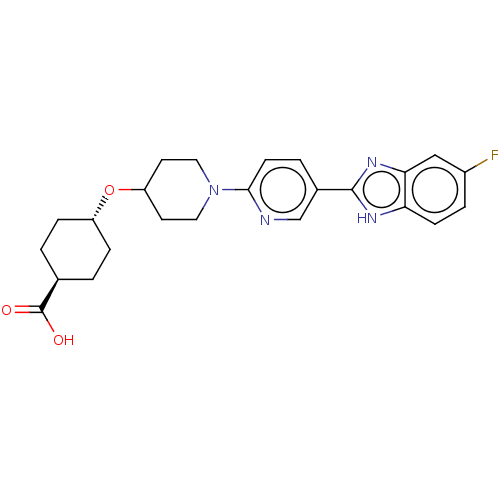
(CHEMBL3342771)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human A2A receptor |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50031503

(CHEMBL3342775)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,wD:3.2,(29.05,-9.85,;29.05,-8.31,;30.39,-7.55,;27.72,-7.54,;27.72,-6,;26.39,-5.22,;25.06,-5.98,;25.06,-7.53,;26.38,-8.3,;23.73,-5.21,;22.39,-5.98,;22.39,-7.52,;21.07,-8.29,;19.73,-7.52,;19.72,-5.98,;21.06,-5.21,;18.4,-8.29,;17.06,-7.53,;15.73,-8.3,;15.73,-9.84,;17.06,-10.61,;18.4,-9.84,;14.4,-10.61,;12.99,-9.98,;11.96,-11.13,;10.43,-11.13,;9.66,-12.46,;10.44,-13.79,;11.97,-13.78,;12.73,-12.46,;14.23,-12.14,;8.12,-12.46,;7.35,-11.13,;7.35,-13.79,;6.58,-12.45,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled MK499 from human ERG |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Homo sapiens (Human)) | BDBM50031504

(CHEMBL3342770)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human A2A receptor |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50031504

(CHEMBL3342770)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled MK499 from human ERG |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50031506
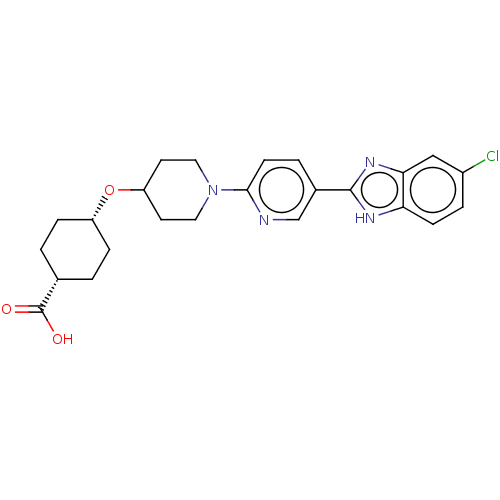
(CHEMBL3342772)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled MK499 from human ERG |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50031505

(CHEMBL3342771)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of radiolabeled MK499 from human ERG |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031506

(CHEMBL3342772)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031502

(CHEMBL3342774)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,3.2,(22.37,-7.8,;22.37,-6.26,;23.71,-5.5,;21.04,-5.48,;21.04,-3.94,;19.71,-3.16,;18.38,-3.93,;18.37,-5.48,;19.7,-6.25,;17.05,-3.16,;15.71,-3.93,;15.71,-5.47,;14.38,-6.24,;13.05,-5.47,;13.04,-3.93,;14.38,-3.16,;11.71,-6.24,;10.38,-5.47,;9.05,-6.25,;9.05,-7.79,;10.38,-8.56,;11.72,-7.79,;7.71,-8.55,;6.31,-7.93,;5.28,-9.07,;3.75,-9.07,;2.98,-10.4,;3.76,-11.73,;5.28,-11.73,;6.05,-10.4,;7.55,-10.08,;1.44,-10.4,;.67,-9.07,;.67,-11.74,;-.1,-10.4,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031501

(CHEMBL3342773)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031503

(CHEMBL3342775)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,wD:3.2,(29.05,-9.85,;29.05,-8.31,;30.39,-7.55,;27.72,-7.54,;27.72,-6,;26.39,-5.22,;25.06,-5.98,;25.06,-7.53,;26.38,-8.3,;23.73,-5.21,;22.39,-5.98,;22.39,-7.52,;21.07,-8.29,;19.73,-7.52,;19.72,-5.98,;21.06,-5.21,;18.4,-8.29,;17.06,-7.53,;15.73,-8.3,;15.73,-9.84,;17.06,-10.61,;18.4,-9.84,;14.4,-10.61,;12.99,-9.98,;11.96,-11.13,;10.43,-11.13,;9.66,-12.46,;10.44,-13.79,;11.97,-13.78,;12.73,-12.46,;14.23,-12.14,;8.12,-12.46,;7.35,-11.13,;7.35,-13.79,;6.58,-12.45,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031504

(CHEMBL3342770)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50031502

(CHEMBL3342774)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,3.2,(22.37,-7.8,;22.37,-6.26,;23.71,-5.5,;21.04,-5.48,;21.04,-3.94,;19.71,-3.16,;18.38,-3.93,;18.37,-5.48,;19.7,-6.25,;17.05,-3.16,;15.71,-3.93,;15.71,-5.47,;14.38,-6.24,;13.05,-5.47,;13.04,-3.93,;14.38,-3.16,;11.71,-6.24,;10.38,-5.47,;9.05,-6.25,;9.05,-7.79,;10.38,-8.56,;11.72,-7.79,;7.71,-8.55,;6.31,-7.93,;5.28,-9.07,;3.75,-9.07,;2.98,-10.4,;3.76,-11.73,;5.28,-11.73,;6.05,-10.4,;7.55,-10.08,;1.44,-10.4,;.67,-9.07,;.67,-11.74,;-.1,-10.4,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50031506

(CHEMBL3342772)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031505

(CHEMBL3342771)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50031503

(CHEMBL3342775)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,wD:3.2,(29.05,-9.85,;29.05,-8.31,;30.39,-7.55,;27.72,-7.54,;27.72,-6,;26.39,-5.22,;25.06,-5.98,;25.06,-7.53,;26.38,-8.3,;23.73,-5.21,;22.39,-5.98,;22.39,-7.52,;21.07,-8.29,;19.73,-7.52,;19.72,-5.98,;21.06,-5.21,;18.4,-8.29,;17.06,-7.53,;15.73,-8.3,;15.73,-9.84,;17.06,-10.61,;18.4,-9.84,;14.4,-10.61,;12.99,-9.98,;11.96,-11.13,;10.43,-11.13,;9.66,-12.46,;10.44,-13.79,;11.97,-13.78,;12.73,-12.46,;14.23,-12.14,;8.12,-12.46,;7.35,-11.13,;7.35,-13.79,;6.58,-12.45,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50031504

(CHEMBL3342770)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50031501

(CHEMBL3342773)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50031505

(CHEMBL3342771)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Sterol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031506

(CHEMBL3342772)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ACAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Sterol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031505

(CHEMBL3342771)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ACAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Sterol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031503

(CHEMBL3342775)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,wD:3.2,(29.05,-9.85,;29.05,-8.31,;30.39,-7.55,;27.72,-7.54,;27.72,-6,;26.39,-5.22,;25.06,-5.98,;25.06,-7.53,;26.38,-8.3,;23.73,-5.21,;22.39,-5.98,;22.39,-7.52,;21.07,-8.29,;19.73,-7.52,;19.72,-5.98,;21.06,-5.21,;18.4,-8.29,;17.06,-7.53,;15.73,-8.3,;15.73,-9.84,;17.06,-10.61,;18.4,-9.84,;14.4,-10.61,;12.99,-9.98,;11.96,-11.13,;10.43,-11.13,;9.66,-12.46,;10.44,-13.79,;11.97,-13.78,;12.73,-12.46,;14.23,-12.14,;8.12,-12.46,;7.35,-11.13,;7.35,-13.79,;6.58,-12.45,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18- | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ACAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Sterol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031502

(CHEMBL3342774)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(ccc2[nH]1)C(F)(F)F |r,wU:6.9,3.2,(22.37,-7.8,;22.37,-6.26,;23.71,-5.5,;21.04,-5.48,;21.04,-3.94,;19.71,-3.16,;18.38,-3.93,;18.37,-5.48,;19.7,-6.25,;17.05,-3.16,;15.71,-3.93,;15.71,-5.47,;14.38,-6.24,;13.05,-5.47,;13.04,-3.93,;14.38,-3.16,;11.71,-6.24,;10.38,-5.47,;9.05,-6.25,;9.05,-7.79,;10.38,-8.56,;11.72,-7.79,;7.71,-8.55,;6.31,-7.93,;5.28,-9.07,;3.75,-9.07,;2.98,-10.4,;3.76,-11.73,;5.28,-11.73,;6.05,-10.4,;7.55,-10.08,;1.44,-10.4,;.67,-9.07,;.67,-11.74,;-.1,-10.4,)| Show InChI InChI=1S/C25H27F3N4O3/c26-25(27,28)17-4-7-20-21(13-17)31-23(30-20)16-3-8-22(29-14-16)32-11-9-19(10-12-32)35-18-5-1-15(2-6-18)24(33)34/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,30,31)(H,33,34)/t15-,18+ | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ACAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Sterol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031501

(CHEMBL3342773)Show SMILES OC(=O)[C@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(Cl)ccc2[nH]1 |r,wU:6.9,wD:3.2,(37.68,-12.65,;37.68,-11.11,;39.02,-10.34,;36.35,-10.33,;36.35,-8.79,;35.02,-8.01,;33.69,-8.77,;33.68,-10.32,;35.01,-11.09,;32.35,-8,;31.02,-8.77,;31.02,-10.31,;29.69,-11.08,;28.36,-10.31,;28.35,-8.78,;29.68,-8,;27.02,-11.08,;25.69,-10.32,;24.36,-11.09,;24.36,-12.64,;25.69,-13.41,;27.03,-12.63,;23.02,-13.4,;21.62,-12.77,;20.59,-13.92,;19.05,-13.92,;18.29,-15.25,;16.75,-15.25,;19.06,-16.58,;20.59,-16.57,;21.35,-15.25,;22.86,-14.93,)| Show InChI InChI=1S/C24H27ClN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18- | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ACAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |
Sterol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50031504

(CHEMBL3342770)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)OC1CCN(CC1)c1ccc(cn1)-c1nc2cc(F)ccc2[nH]1 |r,wU:6.9,3.2,(22.66,-7.8,;22.66,-6.26,;24,-5.5,;21.33,-5.48,;21.33,-3.94,;20,-3.16,;18.67,-3.93,;18.66,-5.48,;19.99,-6.25,;17.33,-3.16,;16,-3.93,;16,-5.47,;14.67,-6.24,;13.34,-5.47,;13.33,-3.93,;14.66,-3.16,;12,-6.24,;10.67,-5.47,;9.34,-6.25,;9.34,-7.79,;10.67,-8.56,;12.01,-7.79,;8,-8.55,;6.6,-7.93,;5.57,-9.07,;4.04,-9.07,;3.27,-10.4,;1.73,-10.4,;4.04,-11.73,;5.57,-11.73,;6.33,-10.4,;7.84,-10.08,)| Show InChI InChI=1S/C24H27FN4O3/c25-17-4-7-20-21(13-17)28-23(27-20)16-3-8-22(26-14-16)29-11-9-19(10-12-29)32-18-5-1-15(2-6-18)24(30)31/h3-4,7-8,13-15,18-19H,1-2,5-6,9-12H2,(H,27,28)(H,30,31)/t15-,18+ | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ACAT1 |
ACS Med Chem Lett 5: 1082-7 (2014)
Article DOI: 10.1021/ml5003426
BindingDB Entry DOI: 10.7270/Q2WQ05CX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data