 Found 65 hits Enz. Inhib. hit(s) with all data for entry = 50017997
Found 65 hits Enz. Inhib. hit(s) with all data for entry = 50017997 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM15234
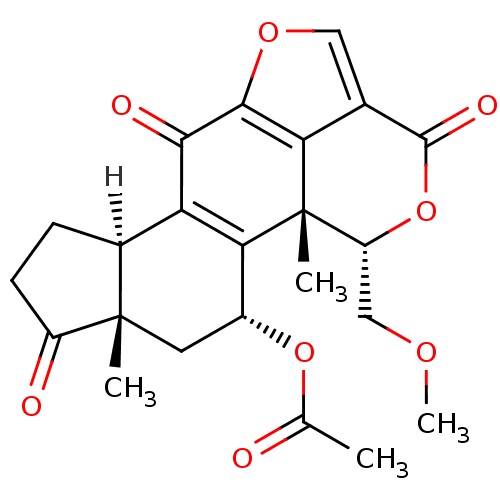
((1R,3R,5S,9R,18S)-18-(methoxymethyl)-1,5-dimethyl-...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)C[C@@H](OC(C)=O)C1=C2C(=O)c2occ3c2[C@]1(C)[C@@H](COC)OC3=O |r,c:15| Show InChI InChI=1S/C23H24O8/c1-10(24)30-13-7-22(2)12(5-6-14(22)25)16-18(13)23(3)15(9-28-4)31-21(27)11-8-29-20(17(11)23)19(16)26/h8,12-13,15H,5-7,9H2,1-4H3/t12-,13+,15+,22-,23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM15234

((1R,3R,5S,9R,18S)-18-(methoxymethyl)-1,5-dimethyl-...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)C[C@@H](OC(C)=O)C1=C2C(=O)c2occ3c2[C@]1(C)[C@@H](COC)OC3=O |r,c:15| Show InChI InChI=1S/C23H24O8/c1-10(24)30-13-7-22(2)12(5-6-14(22)25)16-18(13)23(3)15(9-28-4)31-21(27)11-8-29-20(17(11)23)19(16)26/h8,12-13,15H,5-7,9H2,1-4H3/t12-,13+,15+,22-,23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM15234

((1R,3R,5S,9R,18S)-18-(methoxymethyl)-1,5-dimethyl-...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)C[C@@H](OC(C)=O)C1=C2C(=O)c2occ3c2[C@]1(C)[C@@H](COC)OC3=O |r,c:15| Show InChI InChI=1S/C23H24O8/c1-10(24)30-13-7-22(2)12(5-6-14(22)25)16-18(13)23(3)15(9-28-4)31-21(27)11-8-29-20(17(11)23)19(16)26/h8,12-13,15H,5-7,9H2,1-4H3/t12-,13+,15+,22-,23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM15234

((1R,3R,5S,9R,18S)-18-(methoxymethyl)-1,5-dimethyl-...)Show SMILES [H][C@@]12CCC(=O)[C@@]1(C)C[C@@H](OC(C)=O)C1=C2C(=O)c2occ3c2[C@]1(C)[C@@H](COC)OC3=O |r,c:15| Show InChI InChI=1S/C23H24O8/c1-10(24)30-13-7-22(2)12(5-6-14(22)25)16-18(13)23(3)15(9-28-4)31-21(27)11-8-29-20(17(11)23)19(16)26/h8,12-13,15H,5-7,9H2,1-4H3/t12-,13+,15+,22-,23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kbeta |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189741
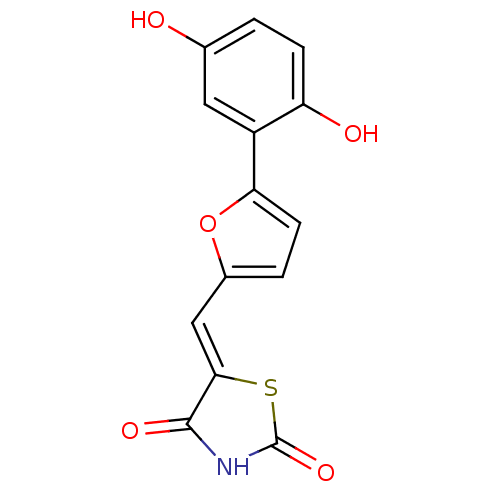
(5-[5-(2,5-dihydroxyphenyl)furan-2-ylmethylene]thia...)Show InChI InChI=1S/C14H9NO5S/c16-7-1-3-10(17)9(5-7)11-4-2-8(20-11)6-12-13(18)15-14(19)21-12/h1-6,16-17H,(H,15,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189760
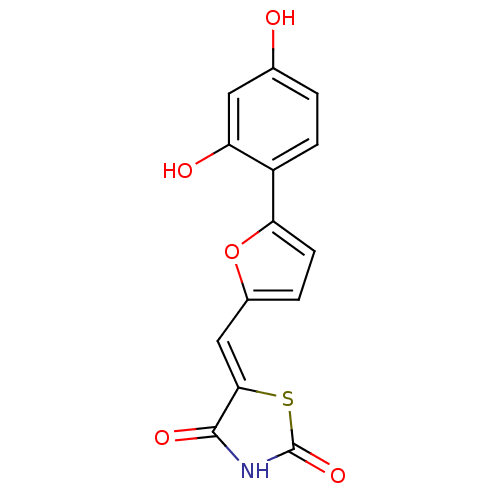
(5-[5-(2,4-dihydroxyphenyl)furan-2-ylmethylene]thia...)Show InChI InChI=1S/C14H9NO5S/c16-7-1-3-9(10(17)5-7)11-4-2-8(20-11)6-12-13(18)15-14(19)21-12/h1-6,16-17H,(H,15,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189748
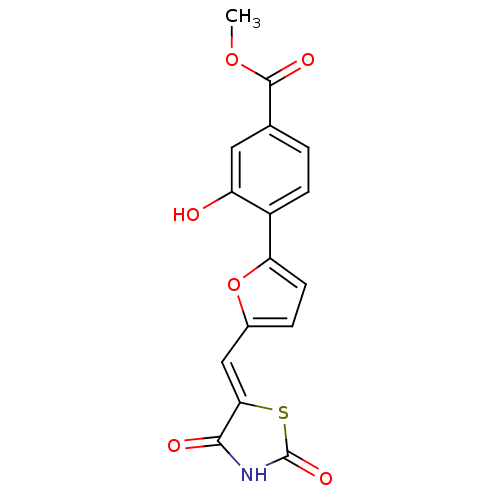
(4-[5-(2,4-dioxothiazolidin-5-ylidenemethyl)furan-2...)Show SMILES COC(=O)c1ccc(-c2ccc(\C=C3/SC(=O)NC3=O)o2)c(O)c1 Show InChI InChI=1S/C16H11NO6S/c1-22-15(20)8-2-4-10(11(18)6-8)12-5-3-9(23-12)7-13-14(19)17-16(21)24-13/h2-7,18H,1H3,(H,17,19,21)/b13-7- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189752
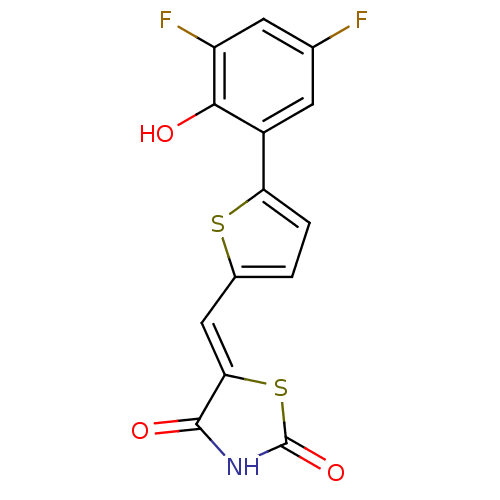
(5-[5-(3,5-difluoro-2-hydroxyphenyl)furan-2-ylmethy...)Show InChI InChI=1S/C14H7F2NO3S2/c15-6-3-8(12(18)9(16)4-6)10-2-1-7(21-10)5-11-13(19)17-14(20)22-11/h1-5,18H,(H,17,19,20)/b11-5- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189744
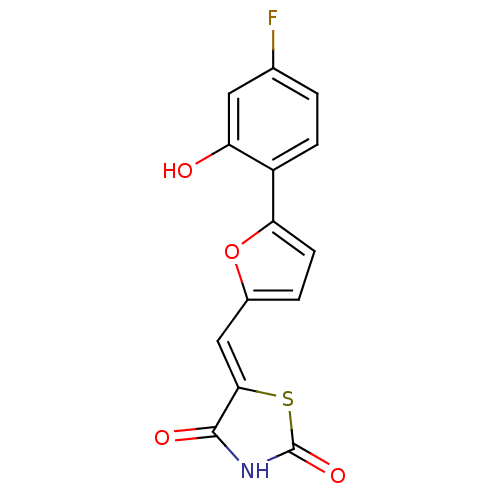
((Z)-5-((5-(4-fluoro-2-hydroxyphenyl)furan-2-yl)met...)Show InChI InChI=1S/C14H8FNO4S/c15-7-1-3-9(10(17)5-7)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189750
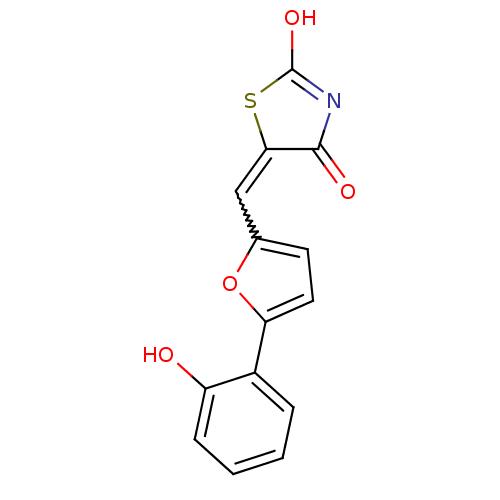
(5-[5-(2-hydroxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccccc1O |w:7.8,t:1| Show InChI InChI=1S/C14H9NO4S/c16-10-4-2-1-3-9(10)11-6-5-8(19-11)7-12-13(17)15-14(18)20-12/h1-7,16H,(H,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189743
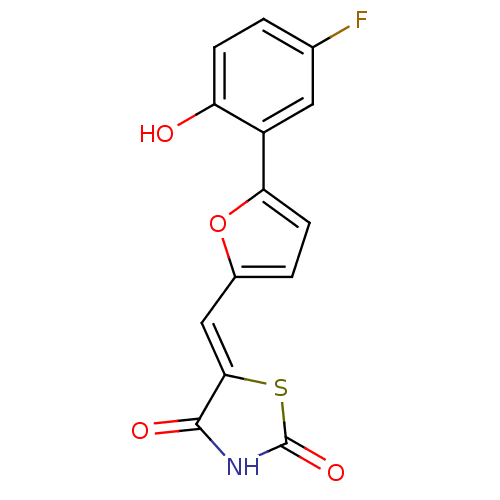
(5-[5-(5-fluoro-2-hydroxyphenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C14H8FNO4S/c15-7-1-3-10(17)9(5-7)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189745
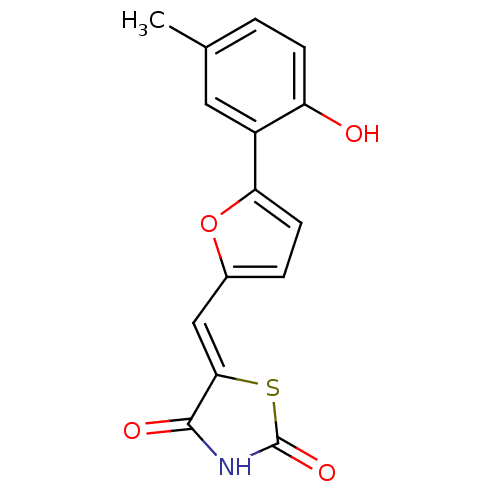
(5-[5-(2-hydroxy-5-methylphenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C15H11NO4S/c1-8-2-4-11(17)10(6-8)12-5-3-9(20-12)7-13-14(18)16-15(19)21-13/h2-7,17H,1H3,(H,16,18,19)/b13-7- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189751
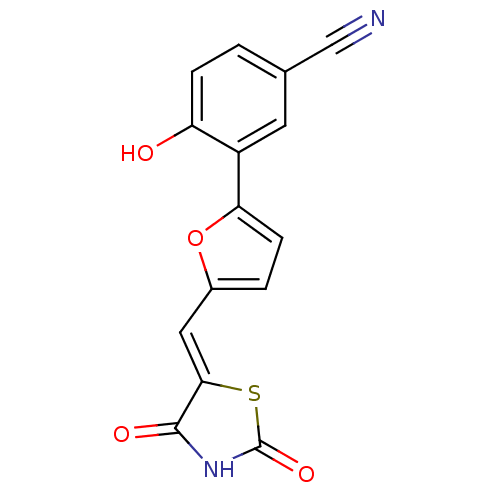
(3-[5-(2,4-dioxothiazolidin-5-ylidenemethyl)furan-2...)Show InChI InChI=1S/C15H8N2O4S/c16-7-8-1-3-11(18)10(5-8)12-4-2-9(21-12)6-13-14(19)17-15(20)22-13/h1-6,18H,(H,17,19,20)/b13-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189753
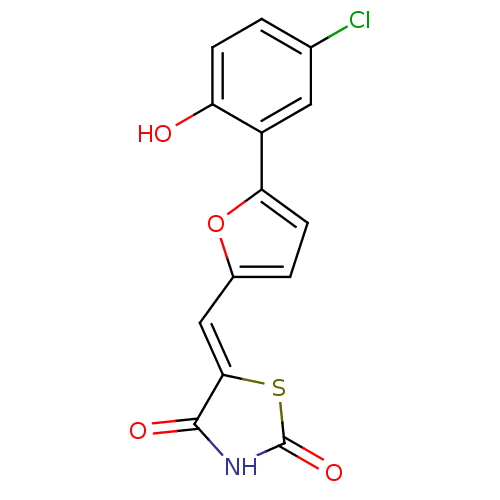
(5-[5-(2-hydroxy-5-chlorophenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C14H8ClNO4S/c15-7-1-3-10(17)9(5-7)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189742
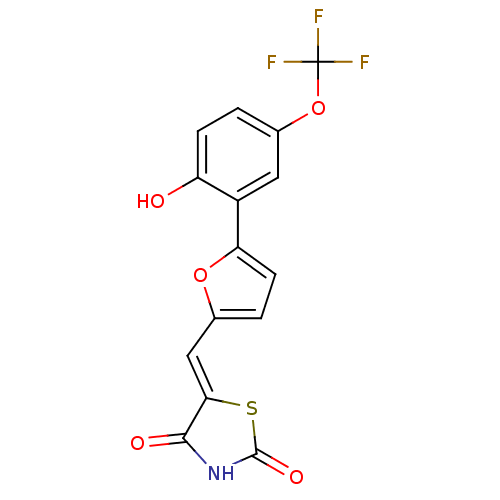
(5-[5-(2-hydroxy-5-trifluoromethoxyphenyl)furan-2-y...)Show SMILES Oc1ccc(OC(F)(F)F)cc1-c1ccc(\C=C2/SC(=O)NC2=O)o1 Show InChI InChI=1S/C15H8F3NO5S/c16-15(17,18)24-8-1-3-10(20)9(5-8)11-4-2-7(23-11)6-12-13(21)19-14(22)25-12/h1-6,20H,(H,19,21,22)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189764
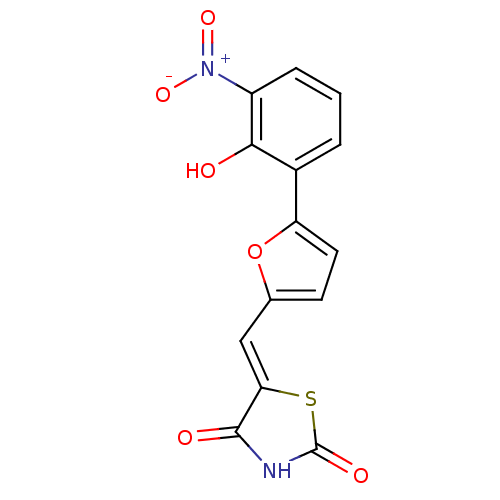
(5-[5-(2-hydroxy-3-nitrophenyl)furan-2-ylmethylene]...)Show SMILES Oc1c(cccc1[N+]([O-])=O)-c1ccc(\C=C2/SC(=O)NC2=O)o1 Show InChI InChI=1S/C14H8N2O6S/c17-12-8(2-1-3-9(12)16(20)21)10-5-4-7(22-10)6-11-13(18)15-14(19)23-11/h1-6,17H,(H,15,18,19)/b11-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189741

(5-[5-(2,5-dihydroxyphenyl)furan-2-ylmethylene]thia...)Show InChI InChI=1S/C14H9NO5S/c16-7-1-3-10(17)9(5-7)11-4-2-8(20-11)6-12-13(18)15-14(19)21-12/h1-6,16-17H,(H,15,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189746
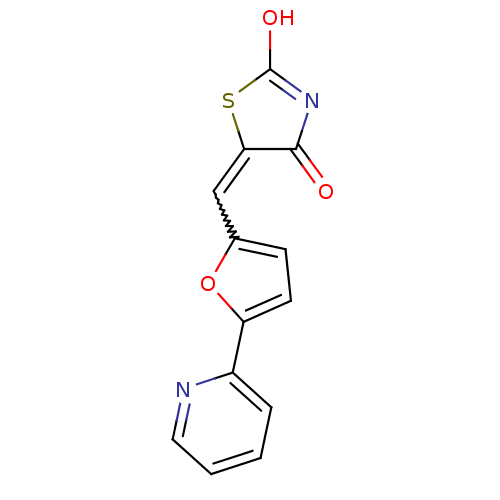
(5-(5-pyridin-2-ylfuran-2-ylmethylene)thiazolidine-...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccccn1 |w:7.8,t:1| Show InChI InChI=1S/C13H8N2O3S/c16-12-11(19-13(17)15-12)7-8-4-5-10(18-8)9-3-1-2-6-14-9/h1-7H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189746

(5-(5-pyridin-2-ylfuran-2-ylmethylene)thiazolidine-...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccccn1 |w:7.8,t:1| Show InChI InChI=1S/C13H8N2O3S/c16-12-11(19-13(17)15-12)7-8-4-5-10(18-8)9-3-1-2-6-14-9/h1-7H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 185 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189760

(5-[5-(2,4-dihydroxyphenyl)furan-2-ylmethylene]thia...)Show InChI InChI=1S/C14H9NO5S/c16-7-1-3-9(10(17)5-7)11-4-2-8(20-11)6-12-13(18)15-14(19)21-12/h1-6,16-17H,(H,15,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189766
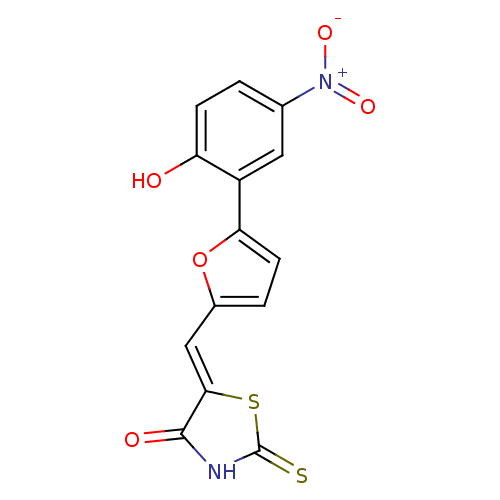
(5-((5-(2-hydroxy-5-nitrophenyl)furan-2-yl)methylen...)Show SMILES Oc1ccc(cc1-c1ccc(\C=C2/SC(=S)NC2=O)o1)[N+]([O-])=O Show InChI InChI=1S/C14H8N2O5S2/c17-10-3-1-7(16(19)20)5-9(10)11-4-2-8(21-11)6-12-13(18)15-14(22)23-12/h1-6,17H,(H,15,18,22)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189748

(4-[5-(2,4-dioxothiazolidin-5-ylidenemethyl)furan-2...)Show SMILES COC(=O)c1ccc(-c2ccc(\C=C3/SC(=O)NC3=O)o2)c(O)c1 Show InChI InChI=1S/C16H11NO6S/c1-22-15(20)8-2-4-10(11(18)6-8)12-5-3-9(23-12)7-13-14(19)17-16(21)24-13/h2-7,18H,1H3,(H,17,19,21)/b13-7- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189751

(3-[5-(2,4-dioxothiazolidin-5-ylidenemethyl)furan-2...)Show InChI InChI=1S/C15H8N2O4S/c16-7-8-1-3-11(18)10(5-8)12-4-2-9(21-12)6-13-14(19)17-15(20)22-13/h1-6,18H,(H,17,19,20)/b13-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189756
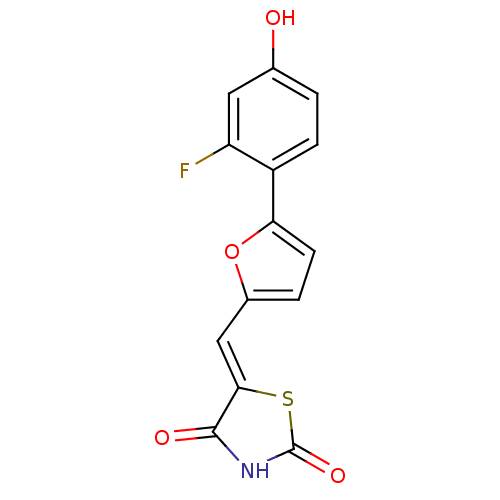
(5-[5-(2-fluoro-4-hydroxyphenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C14H8FNO4S/c15-10-5-7(17)1-3-9(10)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189755
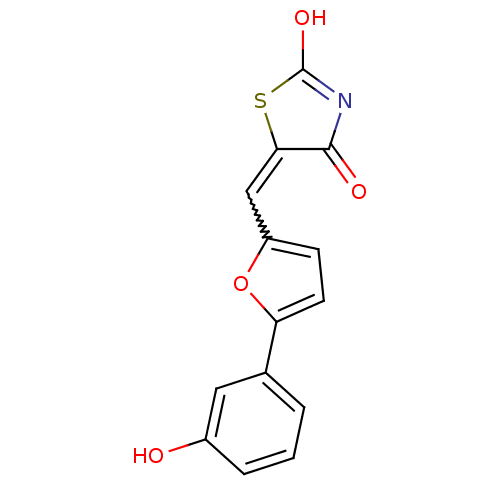
(5-[5-(3-hydroxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1cccc(O)c1 |w:7.8,t:1| Show InChI InChI=1S/C14H9NO4S/c16-9-3-1-2-8(6-9)11-5-4-10(19-11)7-12-13(17)15-14(18)20-12/h1-7,16H,(H,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM12915
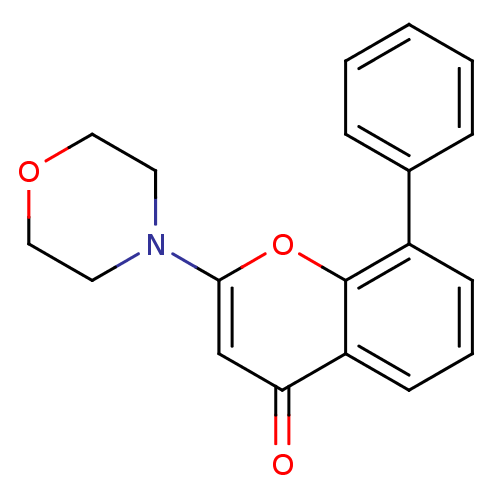
(2-(4-Morpholinyl)-8-phenyl-4H-1-benzopyran-4-one |...)Show InChI InChI=1S/C19H17NO3/c21-17-13-18(20-9-11-22-12-10-20)23-19-15(7-4-8-16(17)19)14-5-2-1-3-6-14/h1-8,13H,9-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 306 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kbeta |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189750

(5-[5-(2-hydroxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccccc1O |w:7.8,t:1| Show InChI InChI=1S/C14H9NO4S/c16-10-4-2-1-3-9(10)11-6-5-8(19-11)7-12-13(17)15-14(18)20-12/h1-7,16H,(H,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189752

(5-[5-(3,5-difluoro-2-hydroxyphenyl)furan-2-ylmethy...)Show InChI InChI=1S/C14H7F2NO3S2/c15-6-3-8(12(18)9(16)4-6)10-2-1-7(21-10)5-11-13(19)17-14(20)22-11/h1-5,18H,(H,17,19,20)/b11-5- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189747
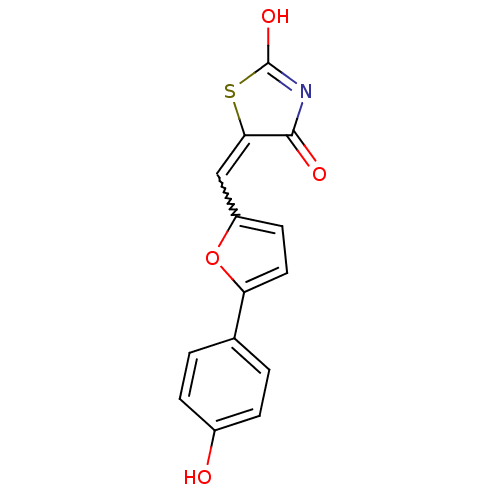
(5-[5-(4-hydroxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccc(O)cc1 |w:7.8,t:1| Show InChI InChI=1S/C14H9NO4S/c16-9-3-1-8(2-4-9)11-6-5-10(19-11)7-12-13(17)15-14(18)20-12/h1-7,16H,(H,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 380 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189743

(5-[5-(5-fluoro-2-hydroxyphenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C14H8FNO4S/c15-7-1-3-10(17)9(5-7)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 455 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189745

(5-[5-(2-hydroxy-5-methylphenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C15H11NO4S/c1-8-2-4-11(17)10(6-8)12-5-3-9(20-12)7-13-14(18)16-15(19)21-13/h2-7,17H,1H3,(H,16,18,19)/b13-7- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189758
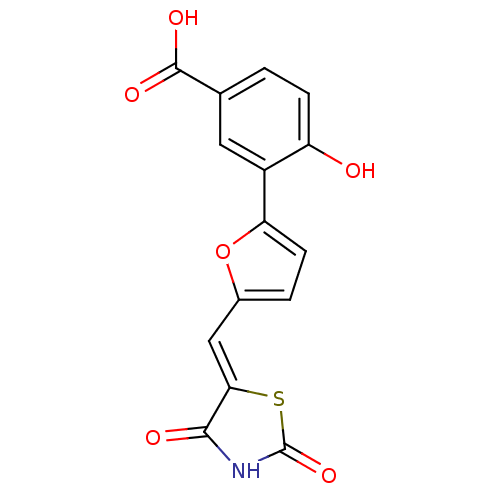
(3-[5-(2,4-dioxothiazolidin-5-ylidenemethyl)furan-2...)Show SMILES OC(=O)c1ccc(O)c(c1)-c1ccc(\C=C2/SC(=O)NC2=O)o1 Show InChI InChI=1S/C15H9NO6S/c17-10-3-1-7(14(19)20)5-9(10)11-4-2-8(22-11)6-12-13(18)16-15(21)23-12/h1-6,17H,(H,19,20)(H,16,18,21)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 530 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189753

(5-[5-(2-hydroxy-5-chlorophenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C14H8ClNO4S/c15-7-1-3-10(17)9(5-7)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 545 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189742

(5-[5-(2-hydroxy-5-trifluoromethoxyphenyl)furan-2-y...)Show SMILES Oc1ccc(OC(F)(F)F)cc1-c1ccc(\C=C2/SC(=O)NC2=O)o1 Show InChI InChI=1S/C15H8F3NO5S/c16-15(17,18)24-8-1-3-10(20)9(5-8)11-4-2-7(23-11)6-12-13(21)19-14(22)25-12/h1-6,20H,(H,19,21,22)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189766

(5-((5-(2-hydroxy-5-nitrophenyl)furan-2-yl)methylen...)Show SMILES Oc1ccc(cc1-c1ccc(\C=C2/SC(=S)NC2=O)o1)[N+]([O-])=O Show InChI InChI=1S/C14H8N2O5S2/c17-10-3-1-7(16(19)20)5-9(10)11-4-2-8(21-11)6-12-13(18)15-14(22)23-12/h1-6,17H,(H,15,18,22)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189765
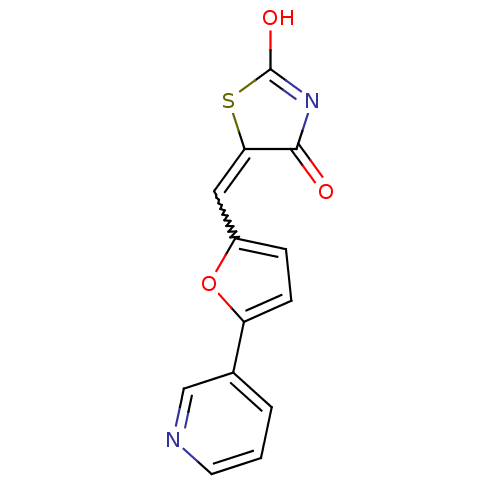
(5-(5-pyridin-3-ylfuran-2-ylmethylene)thiazolidine-...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1cccnc1 |w:7.8,t:1| Show InChI InChI=1S/C13H8N2O3S/c16-12-11(19-13(17)15-12)6-9-3-4-10(18-9)8-2-1-5-14-7-8/h1-7H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 630 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM12915

(2-(4-Morpholinyl)-8-phenyl-4H-1-benzopyran-4-one |...)Show InChI InChI=1S/C19H17NO3/c21-17-13-18(20-9-11-22-12-10-20)23-19-15(7-4-8-16(17)19)14-5-2-1-3-6-14/h1-8,13H,9-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 720 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189768
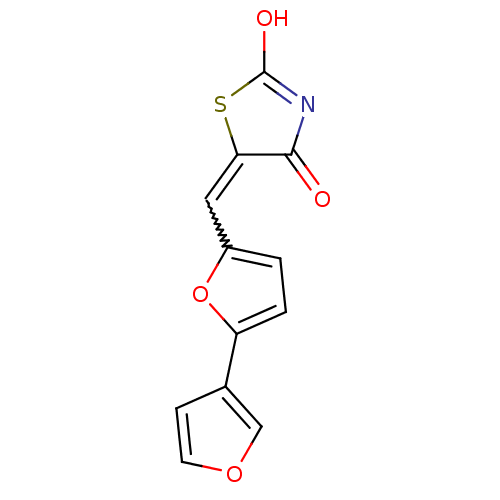
(5-[2,3']bifuranyl-5-ylmethylenethiazolidine-2,4-di...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccoc1 |w:7.8,t:1| Show InChI InChI=1S/C12H7NO4S/c14-11-10(18-12(15)13-11)5-8-1-2-9(17-8)7-3-4-16-6-7/h1-6H,(H,13,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 840 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
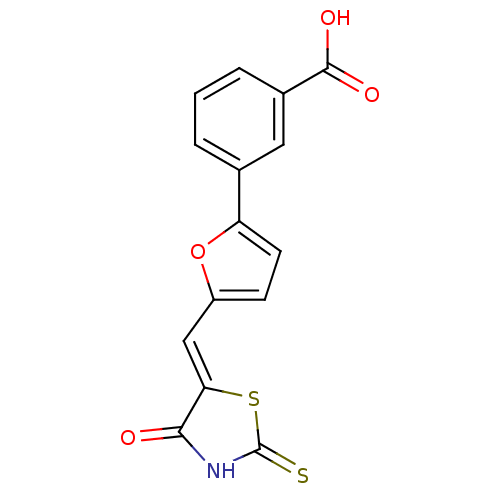
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189763
(3-(5-((4-oxo-2-thioxothiazolidin-5-ylidene)methyl)...)Show SMILES OC(=O)c1cccc(c1)-c1ccc(\C=C2/SC(=S)NC2=O)o1 Show InChI InChI=1S/C15H9NO4S2/c17-13-12(22-15(21)16-13)7-10-4-5-11(20-10)8-2-1-3-9(6-8)14(18)19/h1-7H,(H,18,19)(H,16,17,21)/b12-7- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 920 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189744

((Z)-5-((5-(4-fluoro-2-hydroxyphenyl)furan-2-yl)met...)Show InChI InChI=1S/C14H8FNO4S/c15-7-1-3-9(10(17)5-7)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 940 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189765

(5-(5-pyridin-3-ylfuran-2-ylmethylene)thiazolidine-...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1cccnc1 |w:7.8,t:1| Show InChI InChI=1S/C13H8N2O3S/c16-12-11(19-13(17)15-12)6-9-3-4-10(18-9)8-2-1-5-14-7-8/h1-7H,(H,15,16,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 950 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189756

(5-[5-(2-fluoro-4-hydroxyphenyl)furan-2-ylmethylene...)Show InChI InChI=1S/C14H8FNO4S/c15-10-5-7(17)1-3-9(10)11-4-2-8(20-11)6-12-13(18)16-14(19)21-12/h1-6,17H,(H,16,18,19)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189757

(5-[5-(2-fluorophenyl)furan-2-ylmethylene]thiazolid...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccccc1F |w:7.8,t:1| Show InChI InChI=1S/C14H8FNO3S/c15-10-4-2-1-3-9(10)11-6-5-8(19-11)7-12-13(17)16-14(18)20-12/h1-7H,(H,16,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189764

(5-[5-(2-hydroxy-3-nitrophenyl)furan-2-ylmethylene]...)Show SMILES Oc1c(cccc1[N+]([O-])=O)-c1ccc(\C=C2/SC(=O)NC2=O)o1 Show InChI InChI=1S/C14H8N2O6S/c17-12-8(2-1-3-9(12)16(20)21)10-5-4-7(22-10)6-11-13(18)15-14(19)23-11/h1-6,17H,(H,15,18,19)/b11-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM12915

(2-(4-Morpholinyl)-8-phenyl-4H-1-benzopyran-4-one |...)Show InChI InChI=1S/C19H17NO3/c21-17-13-18(20-9-11-22-12-10-20)23-19-15(7-4-8-16(17)19)14-5-2-1-3-6-14/h1-8,13H,9-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kdelta |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189758

(3-[5-(2,4-dioxothiazolidin-5-ylidenemethyl)furan-2...)Show SMILES OC(=O)c1ccc(O)c(c1)-c1ccc(\C=C2/SC(=O)NC2=O)o1 Show InChI InChI=1S/C15H9NO6S/c17-10-3-1-7(14(19)20)5-9(10)11-4-2-8(22-11)6-12-13(18)16-15(21)23-12/h1-6,17H,(H,19,20)(H,16,18,21)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.35E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189755

(5-[5-(3-hydroxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1cccc(O)c1 |w:7.8,t:1| Show InChI InChI=1S/C14H9NO4S/c16-9-3-1-2-8(6-9)11-5-4-10(19-11)7-12-13(17)15-14(18)20-12/h1-7,16H,(H,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189761

(5-[5-(3-methoxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES COc1cccc(c1)-c1ccc(C=C2SC(O)=NC2=O)o1 |w:12.12,c:17| Show InChI InChI=1S/C15H11NO4S/c1-19-10-4-2-3-9(7-10)12-6-5-11(20-12)8-13-14(17)16-15(18)21-13/h2-8H,1H3,(H,16,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
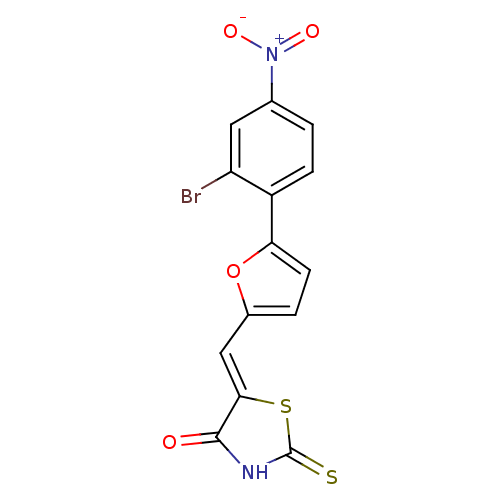
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50189759
(5-((5-(2-bromo-4-nitrophenyl)furan-2-yl)methylene)...)Show SMILES [O-][N+](=O)c1ccc(-c2ccc(\C=C3/SC(=S)NC3=O)o2)c(Br)c1 Show InChI InChI=1S/C14H7BrN2O4S2/c15-10-5-7(17(19)20)1-3-9(10)11-4-2-8(21-11)6-12-13(18)16-14(22)23-12/h1-6H,(H,16,18,22)/b12-6- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50189747

(5-[5-(4-hydroxyphenyl)furan-2-ylmethylene]thiazoli...)Show SMILES OC1=NC(=O)C(S1)=Cc1ccc(o1)-c1ccc(O)cc1 |w:7.8,t:1| Show InChI InChI=1S/C14H9NO4S/c16-9-3-1-8(2-4-9)11-6-5-10(19-11)7-12-13(17)15-14(18)20-12/h1-7,16H,(H,15,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serono Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha |
J Med Chem 49: 3857-71 (2006)
Article DOI: 10.1021/jm0601598
BindingDB Entry DOI: 10.7270/Q2DZ07X0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data