

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50288947 (CHEMBL158033 | N*1*-((S)-1-{(2R,3S)-3-[(1-Isopropy...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against HIV protease was determined | Bioorg Med Chem Lett 6: 589-594 (1996) Article DOI: 10.1016/0960-894X(96)00087-X BindingDB Entry DOI: 10.7270/Q2FQ9WM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50288946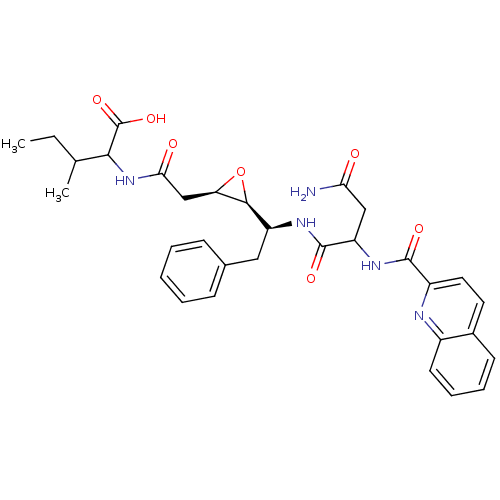 (2-{2-[(2R,3S)-3-((S)-1-{3-Carbamoyl-2-[(quinoline-...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article | 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against HIV protease was determined | Bioorg Med Chem Lett 6: 589-594 (1996) Article DOI: 10.1016/0960-894X(96)00087-X BindingDB Entry DOI: 10.7270/Q2FQ9WM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50288945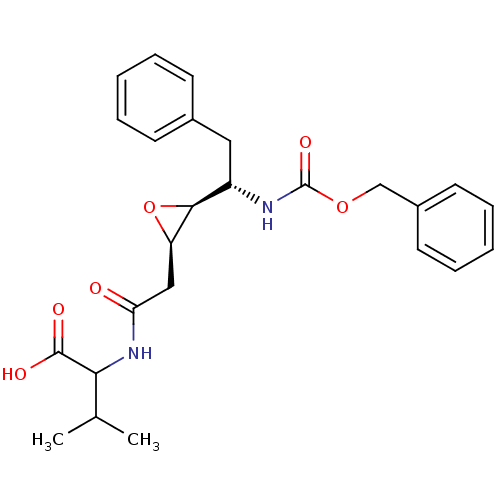 (2-{2-[(2R,3S)-3-((S)-1-Benzyloxycarbonylamino-2-ph...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article | 1.32E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against HIV protease was determined | Bioorg Med Chem Lett 6: 589-594 (1996) Article DOI: 10.1016/0960-894X(96)00087-X BindingDB Entry DOI: 10.7270/Q2FQ9WM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50288948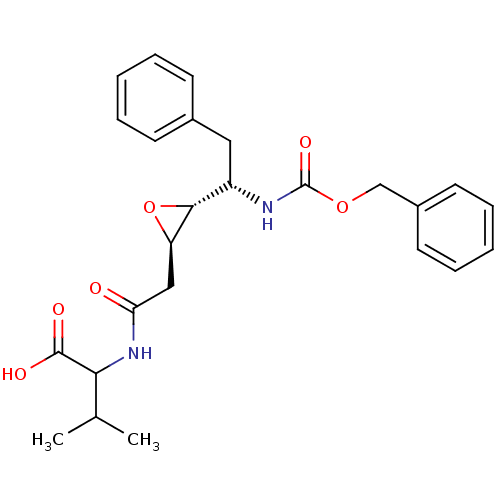 (2-{2-[(2R,3R)-3-((S)-1-Benzyloxycarbonylamino-2-ph...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article | 2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against HIV protease was determined | Bioorg Med Chem Lett 6: 589-594 (1996) Article DOI: 10.1016/0960-894X(96)00087-X BindingDB Entry DOI: 10.7270/Q2FQ9WM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50288944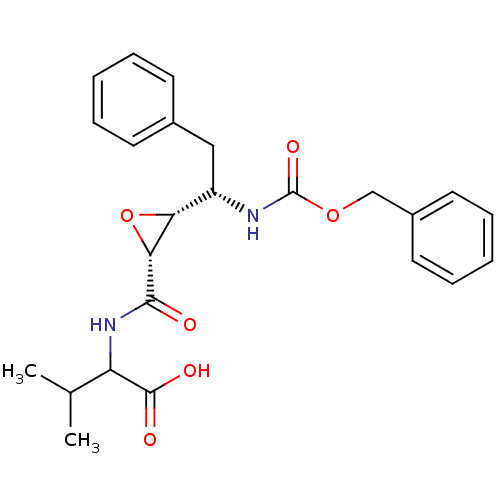 (2-{[(2R,3R)-3-((S)-1-Benzyloxycarbonylamino-2-phen...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article | >1.00E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against HIV protease was determined | Bioorg Med Chem Lett 6: 589-594 (1996) Article DOI: 10.1016/0960-894X(96)00087-X BindingDB Entry DOI: 10.7270/Q2FQ9WM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cathepsin D (Homo sapiens (Human)) | BDBM50288947 (CHEMBL158033 | N*1*-((S)-1-{(2R,3S)-3-[(1-Isopropy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article | n/a | n/a | 1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory activity against human cathepsin D was determined | Bioorg Med Chem Lett 6: 589-594 (1996) Article DOI: 10.1016/0960-894X(96)00087-X BindingDB Entry DOI: 10.7270/Q2FQ9WM4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||