

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-hydroxytryptamine receptor 2A (Homo sapiens (Human)) | BDBM50297306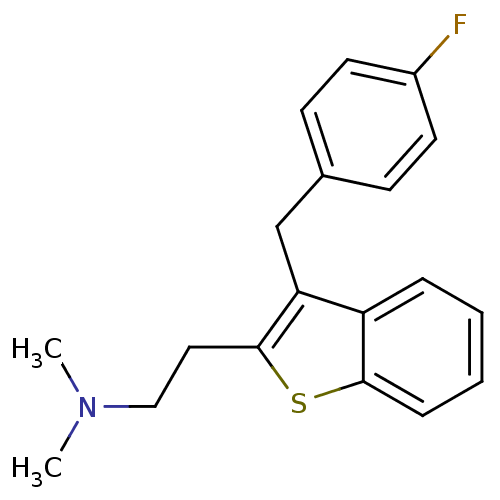 (CHEMBL540982 | {2-[3-(4-Fluoro-benzyl)-benzo[b]thi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]ketanserin from human 5HT2A receptor expressed in HEK293 Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2A (Homo sapiens (Human)) | BDBM50297304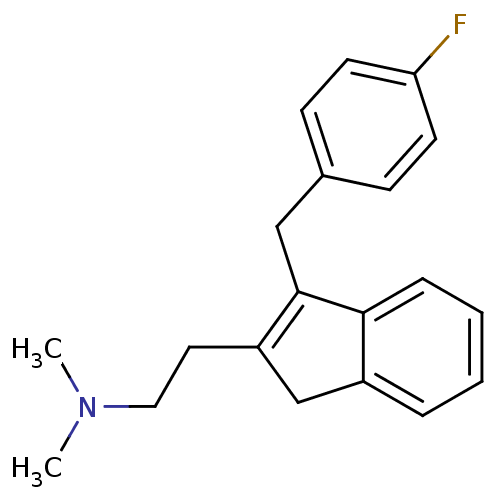 (CHEMBL560741 | {2-[3-(4-Fluoro-benzyl)-1H-inden-2-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]ketanserin from human 5HT2A receptor expressed in HEK293 Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50297310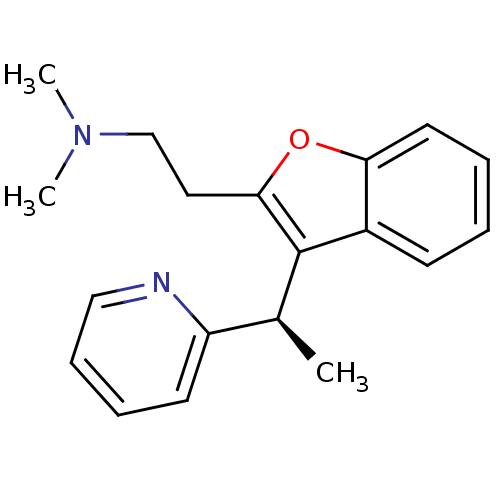 ((-)-Dimethyl-{2-[3-((R)-1-pyridin-2-yl-ethyl)-benz...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]N-methylscopolamine from human muscarinic M1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297307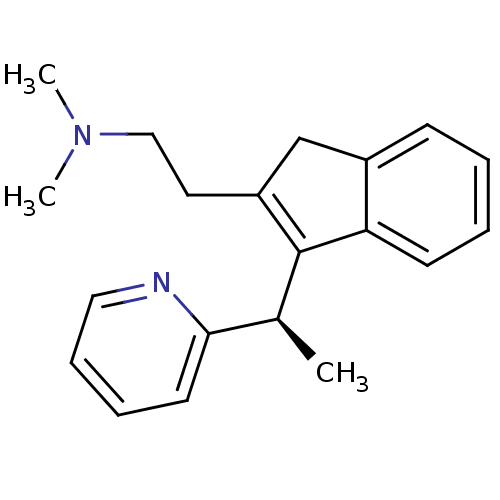 (CHEMBL564226 | R-dimethindene) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2A (Homo sapiens (Human)) | BDBM50297305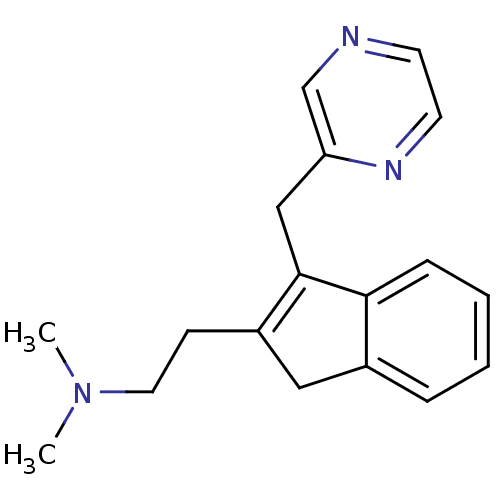 (CHEMBL559251 | Dimethyl-[2-(3-pyrazin-2ylmethyl-1H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]ketanserin from human 5HT2A receptor expressed in HEK293 Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297304 (CHEMBL560741 | {2-[3-(4-Fluoro-benzyl)-1H-inden-2-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297310 ((-)-Dimethyl-{2-[3-((R)-1-pyridin-2-yl-ethyl)-benz...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297308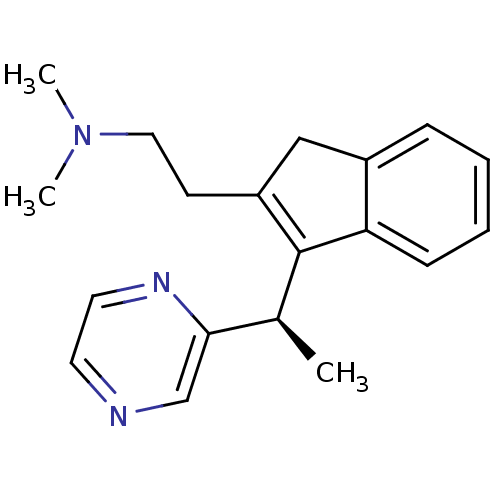 ((-)-Dimethyl-{2-[3-((R)-1-pyrazin-2-yl-ethyl)-1H-i...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50297306 (CHEMBL540982 | {2-[3-(4-Fluoro-benzyl)-benzo[b]thi...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]N-methylscopolamine from human muscarinic M1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50297307 (CHEMBL564226 | R-dimethindene) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]N-methylscopolamine from human muscarinic M1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297309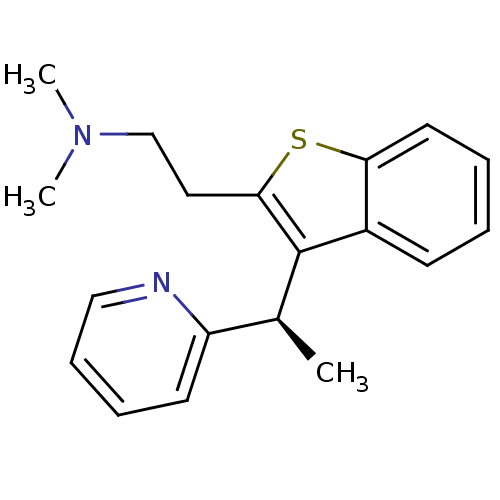 ((-)-(R)-N,N-dimethyl-2-(3-(1-(pyridin-2-yl)ethyl)b...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297305 (CHEMBL559251 | Dimethyl-[2-(3-pyrazin-2ylmethyl-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50297309 ((-)-(R)-N,N-dimethyl-2-(3-(1-(pyridin-2-yl)ethyl)b...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]N-methylscopolamine from human muscarinic M1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297306 (CHEMBL540982 | {2-[3-(4-Fluoro-benzyl)-benzo[b]thi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50297305 (CHEMBL559251 | Dimethyl-[2-(3-pyrazin-2ylmethyl-1H...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]N-methylscopolamine from human muscarinic M1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50297311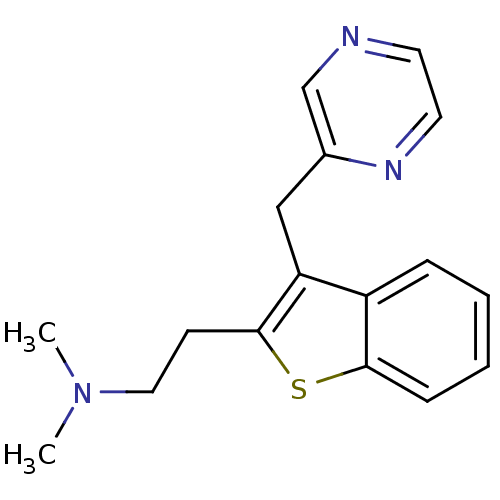 (CHEMBL551340 | Dimethyl-[2-(3-pyrazin-2-ylmethyl-b...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50297310 ((-)-Dimethyl-{2-[3-((R)-1-pyridin-2-yl-ethyl)-benz...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 76 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Displacement of [3H]N-methylscopolamine from human muscarinic M3 receptor expressed in CHO Flp-In cells by liquid scintillation counting | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297306 (CHEMBL540982 | {2-[3-(4-Fluoro-benzyl)-benzo[b]thi...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297304 (CHEMBL560741 | {2-[3-(4-Fluoro-benzyl)-1H-inden-2-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50297307 (CHEMBL564226 | R-dimethindene) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of human ERG expressed in HEK293 cells by patch-clamp technique | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50297310 ((-)-Dimethyl-{2-[3-((R)-1-pyridin-2-yl-ethyl)-benz...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of human ERG expressed in HEK293 cells by patch-clamp technique | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50297309 ((-)-(R)-N,N-dimethyl-2-(3-(1-(pyridin-2-yl)ethyl)b...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of human ERG expressed in HEK293 cells by patch-clamp technique | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50297308 ((-)-Dimethyl-{2-[3-((R)-1-pyrazin-2-yl-ethyl)-1H-i...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of human ERG expressed in HEK293 cells by patch-clamp technique | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297310 ((-)-Dimethyl-{2-[3-((R)-1-pyridin-2-yl-ethyl)-benz...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50297305 (CHEMBL559251 | Dimethyl-[2-(3-pyrazin-2ylmethyl-1H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of human ERG expressed in HEK293 cells by patch-clamp technique | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297305 (CHEMBL559251 | Dimethyl-[2-(3-pyrazin-2ylmethyl-1H...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297309 ((-)-(R)-N,N-dimethyl-2-(3-(1-(pyridin-2-yl)ethyl)b...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.82E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297307 (CHEMBL564226 | R-dimethindene) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 2D6 (Homo sapiens (Human)) | BDBM50297308 ((-)-Dimethyl-{2-[3-((R)-1-pyrazin-2-yl-ethyl)-1H-i...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.05E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Inhibition of recombinant CYP2D6 in presence of NADPH generating system | J Med Chem 52: 5307-10 (2009) Article DOI: 10.1021/jm900933k BindingDB Entry DOI: 10.7270/Q2057G0S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||