 Found 54 hits Enz. Inhib. hit(s) with all data for entry = 50033126
Found 54 hits Enz. Inhib. hit(s) with all data for entry = 50033126 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50262988
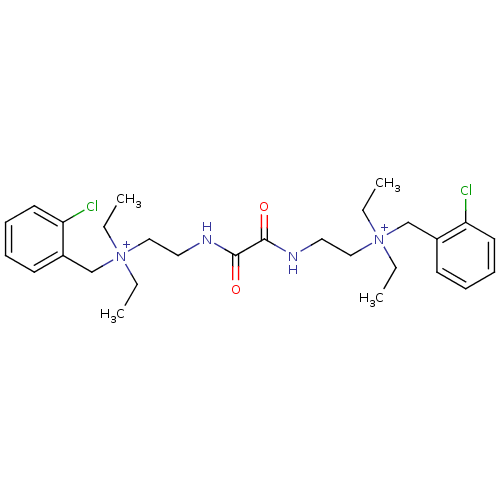
(CHEMBL1200541 | N-(2-chlorobenzyl)-2-(2-(2-((2-chl...)Show SMILES CC[N+](CC)(CCNC(=O)C(=O)NCC[N+](CC)(CC)Cc1ccccc1Cl)Cc1ccccc1Cl Show InChI InChI=1S/C28H40Cl2N4O2/c1-5-33(6-2,21-23-13-9-11-15-25(23)29)19-17-31-27(35)28(36)32-18-20-34(7-3,8-4)22-24-14-10-12-16-26(24)30/h9-16H,5-8,17-22H2,1-4H3/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50262988

(CHEMBL1200541 | N-(2-chlorobenzyl)-2-(2-(2-((2-chl...)Show SMILES CC[N+](CC)(CCNC(=O)C(=O)NCC[N+](CC)(CC)Cc1ccccc1Cl)Cc1ccccc1Cl Show InChI InChI=1S/C28H40Cl2N4O2/c1-5-33(6-2,21-23-13-9-11-15-25(23)29)19-17-31-27(35)28(36)32-18-20-34(7-3,8-4)22-24-14-10-12-16-26(24)30/h9-16H,5-8,17-22H2,1-4H3/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor-substrate complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10624

(1,5-bis(4-allyldimethylammoniumphenyl)-pentan-3-on...)Show SMILES C[N+](C)(CC=C)c1ccc(CCC(=O)CCc2ccc(cc2)[N+](C)(C)CC=C)cc1 Show InChI InChI=1S/C27H38N2O/c1-7-21-28(3,4)25-15-9-23(10-16-25)13-19-27(30)20-14-24-11-17-26(18-12-24)29(5,6)22-8-2/h7-12,15-18H,1-2,13-14,19-22H2,3-6H3/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119797

(1,1'-(decane-1,10-diyl)diquinolinium iodide | 1,10...)Show SMILES C(CCCCC[n+]1cccc2ccccc12)CCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C28H34N2/c1(3-5-11-21-29-23-13-17-25-15-7-9-19-27(25)29)2-4-6-12-22-30-24-14-18-26-16-8-10-20-28(26)30/h7-10,13-20,23-24H,1-6,11-12,21-22H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10624

(1,5-bis(4-allyldimethylammoniumphenyl)-pentan-3-on...)Show SMILES C[N+](C)(CC=C)c1ccc(CCC(=O)CCc2ccc(cc2)[N+](C)(C)CC=C)cc1 Show InChI InChI=1S/C27H38N2O/c1-7-21-28(3,4)25-15-9-23(10-16-25)13-19-27(30)20-14-24-11-17-26(18-12-24)29(5,6)22-8-2/h7-12,15-18H,1-2,13-14,19-22H2,3-6H3/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor-substrate complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119797

(1,1'-(decane-1,10-diyl)diquinolinium iodide | 1,10...)Show SMILES C(CCCCC[n+]1cccc2ccccc12)CCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C28H34N2/c1(3-5-11-21-29-23-13-17-25-15-7-9-19-27(25)29)2-4-6-12-22-30-24-14-18-26-16-8-10-20-28(26)30/h7-10,13-20,23-24H,1-6,11-12,21-22H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor-substrate complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM120262
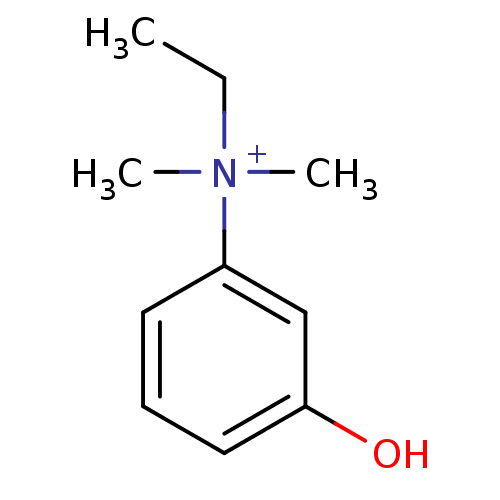
(EDROPHONIUM BROMIDE | EDROPHONIUM CHLORIDE | Edrop...)Show InChI InChI=1S/C10H15NO/c1-4-11(2,3)9-6-5-7-10(12)8-9/h5-8H,4H2,1-3H3/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
Article
PubMed
| 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339942

(1,1'-(naphthalene-2,7-diylbis(methylene))diquinoli...)Show SMILES C(c1ccc2ccc(C[n+]3cccc4ccccc34)cc2c1)[n+]1cccc2ccccc12 Show InChI InChI=1S/C30H24N2/c1-3-11-29-26(7-1)9-5-17-31(29)21-23-13-15-25-16-14-24(20-28(25)19-23)22-32-18-6-10-27-8-2-4-12-30(27)32/h1-20H,21-22H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM120262

(EDROPHONIUM BROMIDE | EDROPHONIUM CHLORIDE | Edrop...)Show InChI InChI=1S/C10H15NO/c1-4-11(2,3)9-6-5-7-10(12)8-9/h5-8H,4H2,1-3H3/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
Article
PubMed
| 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor-substrate complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339942

(1,1'-(naphthalene-2,7-diylbis(methylene))diquinoli...)Show SMILES C(c1ccc2ccc(C[n+]3cccc4ccccc34)cc2c1)[n+]1cccc2ccccc12 Show InChI InChI=1S/C30H24N2/c1-3-11-29-26(7-1)9-5-17-31(29)21-23-13-15-25-16-14-24(20-28(25)19-23)22-32-18-6-10-27-8-2-4-12-30(27)32/h1-20H,21-22H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor-substrate complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8958
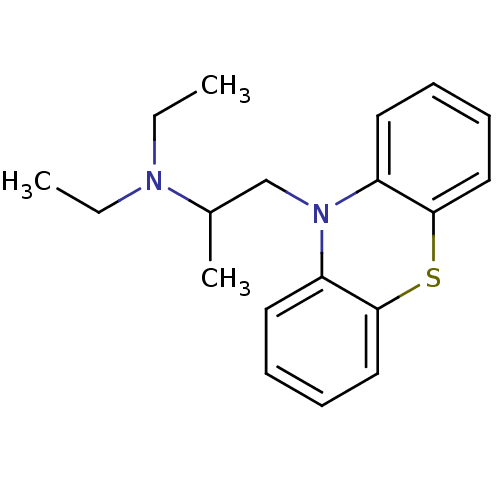
(10-(2-diethylaminopropyl)phenothiazine | CHEMBL120...)Show InChI InChI=1S/C19H24N2S/c1-4-20(5-2)15(3)14-21-16-10-6-8-12-18(16)22-19-13-9-7-11-17(19)21/h6-13,15H,4-5,14H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8958

(10-(2-diethylaminopropyl)phenothiazine | CHEMBL120...)Show InChI InChI=1S/C19H24N2S/c1-4-20(5-2)15(3)14-21-16-10-6-8-12-18(16)22-19-13-9-7-11-17(19)21/h6-13,15H,4-5,14H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.21E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocytes recombinant AChE assessed as dissociation constant for enzyme-inhibitor-substrate complex |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50262988

(CHEMBL1200541 | N-(2-chlorobenzyl)-2-(2-(2-((2-chl...)Show SMILES CC[N+](CC)(CCNC(=O)C(=O)NCC[N+](CC)(CC)Cc1ccccc1Cl)Cc1ccccc1Cl Show InChI InChI=1S/C28H40Cl2N4O2/c1-5-33(6-2,21-23-13-9-11-15-25(23)29)19-17-31-27(35)28(36)32-18-20-34(7-3,8-4)22-24-14-10-12-16-26(24)30/h9-16H,5-8,17-22H2,1-4H3/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119797

(1,1'-(decane-1,10-diyl)diquinolinium iodide | 1,10...)Show SMILES C(CCCCC[n+]1cccc2ccccc12)CCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C28H34N2/c1(3-5-11-21-29-23-13-17-25-15-7-9-19-27(25)29)2-4-6-12-22-30-24-14-18-26-16-8-10-20-28(26)30/h7-10,13-20,23-24H,1-6,11-12,21-22H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119799

(1,11-di(1-quinoliniumyl)undecane; with dibromide i...)Show SMILES C(CCCCC[n+]1cccc2ccccc12)CCCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C29H36N2/c1(2-4-6-12-22-30-24-14-18-26-16-8-10-20-28(26)30)3-5-7-13-23-31-25-15-19-27-17-9-11-21-29(27)31/h8-11,14-21,24-25H,1-7,12-13,22-23H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119789

(1,1'-(dodecane-1,12-diyl)diquinolinium bromide | 1...)Show SMILES C(CCCCCC[n+]1cccc2ccccc12)CCCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C30H38N2/c1(3-5-7-13-23-31-25-15-19-27-17-9-11-21-29(27)31)2-4-6-8-14-24-32-26-16-20-28-18-10-12-22-30(28)32/h9-12,15-22,25-26H,1-8,13-14,23-24H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10624

(1,5-bis(4-allyldimethylammoniumphenyl)-pentan-3-on...)Show SMILES C[N+](C)(CC=C)c1ccc(CCC(=O)CCc2ccc(cc2)[N+](C)(C)CC=C)cc1 Show InChI InChI=1S/C27H38N2O/c1-7-21-28(3,4)25-15-9-23(10-16-25)13-19-27(30)20-14-24-11-17-26(18-12-24)29(5,6)22-8-2/h7-12,15-18H,1-2,13-14,19-22H2,3-6H3/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119787

(1,1''-(octane-1,8-diyl)diquinolinium iodide | 1,8-...)Show SMILES C(CCCC[n+]1cccc2ccccc12)CCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C26H30N2/c1(3-9-19-27-21-11-15-23-13-5-7-17-25(23)27)2-4-10-20-28-22-12-16-24-14-6-8-18-26(24)28/h5-8,11-18,21-22H,1-4,9-10,19-20H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119772

(1,1'-(nonane-1,9-diyl)diquinolinium bromide | 1,9-...)Show SMILES C(CCCC[n+]1cccc2ccccc12)CCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C27H32N2/c1(2-4-10-20-28-22-12-16-24-14-6-8-18-26(24)28)3-5-11-21-29-23-13-17-25-15-7-9-19-27(25)29/h6-9,12-19,22-23H,1-5,10-11,20-21H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339948

(1,1'-hept-1,7-diyl-bis(quinolinium)dibromide | CHE...)Show SMILES C(CCC[n+]1cccc2ccccc12)CCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C25H28N2/c1(2-8-18-26-20-10-14-22-12-4-6-16-24(22)26)3-9-19-27-21-11-15-23-13-5-7-17-25(23)27/h4-7,10-17,20-21H,1-3,8-9,18-19H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50119797

(1,1'-(decane-1,10-diyl)diquinolinium iodide | 1,10...)Show SMILES C(CCCCC[n+]1cccc2ccccc12)CCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C28H34N2/c1(3-5-11-21-29-23-13-17-25-15-7-9-19-27(25)29)2-4-6-12-22-30-24-14-18-26-16-8-10-20-28(26)30/h7-10,13-20,23-24H,1-6,11-12,21-22H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339942

(1,1'-(naphthalene-2,7-diylbis(methylene))diquinoli...)Show SMILES C(c1ccc2ccc(C[n+]3cccc4ccccc34)cc2c1)[n+]1cccc2ccccc12 Show InChI InChI=1S/C30H24N2/c1-3-11-29-26(7-1)9-5-17-31(29)21-23-13-15-25-16-14-24(20-28(25)19-23)22-32-18-6-10-27-8-2-4-12-30(27)32/h1-20H,21-22H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339948

(1,1'-hept-1,7-diyl-bis(quinolinium)dibromide | CHE...)Show SMILES C(CCC[n+]1cccc2ccccc12)CCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C25H28N2/c1(2-8-18-26-20-10-14-22-12-4-6-16-24(22)26)3-9-19-27-21-11-15-23-13-5-7-17-25(23)27/h4-7,10-17,20-21H,1-3,8-9,18-19H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50119789

(1,1'-(dodecane-1,12-diyl)diquinolinium bromide | 1...)Show SMILES C(CCCCCC[n+]1cccc2ccccc12)CCCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C30H38N2/c1(3-5-7-13-23-31-25-15-19-27-17-9-11-21-29(27)31)2-4-6-8-14-24-32-26-16-20-28-18-10-12-22-30(28)32/h9-12,15-22,25-26H,1-8,13-14,23-24H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50119799

(1,11-di(1-quinoliniumyl)undecane; with dibromide i...)Show SMILES C(CCCCC[n+]1cccc2ccccc12)CCCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C29H36N2/c1(2-4-6-12-22-30-24-14-18-26-16-8-10-20-28(26)30)3-5-7-13-23-31-25-15-19-27-17-9-11-21-29(27)31/h8-11,14-21,24-25H,1-7,12-13,22-23H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50119772

(1,1'-(nonane-1,9-diyl)diquinolinium bromide | 1,9-...)Show SMILES C(CCCC[n+]1cccc2ccccc12)CCCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C27H32N2/c1(2-4-10-20-28-22-12-16-24-14-6-8-18-26(24)28)3-5-11-21-29-23-13-17-25-15-7-9-19-27(25)29/h6-9,12-19,22-23H,1-5,10-11,20-21H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50119787

(1,1''-(octane-1,8-diyl)diquinolinium iodide | 1,8-...)Show SMILES C(CCCC[n+]1cccc2ccccc12)CCC[n+]1cccc2ccccc12 Show InChI InChI=1S/C26H30N2/c1(3-9-19-27-21-11-15-23-13-5-7-17-25(23)27)2-4-10-20-28-22-12-16-24-14-6-8-18-26(24)28/h5-8,11-18,21-22H,1-4,9-10,19-20H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339953

((Z)-1,1'-(but-2-ene-1,4-diyl)diquinolinium chlorid...)Show SMILES C(\C=C/C[n+]1cccc2ccccc12)[n+]1cccc2ccccc12 Show InChI InChI=1S/C22H20N2/c1-3-13-21-19(9-1)11-7-17-23(21)15-5-6-16-24-18-8-12-20-10-2-4-14-22(20)24/h1-14,17-18H,15-16H2/q+2/b6-5- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339942

(1,1'-(naphthalene-2,7-diylbis(methylene))diquinoli...)Show SMILES C(c1ccc2ccc(C[n+]3cccc4ccccc34)cc2c1)[n+]1cccc2ccccc12 Show InChI InChI=1S/C30H24N2/c1-3-11-29-26(7-1)9-5-17-31(29)21-23-13-15-25-16-14-24(20-28(25)19-23)22-32-18-6-10-27-8-2-4-12-30(27)32/h1-20H,21-22H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8958

(10-(2-diethylaminopropyl)phenothiazine | CHEMBL120...)Show InChI InChI=1S/C19H24N2S/c1-4-20(5-2)15(3)14-21-16-10-6-8-12-18(16)22-19-13-9-7-11-17(19)21/h6-13,15H,4-5,14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339953

((Z)-1,1'-(but-2-ene-1,4-diyl)diquinolinium chlorid...)Show SMILES C(\C=C/C[n+]1cccc2ccccc12)[n+]1cccc2ccccc12 Show InChI InChI=1S/C22H20N2/c1-3-13-21-19(9-1)11-7-17-23(21)15-5-6-16-24-18-8-12-20-10-2-4-14-22(20)24/h1-14,17-18H,15-16H2/q+2/b6-5- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339946
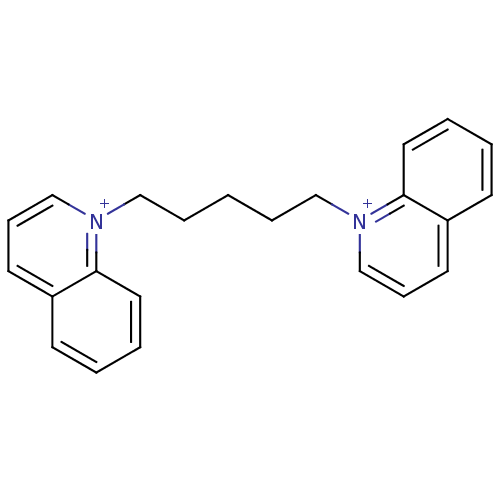
(1,1'-pent-1,5-diyl-bis(quinolinium)dibromide | CHE...)Show InChI InChI=1S/C23H24N2/c1(6-16-24-18-8-12-20-10-2-4-14-22(20)24)7-17-25-19-9-13-21-11-3-5-15-23(21)25/h2-5,8-15,18-19H,1,6-7,16-17H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM120262

(EDROPHONIUM BROMIDE | EDROPHONIUM CHLORIDE | Edrop...)Show InChI InChI=1S/C10H15NO/c1-4-11(2,3)9-6-5-7-10(12)8-9/h5-8H,4H2,1-3H3/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50119796
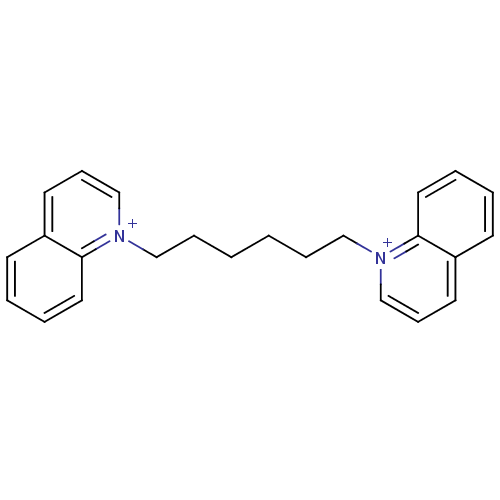
(1,1'-(hexane-1,6-diyl)diquinolinium iodide | 1,6-d...)Show InChI InChI=1S/C24H26N2/c1(7-17-25-19-9-13-21-11-3-5-15-23(21)25)2-8-18-26-20-10-14-22-12-4-6-16-24(22)26/h3-6,9-16,19-20H,1-2,7-8,17-18H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339954

(1,1'-(phenylene-1,2-dimethylenyl)-bis(quinolinium)...)Show SMILES C(c1ccccc1C[n+]1cccc2ccccc12)[n+]1cccc2ccccc12 Show InChI InChI=1S/C26H22N2/c1-2-12-24(20-28-18-8-14-22-10-4-6-16-26(22)28)23(11-1)19-27-17-7-13-21-9-3-5-15-25(21)27/h1-18H,19-20H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339952
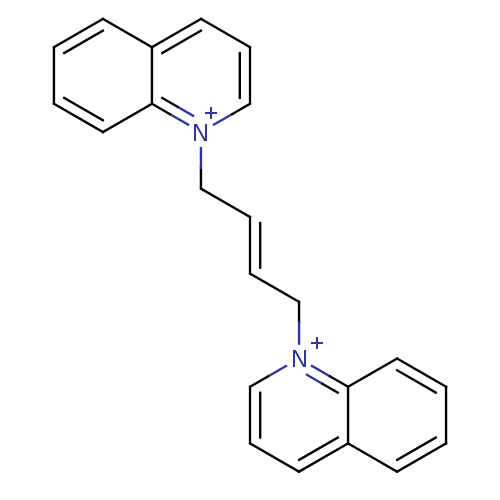
(1,1'-(but-2-ene-1,4-diyl)diquinolinium bromide | C...)Show SMILES C(\C=C\C[n+]1cccc2ccccc12)[n+]1cccc2ccccc12 Show InChI InChI=1S/C22H20N2/c1-3-13-21-19(9-1)11-7-17-23(21)15-5-6-16-24-18-8-12-20-10-2-4-14-22(20)24/h1-14,17-18H,15-16H2/q+2/b6-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50262988

(CHEMBL1200541 | N-(2-chlorobenzyl)-2-(2-(2-((2-chl...)Show SMILES CC[N+](CC)(CCNC(=O)C(=O)NCC[N+](CC)(CC)Cc1ccccc1Cl)Cc1ccccc1Cl Show InChI InChI=1S/C28H40Cl2N4O2/c1-5-33(6-2,21-23-13-9-11-15-25(23)29)19-17-31-27(35)28(36)32-18-20-34(7-3,8-4)22-24-14-10-12-16-26(24)30/h9-16H,5-8,17-22H2,1-4H3/p+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339944
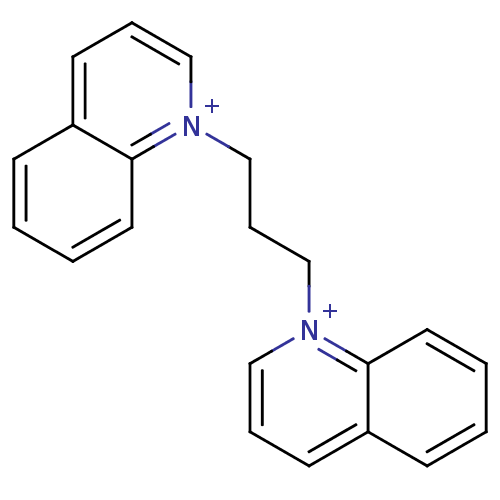
(1,1'-prop-1,3-diyl-bis(quinolinium)dibromide | CHE...)Show InChI InChI=1S/C21H20N2/c1-3-12-20-18(8-1)10-5-14-22(20)16-7-17-23-15-6-11-19-9-2-4-13-21(19)23/h1-6,8-15H,7,16-17H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339944

(1,1'-prop-1,3-diyl-bis(quinolinium)dibromide | CHE...)Show InChI InChI=1S/C21H20N2/c1-3-12-20-18(8-1)10-5-14-22(20)16-7-17-23-15-6-11-19-9-2-4-13-21(19)23/h1-6,8-15H,7,16-17H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339955
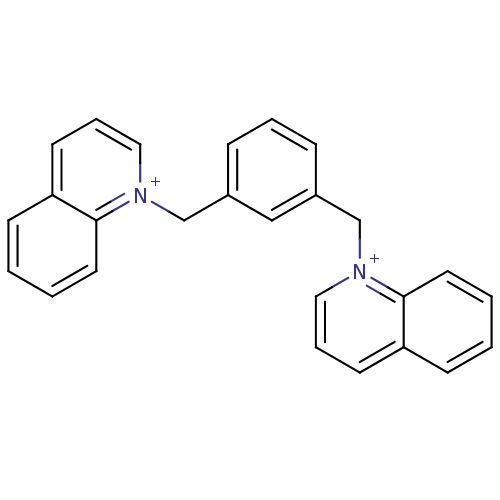
(1,1'-(phenylene-1,3-dimethylenyl)-bis(quinolinium)...)Show SMILES C(c1cccc(C[n+]2cccc3ccccc23)c1)[n+]1cccc2ccccc12 Show InChI InChI=1S/C26H22N2/c1-3-14-25-23(10-1)12-6-16-27(25)19-21-8-5-9-22(18-21)20-28-17-7-13-24-11-2-4-15-26(24)28/h1-18H,19-20H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50119796

(1,1'-(hexane-1,6-diyl)diquinolinium iodide | 1,6-d...)Show InChI InChI=1S/C24H26N2/c1(7-17-25-19-9-13-21-11-3-5-15-23(21)25)2-8-18-26-20-10-14-22-12-4-6-16-24(22)26/h3-6,9-16,19-20H,1-2,7-8,17-18H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339945
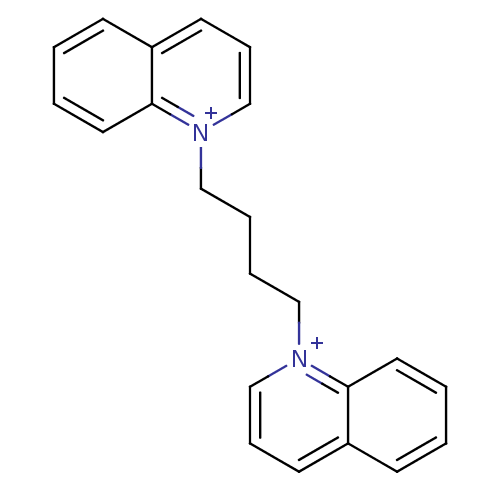
(1,1'-but-1,4-diyl-bis(quinolinium)dibromide | CHEM...)Show InChI InChI=1S/C22H22N2/c1-3-13-21-19(9-1)11-7-17-23(21)15-5-6-16-24-18-8-12-20-10-2-4-14-22(20)24/h1-4,7-14,17-18H,5-6,15-16H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339946

(1,1'-pent-1,5-diyl-bis(quinolinium)dibromide | CHE...)Show InChI InChI=1S/C23H24N2/c1(6-16-24-18-8-12-20-10-2-4-14-22(20)24)7-17-25-19-9-13-21-11-3-5-15-23(21)25/h2-5,8-15,18-19H,1,6-7,16-17H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339951
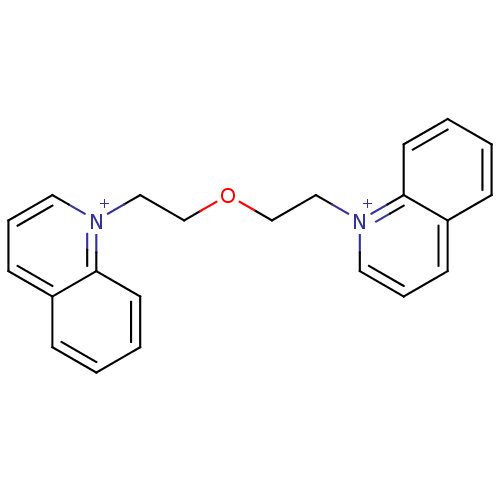
(1,1'-(2,2'-oxybis(ethane-2,1-diyl))diquinolinium b...)Show InChI InChI=1S/C22H22N2O/c1-3-11-21-19(7-1)9-5-13-23(21)15-17-25-18-16-24-14-6-10-20-8-2-4-12-22(20)24/h1-14H,15-18H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339954

(1,1'-(phenylene-1,2-dimethylenyl)-bis(quinolinium)...)Show SMILES C(c1ccccc1C[n+]1cccc2ccccc12)[n+]1cccc2ccccc12 Show InChI InChI=1S/C26H22N2/c1-2-12-24(20-28-18-8-14-22-10-4-6-16-26(22)28)23(11-1)19-27-17-7-13-21-9-3-5-15-25(21)27/h1-18H,19-20H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339945

(1,1'-but-1,4-diyl-bis(quinolinium)dibromide | CHEM...)Show InChI InChI=1S/C22H22N2/c1-3-13-21-19(9-1)11-7-17-23(21)15-5-6-16-24-18-8-12-20-10-2-4-14-22(20)24/h1-4,7-14,17-18H,5-6,15-16H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50339955

(1,1'-(phenylene-1,3-dimethylenyl)-bis(quinolinium)...)Show SMILES C(c1cccc(C[n+]2cccc3ccccc23)c1)[n+]1cccc2ccccc12 Show InChI InChI=1S/C26H22N2/c1-3-14-25-23(10-1)12-6-16-27(25)19-21-8-5-9-22(18-21)20-28-17-7-13-24-11-2-4-15-26(24)28/h1-18H,19-20H2/q+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human erythrocyte recombinant AChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339951

(1,1'-(2,2'-oxybis(ethane-2,1-diyl))diquinolinium b...)Show InChI InChI=1S/C22H22N2O/c1-3-11-21-19(7-1)9-5-13-23(21)15-17-25-18-16-24-14-6-10-20-8-2-4-12-22(20)24/h1-14H,15-18H2/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50339952

(1,1'-(but-2-ene-1,4-diyl)diquinolinium bromide | C...)Show SMILES C(\C=C\C[n+]1cccc2ccccc12)[n+]1cccc2ccccc12 Show InChI InChI=1S/C22H20N2/c1-3-13-21-19(9-1)11-7-17-23(21)15-5-6-16-24-18-8-12-20-10-2-4-14-22(20)24/h1-14,17-18H,15-16H2/q+2/b6-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.08E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM10624

(1,5-bis(4-allyldimethylammoniumphenyl)-pentan-3-on...)Show SMILES C[N+](C)(CC=C)c1ccc(CCC(=O)CCc2ccc(cc2)[N+](C)(C)CC=C)cc1 Show InChI InChI=1S/C27H38N2O/c1-7-21-28(3,4)25-15-9-23(10-16-25)13-19-27(30)20-14-24-11-17-26(18-12-24)29(5,6)22-8-2/h7-12,15-18H,1-2,13-14,19-22H2,3-6H3/q+2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.54E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Charles University
Curated by ChEMBL
| Assay Description
Inhibition of human plasmatic BChE by modified Ellman's method |
Bioorg Med Chem Lett 21: 2505-9 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.047
BindingDB Entry DOI: 10.7270/Q2JW8F6N |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data