 Found 48 hits Enz. Inhib. hit(s) with all data for entry = 50036574
Found 48 hits Enz. Inhib. hit(s) with all data for entry = 50036574 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50449787

(CHEMBL2062154 | PD-134308)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)NC[C@H](NC(=O)CCC(O)=O)c1ccccc1)[C@@]([H])(C2)C3 |wU:30.41,14.15,45.49,3.3,wD:6.6,1.0,TLB:5:3:47:9.6.8,10:9:47:3.48.2,THB:5:6:47:3.48.2,10:9:1.47.8:3.5.48,2:3:9:1.47.8,2:1:9:3.5.48,(-14.99,-2.1,;-13.56,-2.66,;-14.77,-3.94,;-13.26,-3.53,;-13.35,-5.06,;-11.86,-4.09,;-10.83,-2.82,;-9.38,-3.33,;-12.24,-3.16,;-10.83,-1.28,;-9.29,-1.31,;-8.5,.01,;-9.25,1.36,;-6.96,-.01,;-6.19,1.3,;-5.42,-.02,;-7.44,2.2,;-7.28,3.74,;-8.44,4.76,;-7.81,6.18,;-6.29,6.03,;-5.14,7.07,;-3.69,6.6,;-3.34,5.08,;-4.49,4.06,;-5.94,4.52,;-4.66,1.42,;-3.99,2.8,;-3.79,.15,;-2.27,.27,;-1.4,-1.02,;-2.08,-2.4,;-1.22,-3.69,;.32,-3.58,;-1.9,-5.07,;-3.43,-5.16,;-4.1,-6.57,;-5.64,-6.67,;-3.25,-7.83,;.14,-.91,;.99,-2.2,;2.51,-2.08,;3.19,-.7,;2.32,.59,;.8,.47,;-12.23,-.7,;-12.2,.82,;-13.58,-1.18,;-13.27,-1.94,)| Show InChI InChI=1S/C35H42N4O6/c1-35(18-26-19-36-28-10-6-5-9-27(26)28,39-34(44)45-32-24-14-21-13-22(16-24)17-25(32)15-21)33(43)37-20-29(23-7-3-2-4-8-23)38-30(40)11-12-31(41)42/h2-10,19,21-22,24-25,29,32,36H,11-18,20H2,1H3,(H,37,43)(H,38,40)(H,39,44)(H,41,42)/t21-,22+,24-,25+,29-,32?,35+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061266
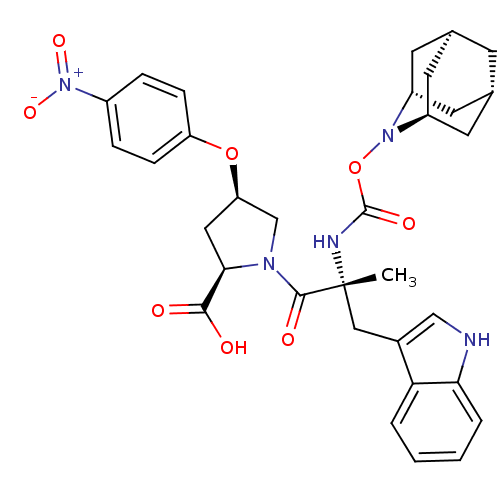
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(cc1)[N+]([O-])=O |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H37N5O8/c1-33(16-21-17-34-28-5-3-2-4-27(21)28,35-32(42)46-37-23-11-19-10-20(13-23)14-24(37)12-19)31(41)36-18-26(15-29(36)30(39)40)45-25-8-6-22(7-9-25)38(43)44/h2-9,17,19-20,23-24,26,29,34H,10-16,18H2,1H3,(H,35,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 1) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061270
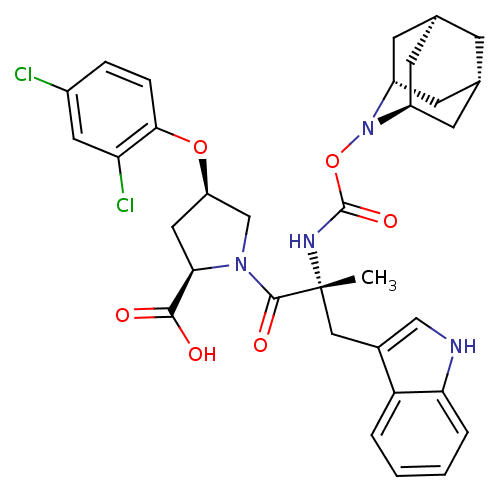
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1Cl |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36Cl2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 1) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM81962
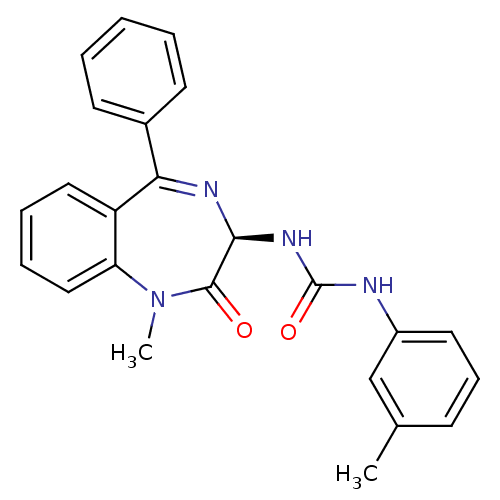
(S-L-365,260)Show SMILES CN1c2ccccc2C(=N[C@@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1 |r,c:9| Show InChI InChI=1S/C24H22N4O2/c1-16-9-8-12-18(15-16)25-24(30)27-22-23(29)28(2)20-14-7-6-13-19(20)21(26-22)17-10-4-3-5-11-17/h3-15,22H,1-2H3,(H2,25,27,30)/t22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061276
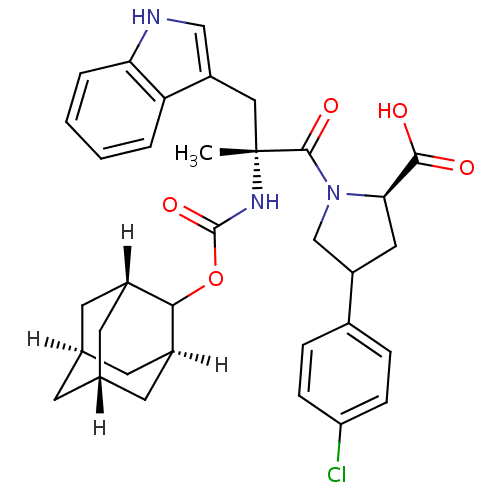
((R)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-3-(1...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)OC1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1CC(C[C@@H]1C(O)=O)c1ccc(Cl)cc1 |TLB:15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25| Show InChI InChI=1S/C34H38ClN3O5/c1-34(16-25-17-36-28-5-3-2-4-27(25)28,37-33(42)43-30-22-11-19-10-20(13-22)14-23(30)12-19)32(41)38-18-24(15-29(38)31(39)40)21-6-8-26(35)9-7-21/h2-9,17,19-20,22-24,29-30,36H,10-16,18H2,1H3,(H,37,42)(H,39,40)/t19-,20+,22-,23+,24?,29-,30?,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061267
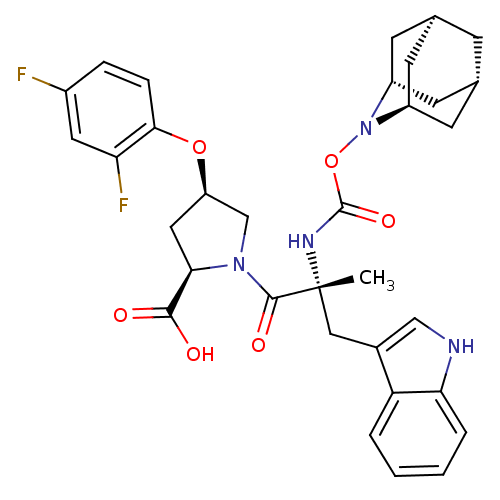
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1F |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36F2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 1) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50449787

(CHEMBL2062154 | PD-134308)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)NC[C@H](NC(=O)CCC(O)=O)c1ccccc1)[C@@]([H])(C2)C3 |wU:30.41,14.15,45.49,3.3,wD:6.6,1.0,TLB:5:3:47:9.6.8,10:9:47:3.48.2,THB:5:6:47:3.48.2,10:9:1.47.8:3.5.48,2:3:9:1.47.8,2:1:9:3.5.48,(-14.99,-2.1,;-13.56,-2.66,;-14.77,-3.94,;-13.26,-3.53,;-13.35,-5.06,;-11.86,-4.09,;-10.83,-2.82,;-9.38,-3.33,;-12.24,-3.16,;-10.83,-1.28,;-9.29,-1.31,;-8.5,.01,;-9.25,1.36,;-6.96,-.01,;-6.19,1.3,;-5.42,-.02,;-7.44,2.2,;-7.28,3.74,;-8.44,4.76,;-7.81,6.18,;-6.29,6.03,;-5.14,7.07,;-3.69,6.6,;-3.34,5.08,;-4.49,4.06,;-5.94,4.52,;-4.66,1.42,;-3.99,2.8,;-3.79,.15,;-2.27,.27,;-1.4,-1.02,;-2.08,-2.4,;-1.22,-3.69,;.32,-3.58,;-1.9,-5.07,;-3.43,-5.16,;-4.1,-6.57,;-5.64,-6.67,;-3.25,-7.83,;.14,-.91,;.99,-2.2,;2.51,-2.08,;3.19,-.7,;2.32,.59,;.8,.47,;-12.23,-.7,;-12.2,.82,;-13.58,-1.18,;-13.27,-1.94,)| Show InChI InChI=1S/C35H42N4O6/c1-35(18-26-19-36-28-10-6-5-9-27(26)28,39-34(44)45-32-24-14-21-13-22(16-24)17-25(32)15-21)33(43)37-20-29(23-7-3-2-4-8-23)38-30(40)11-12-31(41)42/h2-10,19,21-22,24-25,29,32,36H,11-18,20H2,1H3,(H,37,43)(H,38,40)(H,39,44)(H,41,42)/t21-,22+,24-,25+,29-,32?,35+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061284

((R)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-3-(1...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)OC1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1CC(C[C@@H]1C(O)=O)c1ccccc1 |TLB:15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25| Show InChI InChI=1S/C34H39N3O5/c1-34(17-26-18-35-28-10-6-5-9-27(26)28,36-33(41)42-30-23-12-20-11-21(14-23)15-24(30)13-20)32(40)37-19-25(16-29(37)31(38)39)22-7-3-2-4-8-22/h2-10,18,20-21,23-25,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,23-,24+,25?,29-,30?,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061276

((R)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-3-(1...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)OC1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1CC(C[C@@H]1C(O)=O)c1ccc(Cl)cc1 |TLB:15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25| Show InChI InChI=1S/C34H38ClN3O5/c1-34(16-25-17-36-28-5-3-2-4-27(25)28,37-33(42)43-30-22-11-19-10-20(13-22)14-23(30)12-19)32(41)38-18-24(15-29(38)31(39)40)21-6-8-26(35)9-7-21/h2-9,17,19-20,22-24,29-30,36H,10-16,18H2,1H3,(H,37,42)(H,39,40)/t19-,20+,22-,23+,24?,29-,30?,34-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compounds were tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM81962

(S-L-365,260)Show SMILES CN1c2ccccc2C(=N[C@@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1 |r,c:9| Show InChI InChI=1S/C24H22N4O2/c1-16-9-8-12-18(15-16)25-24(30)27-22-23(29)28(2)20-14-7-6-13-19(20)21(26-22)17-10-4-3-5-11-17/h3-15,22H,1-2H3,(H2,25,27,30)/t22-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061270

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1Cl |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36Cl2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061267

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1F |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36F2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061267

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1F |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36F2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061273

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37FN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061285
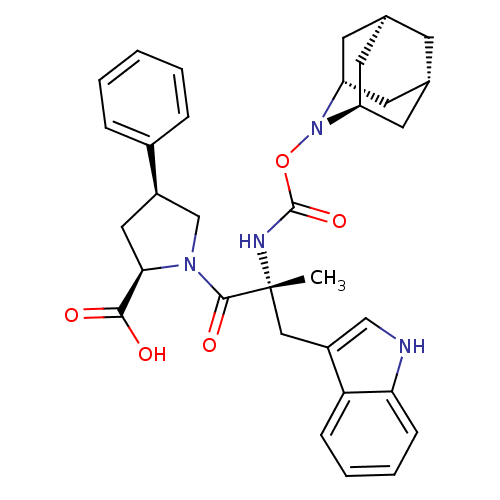
((2R,4S)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)c1ccccc1 |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:16.23.24:18,15:16:25:19.18.20| Show InChI InChI=1S/C33H38N4O5/c1-33(17-24-18-34-28-10-6-5-9-27(24)28,35-32(41)42-37-25-12-20-11-21(14-25)15-26(37)13-20)31(40)36-19-23(16-29(36)30(38)39)22-7-3-2-4-8-22/h2-10,18,20-21,23,25-26,29,34H,11-17,19H2,1H3,(H,35,41)(H,38,39)/t20-,21+,23-,25-,26+,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061271

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37ClN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061271

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37ClN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061283
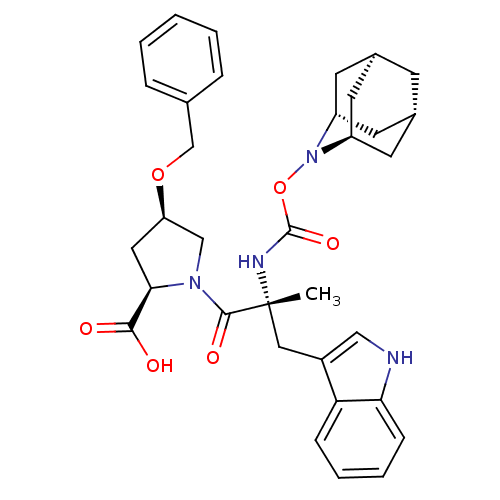
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)OCc1ccccc1 |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:16.23.24:18,15:16:25:19.18.20| Show InChI InChI=1S/C34H40N4O6/c1-34(17-24-18-35-29-10-6-5-9-28(24)29,36-33(42)44-38-25-12-22-11-23(14-25)15-26(38)13-22)32(41)37-19-27(16-30(37)31(39)40)43-20-21-7-3-2-4-8-21/h2-10,18,22-23,25-27,30,35H,11-17,19-20H2,1H3,(H,36,42)(H,39,40)/t22-,23+,25-,26+,27-,30-,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061266

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(cc1)[N+]([O-])=O |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H37N5O8/c1-33(16-21-17-34-28-5-3-2-4-27(21)28,35-32(42)46-37-23-11-19-10-20(13-23)14-24(37)12-19)31(41)36-18-26(15-29(36)30(39)40)45-25-8-6-22(7-9-25)38(43)44/h2-9,17,19-20,23-24,26,29,34H,10-16,18H2,1H3,(H,35,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061273

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37FN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061280
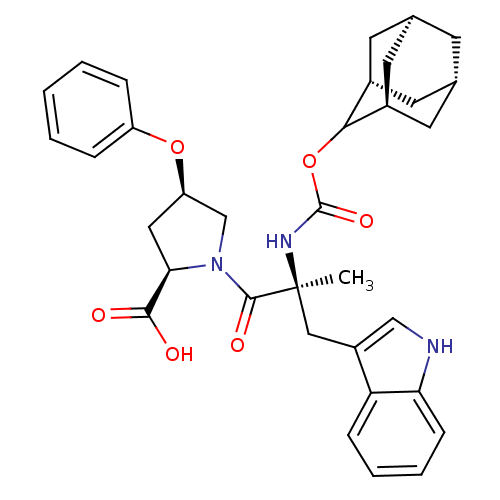
((2R,4R)-1-[(S)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:43.48,3.3,wD:14.28,32.36,30.39,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.49,-5.79,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29-,30?,34+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061270

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1Cl |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36Cl2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 2) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061287
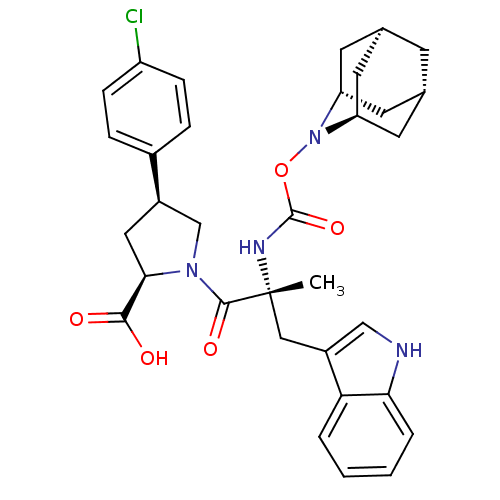
((2R,4S)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)c1ccc(Cl)cc1 |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:16.23.24:18,15:16:25:19.18.20| Show InChI InChI=1S/C33H37ClN4O5/c1-33(16-23-17-35-28-5-3-2-4-27(23)28,36-32(42)43-38-25-11-19-10-20(13-25)14-26(38)12-19)31(41)37-18-22(15-29(37)30(39)40)21-6-8-24(34)9-7-21/h2-9,17,19-20,22,25-26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,22-,25-,26+,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061266

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(cc1)[N+]([O-])=O |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H37N5O8/c1-33(16-21-17-34-28-5-3-2-4-27(21)28,35-32(42)46-37-23-11-19-10-20(13-23)14-24(37)12-19)31(41)36-18-26(15-29(36)30(39)40)45-25-8-6-22(7-9-25)38(43)44/h2-9,17,19-20,23-24,26,29,34H,10-16,18H2,1H3,(H,35,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061268
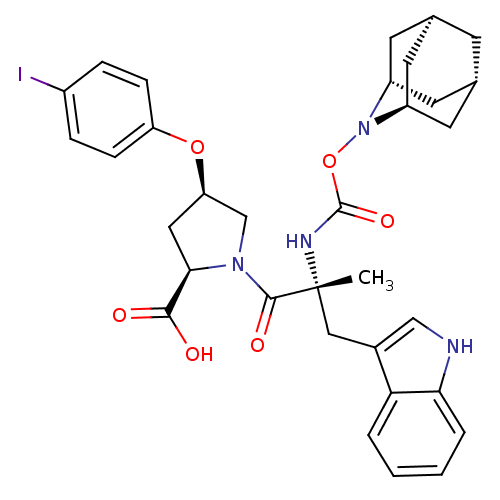
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(I)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37IN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 45 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061270

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1Cl |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36Cl2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 46 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061282
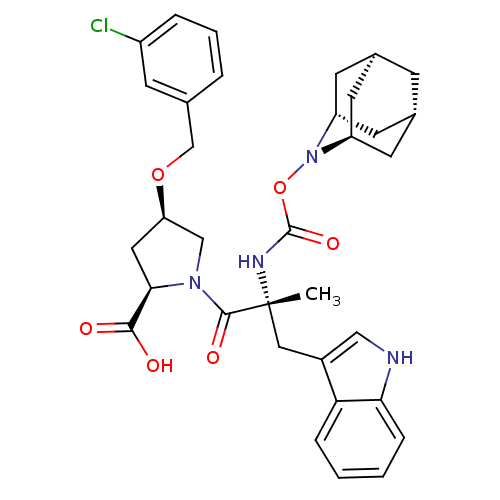
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)OCc1cccc(Cl)c1 |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:16.23.24:18,15:16:25:19.18.20| Show InChI InChI=1S/C34H39ClN4O6/c1-34(16-23-17-36-29-8-3-2-7-28(23)29,37-33(43)45-39-25-11-21-9-22(13-25)14-26(39)12-21)32(42)38-18-27(15-30(38)31(40)41)44-19-20-5-4-6-24(35)10-20/h2-8,10,17,21-22,25-27,30,36H,9,11-16,18-19H2,1H3,(H,37,43)(H,40,41)/t21-,22+,25-,26+,27-,30-,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061275
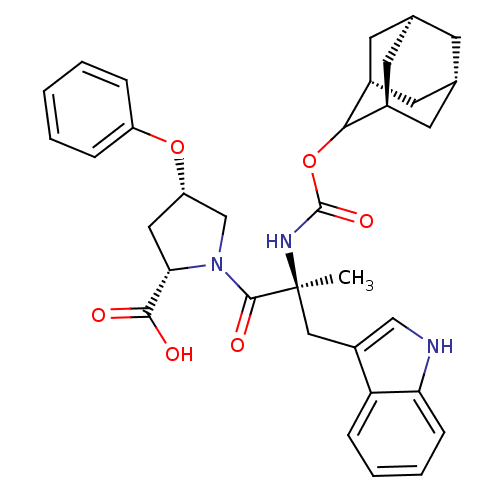
((2S,4S)-1-[(S)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@H](C[C@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:32.36,30.39,43.48,3.3,wD:14.28,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.49,-6.05,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29-,30?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061268

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(I)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37IN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for binding affinity against rat brain Cholecystokinin type B receptor expressed in CHO cells. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061266

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(cc1)[N+]([O-])=O |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H37N5O8/c1-33(16-21-17-34-28-5-3-2-4-27(21)28,35-32(42)46-37-23-11-19-10-20(13-23)14-24(37)12-19)31(41)36-18-26(15-29(36)30(39)40)45-25-8-6-22(7-9-25)38(43)44/h2-9,17,19-20,23-24,26,29,34H,10-16,18H2,1H3,(H,35,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 76 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 2) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061286
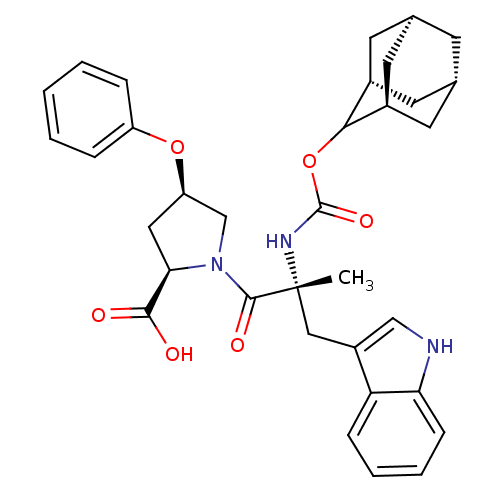
((2R,4R)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:14.28,43.48,3.3,wD:32.36,30.39,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.56,-5.72,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29-,30?,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 91 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061288
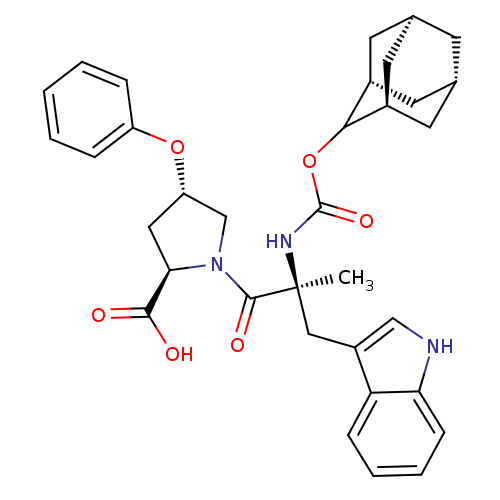
((2R,4S)-1-[(S)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@H](C[C@@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:30.39,43.48,3.3,wD:14.28,32.36,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.17,-5.95,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29+,30?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 98 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061281
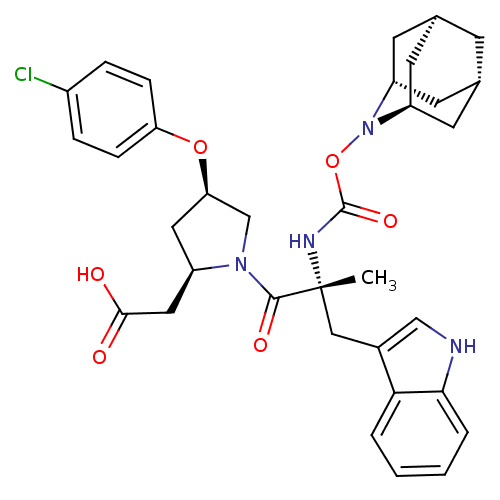
(CHEMBL338252 | [(2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1CC(O)=O)Oc1ccc(Cl)cc1 |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:16.23.24:18,15:16:25:19.18.20| Show InChI InChI=1S/C34H39ClN4O6/c1-34(17-22-18-36-30-5-3-2-4-29(22)30,37-33(43)45-39-25-11-20-10-21(13-25)14-26(39)12-20)32(42)38-19-28(15-24(38)16-31(40)41)44-27-8-6-23(35)7-9-27/h2-9,18,20-21,24-26,28,36H,10-17,19H2,1H3,(H,37,43)(H,40,41)/t20-,21+,24-,25-,26+,28-,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 122 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061272
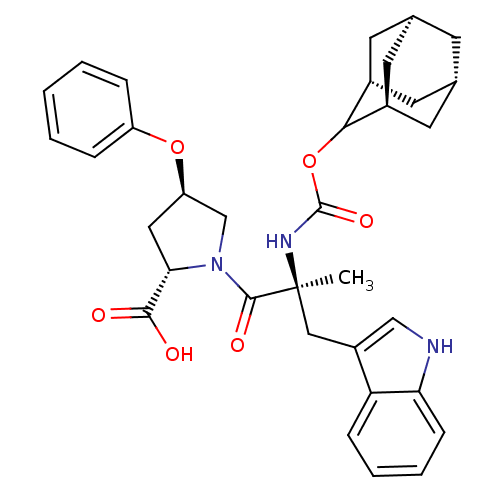
((2S,4R)-1-[(S)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@@H](C[C@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:32.36,43.48,3.3,wD:14.28,30.39,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.56,-5.88,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29+,30?,34+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 285 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061267

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1F |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36F2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 324 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 2) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061279
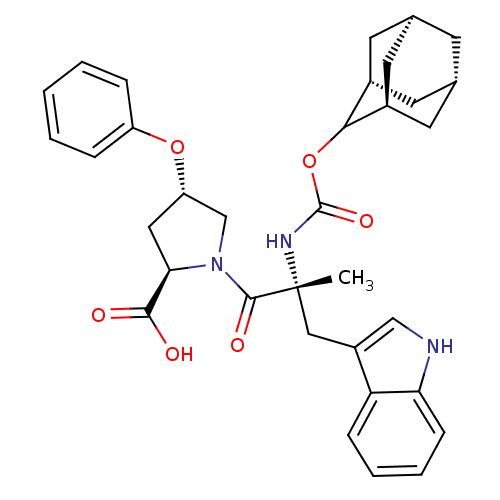
((2R,4S)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@H](C[C@@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:14.28,30.39,43.48,3.3,wD:32.36,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.49,-6.05,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29+,30?,34+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061278
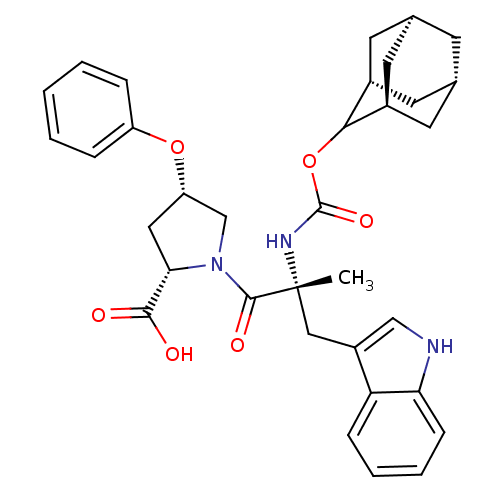
((2S,4S)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@H](C[C@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:14.28,32.36,30.39,43.48,3.3,wD:6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.4,-5.72,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29-,30?,34+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061269

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1cccc2ccccc12 |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:16.23.24:18,15:16:25:19.18.20| Show InChI InChI=1S/C37H40N4O6/c1-37(19-25-20-38-31-11-5-4-9-29(25)31,39-36(45)47-41-26-14-22-13-23(16-26)17-27(41)15-22)35(44)40-21-28(18-32(40)34(42)43)46-33-12-6-8-24-7-2-3-10-30(24)33/h2-12,20,22-23,26-28,32,38H,13-19,21H2,1H3,(H,39,45)(H,42,43)/t22-,23+,26-,27+,28-,32-,37-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 775 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061277
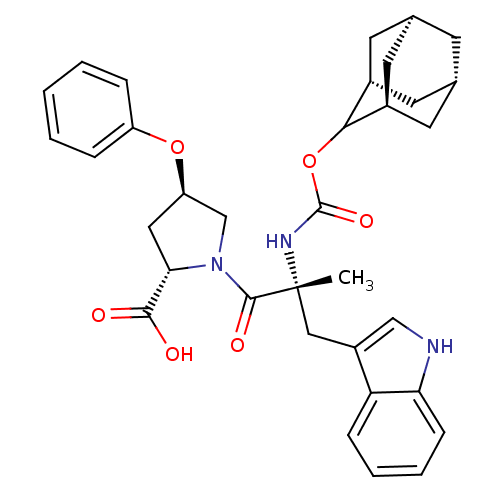
((2S,4R)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-...)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)N1C[C@@H](C[C@H]1C(O)=O)Oc1ccccc1)[C@@]([H])(C2)C3 |wU:14.28,32.36,43.48,3.3,wD:30.39,6.6,1.0,TLB:5:3:45:9.6.8,10:9:45:3.46.2,THB:5:6:45:3.46.2,2:3:9:1.45.8,2:1:9:3.5.46,(5.34,-8.16,;6.78,-8.71,;5.58,-10,;7.08,-9.56,;6.98,-11.1,;8.47,-10.13,;9.49,-8.85,;10.92,-9.37,;8.08,-9.21,;9.5,-7.34,;10.82,-6.56,;12.14,-7.3,;12.97,-5.88,;12.17,-8.85,;13.69,-8.85,;13.69,-7.3,;13.69,-10.37,;12.82,-11.81,;13.47,-13.37,;12.21,-14.47,;10.78,-13.56,;9.18,-14.05,;7.95,-12.89,;8.34,-11.26,;9.95,-10.79,;11.17,-11.95,;15.23,-8.85,;15.98,-10.18,;15.98,-7.52,;15.37,-6.11,;16.5,-5.1,;17.84,-5.85,;17.52,-7.36,;18.53,-8.5,;20.04,-8.17,;18.07,-9.95,;16.34,-3.56,;17.6,-2.66,;17.43,-1.14,;18.65,-.24,;20.07,-.87,;20.23,-2.4,;18.99,-3.29,;8.11,-6.76,;8.14,-5.24,;6.76,-7.24,;7.07,-8,)| Show InChI InChI=1S/C34H39N3O6/c1-34(17-24-18-35-28-10-6-5-9-27(24)28,36-33(41)43-30-22-12-20-11-21(14-22)15-23(30)13-20)32(40)37-19-26(16-29(37)31(38)39)42-25-7-3-2-4-8-25/h2-10,18,20-23,26,29-30,35H,11-17,19H2,1H3,(H,36,41)(H,38,39)/t20-,21+,22-,23+,26-,29+,30?,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 791 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50449787

(CHEMBL2062154 | PD-134308)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)NC[C@H](NC(=O)CCC(O)=O)c1ccccc1)[C@@]([H])(C2)C3 |wU:30.41,14.15,45.49,3.3,wD:6.6,1.0,TLB:5:3:47:9.6.8,10:9:47:3.48.2,THB:5:6:47:3.48.2,10:9:1.47.8:3.5.48,2:3:9:1.47.8,2:1:9:3.5.48,(-14.99,-2.1,;-13.56,-2.66,;-14.77,-3.94,;-13.26,-3.53,;-13.35,-5.06,;-11.86,-4.09,;-10.83,-2.82,;-9.38,-3.33,;-12.24,-3.16,;-10.83,-1.28,;-9.29,-1.31,;-8.5,.01,;-9.25,1.36,;-6.96,-.01,;-6.19,1.3,;-5.42,-.02,;-7.44,2.2,;-7.28,3.74,;-8.44,4.76,;-7.81,6.18,;-6.29,6.03,;-5.14,7.07,;-3.69,6.6,;-3.34,5.08,;-4.49,4.06,;-5.94,4.52,;-4.66,1.42,;-3.99,2.8,;-3.79,.15,;-2.27,.27,;-1.4,-1.02,;-2.08,-2.4,;-1.22,-3.69,;.32,-3.58,;-1.9,-5.07,;-3.43,-5.16,;-4.1,-6.57,;-5.64,-6.67,;-3.25,-7.83,;.14,-.91,;.99,-2.2,;2.51,-2.08,;3.19,-.7,;2.32,.59,;.8,.47,;-12.23,-.7,;-12.2,.82,;-13.58,-1.18,;-13.27,-1.94,)| Show InChI InChI=1S/C35H42N4O6/c1-35(18-26-19-36-28-10-6-5-9-27(26)28,39-34(44)45-32-24-14-21-13-22(16-24)17-25(32)15-21)33(43)37-20-29(23-7-3-2-4-8-23)38-30(40)11-12-31(41)42/h2-10,19,21-22,24-25,29,32,36H,11-18,20H2,1H3,(H,37,43)(H,38,40)(H,39,44)(H,41,42)/t21-,22+,24-,25+,29-,32?,35+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061270

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1Cl |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36Cl2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM81962

(S-L-365,260)Show SMILES CN1c2ccccc2C(=N[C@@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1 |r,c:9| Show InChI InChI=1S/C24H22N4O2/c1-16-9-8-12-18(15-16)25-24(30)27-22-23(29)28(2)20-14-7-6-13-19(20)21(26-22)17-10-4-3-5-11-17/h3-15,22H,1-2H3,(H2,25,27,30)/t22-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061268

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(I)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37IN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061273

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37FN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061271

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(Cl)cc1 |TLB:25:17:24:21.22.20,15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25,15:16:24:21.22.20| Show InChI InChI=1S/C33H37ClN4O6/c1-33(16-21-17-35-28-5-3-2-4-27(21)28,36-32(42)44-38-23-11-19-10-20(13-23)14-24(38)12-19)31(41)37-18-26(15-29(37)30(39)40)43-25-8-6-22(34)7-9-25/h2-9,17,19-20,23-24,26,29,35H,10-16,18H2,1H3,(H,36,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 72 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061276

((R)-1-[(R)-2-(Adamantan-2-yloxycarbonylamino)-3-(1...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)OC1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1CC(C[C@@H]1C(O)=O)c1ccc(Cl)cc1 |TLB:15:16:22:19.24.20,THB:18:17:22:19.24.20,18:19:22:16.17.25| Show InChI InChI=1S/C34H38ClN3O5/c1-34(16-25-17-36-28-5-3-2-4-27(25)28,37-33(42)43-30-22-11-19-10-20(13-22)14-23(30)12-19)32(41)38-18-24(15-29(38)31(39)40)21-6-8-26(35)9-7-21/h2-9,17,19-20,22-24,29-30,36H,10-16,18H2,1H3,(H,37,42)(H,39,40)/t19-,20+,22-,23+,24?,29-,30?,34-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 217 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061267

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(F)cc1F |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H36F2N4O6/c1-33(15-20-16-36-27-5-3-2-4-25(20)27,37-32(43)45-39-22-9-18-8-19(11-22)12-23(39)10-18)31(42)38-17-24(14-28(38)30(40)41)44-29-7-6-21(34)13-26(29)35/h2-7,13,16,18-19,22-24,28,36H,8-12,14-15,17H2,1H3,(H,37,43)(H,40,41)/t18-,19+,22-,23+,24-,28-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 389 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061266

((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(cc1)[N+]([O-])=O |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H37N5O8/c1-33(16-21-17-34-28-5-3-2-4-27(21)28,35-32(42)46-37-23-11-19-10-20(13-23)14-24(37)12-19)31(41)36-18-26(15-29(36)30(39)40)45-25-8-6-22(7-9-25)38(43)44/h2-9,17,19-20,23-24,26,29,34H,10-16,18H2,1H3,(H,35,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 507 | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of inositol phosphate production induced by Cholecystokinin type B receptor (0.5 nM) |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data