 Found 34 hits Enz. Inhib. hit(s) with all data for entry = 50037991
Found 34 hits Enz. Inhib. hit(s) with all data for entry = 50037991 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50225209

(N,N,N-triethyl-2-(4-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H47N5O3/c1-4-39(5-2,6-3)24-25-42-27-21-19-26(20-22-27)34(41)35-23-13-7-8-18-32(40)37-38-33-28-14-9-11-16-30(28)36-31-17-12-10-15-29(31)33/h9,11,14,16,19-22H,4-8,10,12-13,15,17-18,23-25H2,1-3H3,(H2-,35,36,37,38,40,41)/p+1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.467 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50225215
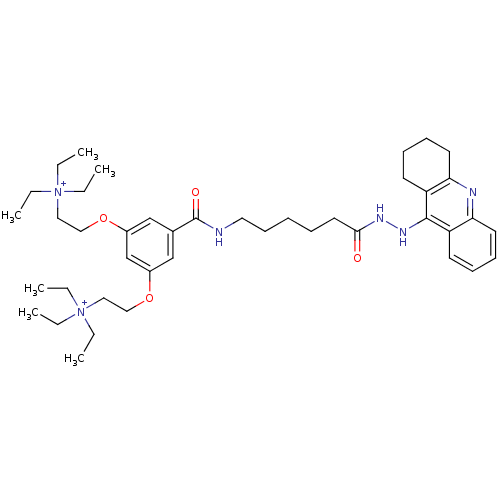
(N,N,N-triethyl-2-(3-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C42H64N6O4/c1-7-47(8-2,9-3)26-28-51-34-30-33(31-35(32-34)52-29-27-48(10-4,11-5)12-6)42(50)43-25-19-13-14-24-40(49)45-46-41-36-20-15-17-22-38(36)44-39-23-18-16-21-37(39)41/h15,17,20,22,30-32H,7-14,16,18-19,21,23-29H2,1-6H3,(H-2,43,44,45,46,49,50)/p+2 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.28 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50371198

(CHEMBL540131)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C34H46ClN5O3/c1-4-40(5-2,6-3)22-23-43-27-18-15-25(16-19-27)34(42)36-21-11-7-8-14-32(41)38-39-33-28-12-9-10-13-30(28)37-31-24-26(35)17-20-29(31)33/h15-20,24H,4-14,21-23H2,1-3H3,(H2-,36,37,38,39,41,42)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.44 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50371197
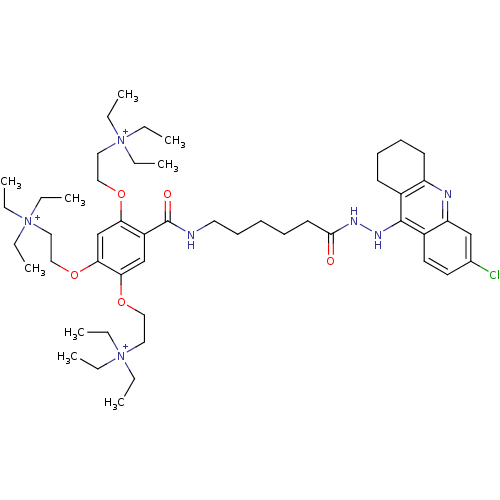
(CHEMBL555247)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C50H81ClN7O5/c1-10-56(11-2,12-3)30-33-61-45-38-47(63-35-32-58(16-7,17-8)18-9)46(62-34-31-57(13-4,14-5)15-6)37-42(45)50(60)52-29-23-19-20-26-48(59)54-55-49-40-24-21-22-25-43(40)53-44-36-39(51)27-28-41(44)49/h27-28,36-38H,10-26,29-35H2,1-9H3,(H-2,52,53,54,55,59,60)/q+1/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.54 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50371199

(CHEMBL540132)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C42H63ClN6O4/c1-7-48(8-2,9-3)24-26-52-34-28-32(29-35(31-34)53-27-25-49(10-4,11-5)12-6)42(51)44-23-17-13-14-20-40(50)46-47-41-36-18-15-16-19-38(36)45-39-30-33(43)21-22-37(39)41/h21-22,28-31H,7-20,23-27H2,1-6H3,(H-2,44,45,46,47,50,51)/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.75 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50225209

(N,N,N-triethyl-2-(4-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H47N5O3/c1-4-39(5-2,6-3)24-25-42-27-21-19-26(20-22-27)34(41)35-23-13-7-8-18-32(40)37-38-33-28-14-9-11-16-30(28)36-31-17-12-10-15-29(31)33/h9,11,14,16,19-22H,4-8,10,12-13,15,17-18,23-25H2,1-3H3,(H2-,35,36,37,38,40,41)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.61 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50225215

(N,N,N-triethyl-2-(3-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C42H64N6O4/c1-7-47(8-2,9-3)26-28-51-34-30-33(31-35(32-34)52-29-27-48(10-4,11-5)12-6)42(50)43-25-19-13-14-24-40(49)45-46-41-36-20-15-17-22-38(36)44-39-23-18-16-21-37(39)41/h15,17,20,22,30-32H,7-14,16,18-19,21,23-29H2,1-6H3,(H-2,43,44,45,46,49,50)/p+2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.59 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Torpedo californica Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50225215

(N,N,N-triethyl-2-(3-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C42H64N6O4/c1-7-47(8-2,9-3)26-28-51-34-30-33(31-35(32-34)52-29-27-48(10-4,11-5)12-6)42(50)43-25-19-13-14-24-40(49)45-46-41-36-20-15-17-22-38(36)44-39-23-18-16-21-37(39)41/h15,17,20,22,30-32H,7-14,16,18-19,21,23-29H2,1-6H3,(H-2,43,44,45,46,49,50)/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15.8 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50225209

(N,N,N-triethyl-2-(4-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H47N5O3/c1-4-39(5-2,6-3)24-25-42-27-21-19-26(20-22-27)34(41)35-23-13-7-8-18-32(40)37-38-33-28-14-9-11-16-30(28)36-31-17-12-10-15-29(31)33/h9,11,14,16,19-22H,4-8,10,12-13,15,17-18,23-25H2,1-3H3,(H2-,35,36,37,38,40,41)/p+1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17.6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Torpedo californica Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50225210
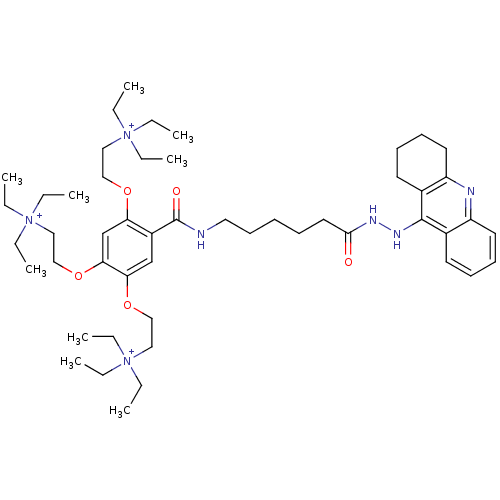
(N,N,N-triethyl-2-(5-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C50H82N7O5/c1-10-55(11-2,12-3)32-35-60-45-39-47(62-37-34-57(16-7,17-8)18-9)46(61-36-33-56(13-4,14-5)15-6)38-42(45)50(59)51-31-25-19-20-30-48(58)53-54-49-40-26-21-23-28-43(40)52-44-29-24-22-27-41(44)49/h21,23,26,28,38-39H,10-20,22,24-25,27,29-37H2,1-9H3,(H-2,51,52,53,54,58,59)/q+1/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23.2 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50225210

(N,N,N-triethyl-2-(5-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C50H82N7O5/c1-10-55(11-2,12-3)32-35-60-45-39-47(62-37-34-57(16-7,17-8)18-9)46(61-36-33-56(13-4,14-5)15-6)38-42(45)50(59)51-31-25-19-20-30-48(58)53-54-49-40-26-21-23-28-43(40)52-44-29-24-22-27-41(44)49/h21,23,26,28,38-39H,10-20,22,24-25,27,29-37H2,1-9H3,(H-2,51,52,53,54,58,59)/q+1/p+2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23.2 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50371198

(CHEMBL540131)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C34H46ClN5O3/c1-4-40(5-2,6-3)22-23-43-27-18-15-25(16-19-27)34(42)36-21-11-7-8-14-32(41)38-39-33-28-12-9-10-13-30(28)37-31-24-26(35)17-20-29(31)33/h15-20,24H,4-14,21-23H2,1-3H3,(H2-,36,37,38,39,41,42)/p+1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50225210

(N,N,N-triethyl-2-(5-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C50H82N7O5/c1-10-55(11-2,12-3)32-35-60-45-39-47(62-37-34-57(16-7,17-8)18-9)46(61-36-33-56(13-4,14-5)15-6)38-42(45)50(59)51-31-25-19-20-30-48(58)53-54-49-40-26-21-23-28-43(40)52-44-29-24-22-27-41(44)49/h21,23,26,28,38-39H,10-20,22,24-25,27,29-37H2,1-9H3,(H-2,51,52,53,54,58,59)/q+1/p+2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27.7 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Torpedo californica Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50225210

(N,N,N-triethyl-2-(5-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C50H82N7O5/c1-10-55(11-2,12-3)32-35-60-45-39-47(62-37-34-57(16-7,17-8)18-9)46(61-36-33-56(13-4,14-5)15-6)38-42(45)50(59)51-31-25-19-20-30-48(58)53-54-49-40-26-21-23-28-43(40)52-44-29-24-22-27-41(44)49/h21,23,26,28,38-39H,10-20,22,24-25,27,29-37H2,1-9H3,(H-2,51,52,53,54,58,59)/q+1/p+2 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28.9 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50225210

(N,N,N-triethyl-2-(5-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C50H82N7O5/c1-10-55(11-2,12-3)32-35-60-45-39-47(62-37-34-57(16-7,17-8)18-9)46(61-36-33-56(13-4,14-5)15-6)38-42(45)50(59)51-31-25-19-20-30-48(58)53-54-49-40-26-21-23-28-43(40)52-44-29-24-22-27-41(44)49/h21,23,26,28,38-39H,10-20,22,24-25,27,29-37H2,1-9H3,(H-2,51,52,53,54,58,59)/q+1/p+2 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28.9 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 42.3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50371199

(CHEMBL540132)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C42H63ClN6O4/c1-7-48(8-2,9-3)24-26-52-34-28-32(29-35(31-34)53-27-25-49(10-4,11-5)12-6)42(51)44-23-17-13-14-20-40(50)46-47-41-36-18-15-16-19-38(36)45-39-30-33(43)21-22-37(39)41/h21-22,28-31H,7-20,23-27H2,1-6H3,(H-2,44,45,46,47,50,51)/p+2 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50371197

(CHEMBL555247)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C50H81ClN7O5/c1-10-56(11-2,12-3)30-33-61-45-38-47(63-35-32-58(16-7,17-8)18-9)46(62-34-31-57(13-4,14-5)15-6)37-42(45)50(60)52-29-23-19-20-26-48(59)54-55-49-40-24-21-22-25-43(40)53-44-36-39(51)27-28-41(44)49/h27-28,36-38H,10-26,29-35H2,1-9H3,(H-2,52,53,54,55,59,60)/q+1/p+2 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50371198

(CHEMBL540131)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C34H46ClN5O3/c1-4-40(5-2,6-3)22-23-43-27-18-15-25(16-19-27)34(42)36-21-11-7-8-14-32(41)38-39-33-28-12-9-10-13-30(28)37-31-24-26(35)17-20-29(31)33/h15-20,24H,4-14,21-23H2,1-3H3,(H2-,36,37,38,39,41,42)/p+1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 127 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Torpedo californica Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 136 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50371199

(CHEMBL540132)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C42H63ClN6O4/c1-7-48(8-2,9-3)24-26-52-34-28-32(29-35(31-34)53-27-25-49(10-4,11-5)12-6)42(51)44-23-17-13-14-20-40(50)46-47-41-36-18-15-16-19-38(36)45-39-30-33(43)21-22-37(39)41/h21-22,28-31H,7-20,23-27H2,1-6H3,(H-2,44,45,46,47,50,51)/p+2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 301 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Torpedo californica Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50371197

(CHEMBL555247)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C50H81ClN7O5/c1-10-56(11-2,12-3)30-33-61-45-38-47(63-35-32-58(16-7,17-8)18-9)46(62-34-31-57(13-4,14-5)15-6)37-42(45)50(60)52-29-23-19-20-26-48(59)54-55-49-40-24-21-22-25-43(40)53-44-36-39(51)27-28-41(44)49/h27-28,36-38H,10-26,29-35H2,1-9H3,(H-2,52,53,54,55,59,60)/q+1/p+2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 334 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Torpedo californica Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 926 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50149891
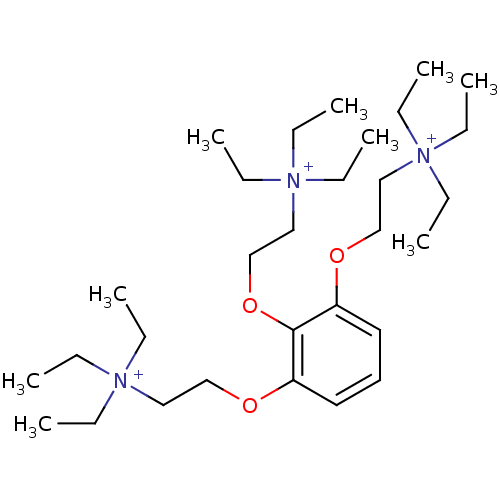
(2,2',2''-[1,2,3-BENZENE-TRIYLTRIS(OXY)]TRIS[N,N,N-...)Show SMILES CC[N+](CC)(CC)CCOc1cccc(OCC[N+](CC)(CC)CC)c1OCC[N+](CC)(CC)CC Show InChI InChI=1S/C30H60N3O3/c1-10-31(11-2,12-3)22-25-34-28-20-19-21-29(35-26-23-32(13-4,14-5)15-6)30(28)36-27-24-33(16-7,17-8)18-9/h19-21H,10-18,22-27H2,1-9H3/q+3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 2.11E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of human Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50149891

(2,2',2''-[1,2,3-BENZENE-TRIYLTRIS(OXY)]TRIS[N,N,N-...)Show SMILES CC[N+](CC)(CC)CCOc1cccc(OCC[N+](CC)(CC)CC)c1OCC[N+](CC)(CC)CC Show InChI InChI=1S/C30H60N3O3/c1-10-31(11-2,12-3)22-25-34-28-20-19-21-29(35-26-23-32(13-4,14-5)15-6)30(28)36-27-24-33(16-7,17-8)18-9/h19-21H,10-18,22-27H2,1-9H3/q+3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.15E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of Electrophorus electricus Acetylcholinesterase |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50225209

(N,N,N-triethyl-2-(4-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H47N5O3/c1-4-39(5-2,6-3)24-25-42-27-21-19-26(20-22-27)34(41)35-23-13-7-8-18-32(40)37-38-33-28-14-9-11-16-30(28)36-31-17-12-10-15-29(31)33/h9,11,14,16,19-22H,4-8,10,12-13,15,17-18,23-25H2,1-3H3,(H2-,35,36,37,38,40,41)/p+1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50371197

(CHEMBL555247)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C50H81ClN7O5/c1-10-56(11-2,12-3)30-33-61-45-38-47(63-35-32-58(16-7,17-8)18-9)46(62-34-31-57(13-4,14-5)15-6)37-42(45)50(60)52-29-23-19-20-26-48(59)54-55-49-40-24-21-22-25-43(40)53-44-36-39(51)27-28-41(44)49/h27-28,36-38H,10-26,29-35H2,1-9H3,(H-2,52,53,54,55,59,60)/q+1/p+2 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50149891

(2,2',2''-[1,2,3-BENZENE-TRIYLTRIS(OXY)]TRIS[N,N,N-...)Show SMILES CC[N+](CC)(CC)CCOc1cccc(OCC[N+](CC)(CC)CC)c1OCC[N+](CC)(CC)CC Show InChI InChI=1S/C30H60N3O3/c1-10-31(11-2,12-3)22-25-34-28-20-19-21-29(35-26-23-32(13-4,14-5)15-6)30(28)36-27-24-33(16-7,17-8)18-9/h19-21H,10-18,22-27H2,1-9H3/q+3 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 130 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50225215

(N,N,N-triethyl-2-(3-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C42H64N6O4/c1-7-47(8-2,9-3)26-28-51-34-30-33(31-35(32-34)52-29-27-48(10-4,11-5)12-6)42(50)43-25-19-13-14-24-40(49)45-46-41-36-20-15-17-22-38(36)44-39-23-18-16-21-37(39)41/h15,17,20,22,30-32H,7-14,16,18-19,21,23-29H2,1-6H3,(H-2,43,44,45,46,49,50)/p+2 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50371199

(CHEMBL540132)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)cc(c1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C42H63ClN6O4/c1-7-48(8-2,9-3)24-26-52-34-28-32(29-35(31-34)53-27-25-49(10-4,11-5)12-6)42(51)44-23-17-13-14-20-40(50)46-47-41-36-18-15-16-19-38(36)45-39-30-33(43)21-22-37(39)41/h21-22,28-31H,7-20,23-27H2,1-6H3,(H-2,44,45,46,47,50,51)/p+2 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.30E+3 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50371198

(CHEMBL540131)Show SMILES CC[N+](CC)(CC)CCOc1ccc(cc1)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2cc(Cl)ccc12 Show InChI InChI=1S/C34H46ClN5O3/c1-4-40(5-2,6-3)22-23-43-27-18-15-25(16-19-27)34(42)36-21-11-7-8-14-32(41)38-39-33-28-12-9-10-13-30(28)37-31-24-26(35)17-20-29(31)33/h15-20,24H,4-14,21-23H2,1-3H3,(H2-,36,37,38,39,41,42)/p+1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(GUINEA PIG) | BDBM50225210

(N,N,N-triethyl-2-(5-(((6-(2-(1,2,3,4-tetrahydroacr...)Show SMILES CC[N+](CC)(CC)CCOc1cc(OCC[N+](CC)(CC)CC)c(cc1OCC[N+](CC)(CC)CC)C(=O)NCCCCCC(=O)NNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C50H82N7O5/c1-10-55(11-2,12-3)32-35-60-45-39-47(62-37-34-57(16-7,17-8)18-9)46(61-36-33-56(13-4,14-5)15-6)38-42(45)50(59)51-31-25-19-20-30-48(58)53-54-49-40-26-21-23-28-43(40)52-44-29-24-22-27-41(44)49/h21,23,26,28,38-39H,10-20,22,24-25,27,29-37H2,1-9H3,(H-2,51,52,53,54,58,59)/q+1/p+2 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Inhibition of [3H]NMS dissociation from porcine muscarinic M2 receptor |
J Med Chem 50: 5685-95 (2007)
Article DOI: 10.1021/jm070859s
BindingDB Entry DOI: 10.7270/Q2NS0VRN |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data