 Found 96 hits Enz. Inhib. hit(s) with all data for entry = 50040971
Found 96 hits Enz. Inhib. hit(s) with all data for entry = 50040971 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50046145
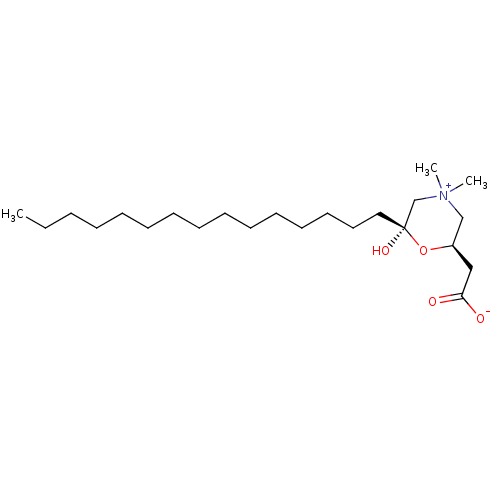
(2-[6-hydroxy-4,4-dimethyl-6-pentadecyl-(2R,6S)-1,4...)Show SMILES CCCCCCCCCCCCCCC[C@@]1(O)C[N+](C)(C)C[C@@H](CC([O-])=O)O1 Show InChI InChI=1S/C23H45NO4/c1-4-5-6-7-8-9-10-11-12-13-14-15-16-17-23(27)20-24(2,3)19-21(28-23)18-22(25)26/h21,27H,4-20H2,1-3H3/t21-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Competitive inhibition of CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50046145

(2-[6-hydroxy-4,4-dimethyl-6-pentadecyl-(2R,6S)-1,4...)Show SMILES CCCCCCCCCCCCCCC[C@@]1(O)C[N+](C)(C)C[C@@H](CC([O-])=O)O1 Show InChI InChI=1S/C23H45NO4/c1-4-5-6-7-8-9-10-11-12-13-14-15-16-17-23(27)20-24(2,3)19-21(28-23)18-22(25)26/h21,27H,4-20H2,1-3H3/t21-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Competitive inhibition of CPT2 in heart mitochondria using carnitine |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50046145

(2-[6-hydroxy-4,4-dimethyl-6-pentadecyl-(2R,6S)-1,4...)Show SMILES CCCCCCCCCCCCCCC[C@@]1(O)C[N+](C)(C)C[C@@H](CC([O-])=O)O1 Show InChI InChI=1S/C23H45NO4/c1-4-5-6-7-8-9-10-11-12-13-14-15-16-17-23(27)20-24(2,3)19-21(28-23)18-22(25)26/h21,27H,4-20H2,1-3H3/t21-,23+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Competitive inhibition of CPT2 in liver mitochondria using carnitine |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403039
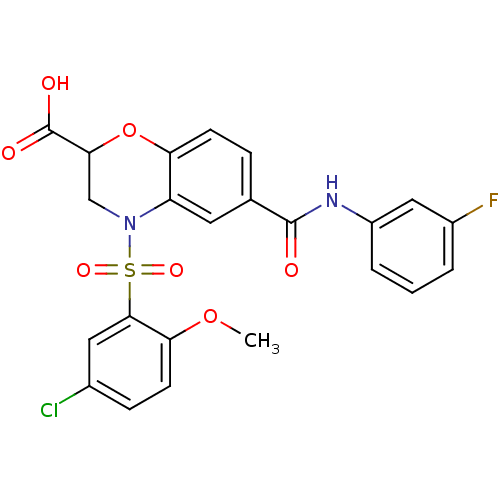
(CHEMBL2216778)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CC(Oc2ccc(cc12)C(=O)Nc1cccc(F)c1)C(O)=O Show InChI InChI=1S/C23H18ClFN2O7S/c1-33-19-8-6-14(24)10-21(19)35(31,32)27-12-20(23(29)30)34-18-7-5-13(9-17(18)27)22(28)26-16-4-2-3-15(25)11-16/h2-11,20H,12H2,1H3,(H,26,28)(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403037
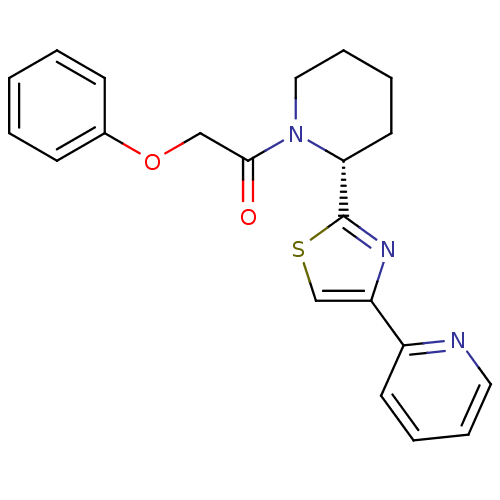
(CHEMBL2216774)Show SMILES O=C(COc1ccccc1)N1CCCC[C@@H]1c1nc(cs1)-c1ccccn1 |r| Show InChI InChI=1S/C21H21N3O2S/c25-20(14-26-16-8-2-1-3-9-16)24-13-7-5-11-19(24)21-23-18(15-27-21)17-10-4-6-12-22-17/h1-4,6,8-10,12,15,19H,5,7,11,13-14H2/t19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403040
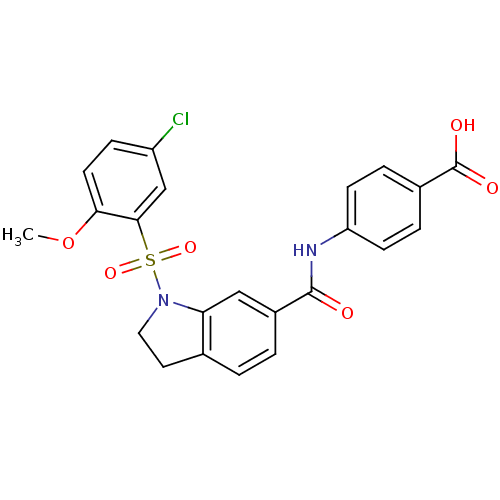
(CHEMBL2216779)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CCc2ccc(cc12)C(=O)Nc1ccc(cc1)C(O)=O Show InChI InChI=1S/C23H19ClN2O6S/c1-32-20-9-6-17(24)13-21(20)33(30,31)26-11-10-14-2-3-16(12-19(14)26)22(27)25-18-7-4-15(5-8-18)23(28)29/h2-9,12-13H,10-11H2,1H3,(H,25,27)(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403041
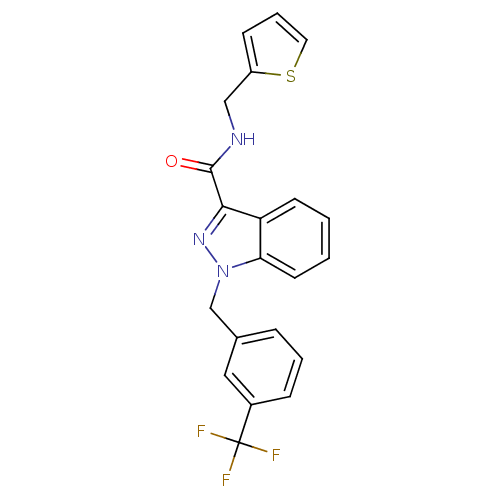
(CHEMBL2216776)Show SMILES FC(F)(F)c1cccc(Cn2nc(C(=O)NCc3cccs3)c3ccccc23)c1 Show InChI InChI=1S/C21H16F3N3OS/c22-21(23,24)15-6-3-5-14(11-15)13-27-18-9-2-1-8-17(18)19(26-27)20(28)25-12-16-7-4-10-29-16/h1-11H,12-13H2,(H,25,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403039

(CHEMBL2216778)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CC(Oc2ccc(cc12)C(=O)Nc1cccc(F)c1)C(O)=O Show InChI InChI=1S/C23H18ClFN2O7S/c1-33-19-8-6-14(24)10-21(19)35(31,32)27-12-20(23(29)30)34-18-7-5-13(9-17(18)27)22(28)26-16-4-2-3-15(25)11-16/h2-11,20H,12H2,1H3,(H,26,28)(H,29,30) | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403029
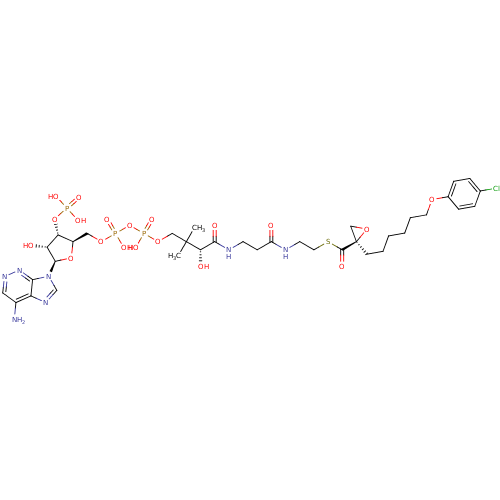
(CHEMBL2216792)Show SMILES CC(C)(COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)n1cnc2c(N)cnnc12)[C@@H](O)C(=O)NCCC(=O)NCCSC(=O)[C@@]1(CCCCCCOc2ccc(Cl)cc2)CO1 |r| Show InChI InChI=1S/C36H53ClN7O19P3S/c1-35(2,30(47)32(48)40-13-11-26(45)39-14-16-67-34(49)36(20-58-36)12-5-3-4-6-15-57-23-9-7-22(37)8-10-23)19-60-66(55,56)63-65(53,54)59-18-25-29(62-64(50,51)52)28(46)33(61-25)44-21-41-27-24(38)17-42-43-31(27)44/h7-10,17,21,25,28-30,33,46-47H,3-6,11-16,18-20H2,1-2H3,(H2,38,43)(H,39,45)(H,40,48)(H,53,54)(H,55,56)(H2,50,51,52)/t25-,28-,29-,30+,33-,36-/m1/s1 | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT1B expressed in intact Saccharomyces cerevisiae using [methyl-3H]carnitine by radiometric assay |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403040

(CHEMBL2216779)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CCc2ccc(cc12)C(=O)Nc1ccc(cc1)C(O)=O Show InChI InChI=1S/C23H19ClN2O6S/c1-32-20-9-6-17(24)13-21(20)33(30,31)26-11-10-14-2-3-16(12-19(14)26)22(27)25-18-7-4-15(5-8-18)23(28)29/h2-9,12-13H,10-11H2,1H3,(H,25,27)(H,28,29) | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403042
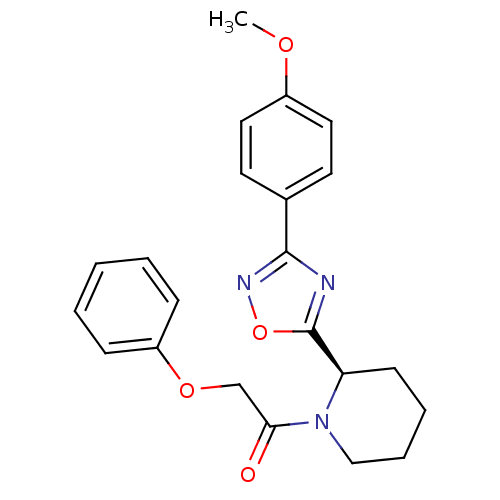
(CHEMBL2216775)Show SMILES COc1ccc(cc1)-c1noc(n1)[C@H]1CCCCN1C(=O)COc1ccccc1 |r| Show InChI InChI=1S/C22H23N3O4/c1-27-17-12-10-16(11-13-17)21-23-22(29-24-21)19-9-5-6-14-25(19)20(26)15-28-18-7-3-2-4-8-18/h2-4,7-8,10-13,19H,5-6,9,14-15H2,1H3/t19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403037

(CHEMBL2216774)Show SMILES O=C(COc1ccccc1)N1CCCC[C@@H]1c1nc(cs1)-c1ccccn1 |r| Show InChI InChI=1S/C21H21N3O2S/c25-20(14-26-16-8-2-1-3-9-16)24-13-7-5-11-19(24)21-23-18(15-27-21)17-10-4-6-12-22-17/h1-4,6,8-10,12,15,19H,5,7,11,13-14H2/t19-/m1/s1 | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403033
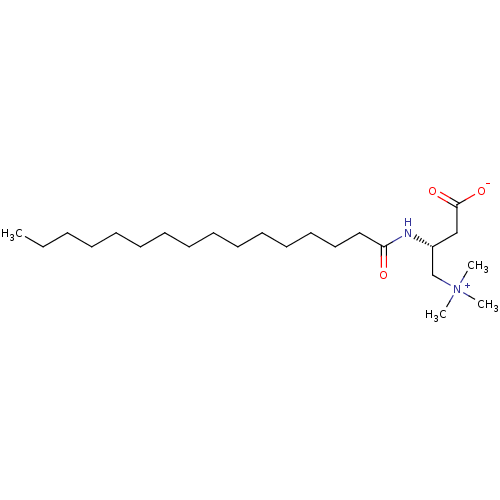
(CHEMBL1231507)Show SMILES CCCCCCCCCCCCCCCC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C |r| Show InChI InChI=1S/C23H46N2O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-18-22(26)24-21(19-23(27)28)20-25(2,3)4/h21H,5-20H2,1-4H3,(H-,24,26,27,28)/t21-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403041

(CHEMBL2216776)Show SMILES FC(F)(F)c1cccc(Cn2nc(C(=O)NCc3cccs3)c3ccccc23)c1 Show InChI InChI=1S/C21H16F3N3OS/c22-21(23,24)15-6-3-5-14(11-15)13-27-18-9-2-1-8-17(18)19(26-27)20(28)25-12-16-7-4-10-29-16/h1-11H,12-13H2,(H,25,28) | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403038
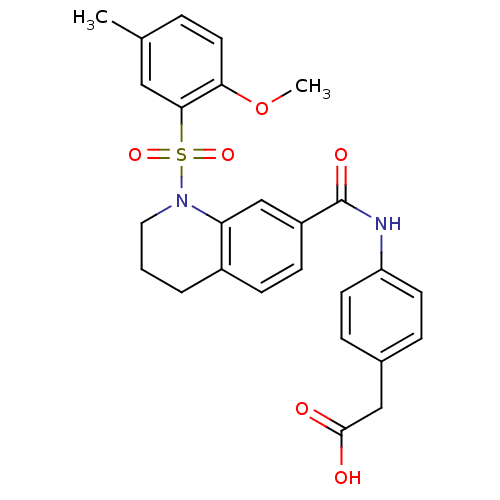
(CHEMBL2216777)Show SMILES COc1ccc(C)cc1S(=O)(=O)N1CCCc2ccc(cc12)C(=O)Nc1ccc(CC(O)=O)cc1 Show InChI InChI=1S/C26H26N2O6S/c1-17-5-12-23(34-2)24(14-17)35(32,33)28-13-3-4-19-8-9-20(16-22(19)28)26(31)27-21-10-6-18(7-11-21)15-25(29)30/h5-12,14,16H,3-4,13,15H2,1-2H3,(H,27,31)(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50403031
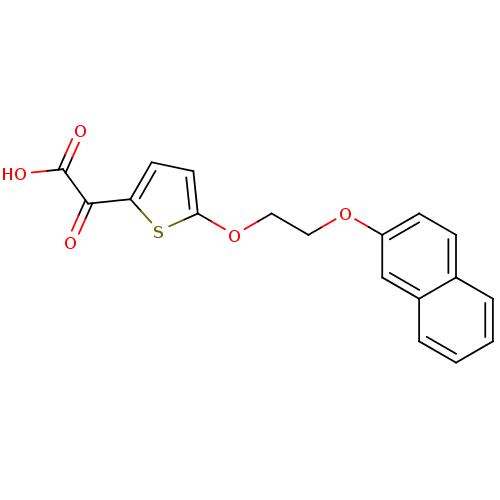
(CHEMBL2216771)Show InChI InChI=1S/C18H14O5S/c19-17(18(20)21)15-7-8-16(24-15)23-10-9-22-14-6-5-12-3-1-2-4-13(12)11-14/h1-8,11H,9-10H2,(H,20,21) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50122839
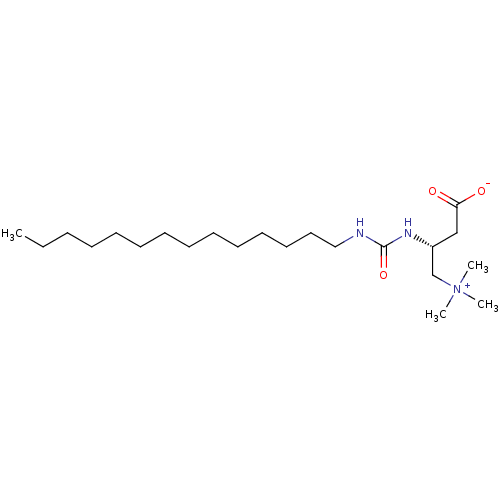
(CHEMBL106227 | [3-Carboxy-2-(3-tetradecyl-ureido)-...)Show SMILES CCCCCCCCCCCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C Show InChI InChI=1S/C22H45N3O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-23-22(28)24-20(18-21(26)27)19-25(2,3)4/h20H,5-19H2,1-4H3,(H2-,23,24,26,27,28)/t20-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403038

(CHEMBL2216777)Show SMILES COc1ccc(C)cc1S(=O)(=O)N1CCCc2ccc(cc12)C(=O)Nc1ccc(CC(O)=O)cc1 Show InChI InChI=1S/C26H26N2O6S/c1-17-5-12-23(34-2)24(14-17)35(32,33)28-13-3-4-19-8-9-20(16-22(19)28)26(31)27-21-10-6-18(7-11-21)15-25(29)30/h5-12,14,16H,3-4,13,15H2,1-2H3,(H,27,31)(H,29,30) | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403043
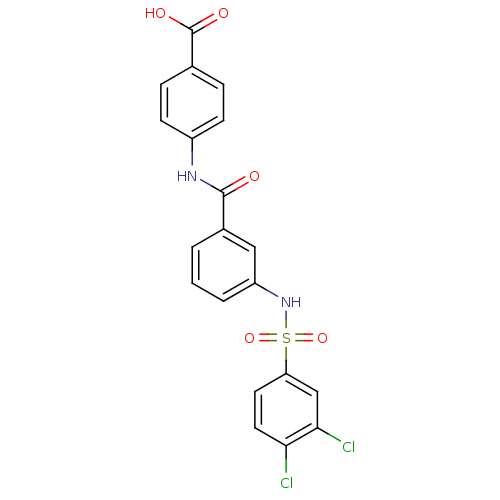
(CHEMBL2216780)Show SMILES OC(=O)c1ccc(NC(=O)c2cccc(NS(=O)(=O)c3ccc(Cl)c(Cl)c3)c2)cc1 Show InChI InChI=1S/C20H14Cl2N2O5S/c21-17-9-8-16(11-18(17)22)30(28,29)24-15-3-1-2-13(10-15)19(25)23-14-6-4-12(5-7-14)20(26)27/h1-11,24H,(H,23,25)(H,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403028
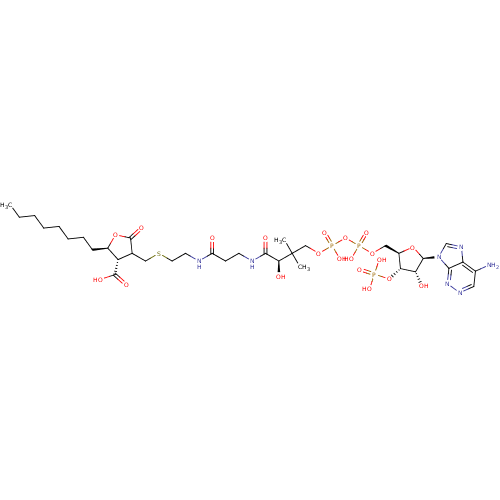
(CHEMBL2216769)Show SMILES CCCCCCCC[C@H]1OC(=O)C(CSCCNC(=O)CCNC(=O)[C@H](O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2OP(O)(O)=O)n2cnc3c(N)cnnc23)[C@@H]1C(O)=O |r| Show InChI InChI=1S/C35H58N7O20P3S/c1-4-5-6-7-8-9-10-22-25(33(47)48)20(34(49)60-22)17-66-14-13-37-24(43)11-12-38-31(46)29(45)35(2,3)18-58-65(55,56)62-64(53,54)57-16-23-28(61-63(50,51)52)27(44)32(59-23)42-19-39-26-21(36)15-40-41-30(26)42/h15,19-20,22-23,25,27-29,32,44-45H,4-14,16-18H2,1-3H3,(H2,36,41)(H,37,43)(H,38,46)(H,47,48)(H,53,54)(H,55,56)(H2,50,51,52)/t20?,22-,23-,25+,27-,28-,29+,32-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403030
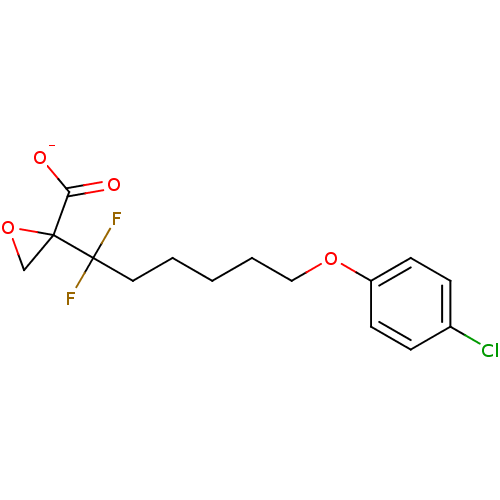
(CHEMBL2216768)Show InChI InChI=1S/C15H17ClF2O4/c16-11-4-6-12(7-5-11)21-9-3-1-2-8-15(17,18)14(10-22-14)13(19)20/h4-7H,1-3,8-10H2,(H,19,20)/p-1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50403028

(CHEMBL2216769)Show SMILES CCCCCCCC[C@H]1OC(=O)C(CSCCNC(=O)CCNC(=O)[C@H](O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2OP(O)(O)=O)n2cnc3c(N)cnnc23)[C@@H]1C(O)=O |r| Show InChI InChI=1S/C35H58N7O20P3S/c1-4-5-6-7-8-9-10-22-25(33(47)48)20(34(49)60-22)17-66-14-13-37-24(43)11-12-38-31(46)29(45)35(2,3)18-58-65(55,56)62-64(53,54)57-16-23-28(61-63(50,51)52)27(44)32(59-23)42-19-39-26-21(36)15-40-41-30(26)42/h15,19-20,22-23,25,27-29,32,44-45H,4-14,16-18H2,1-3H3,(H2,36,41)(H,37,43)(H,38,46)(H,47,48)(H,53,54)(H,55,56)(H2,50,51,52)/t20?,22-,23-,25+,27-,28-,29+,32-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 360 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Rattus norvegicus) | BDBM50122839

(CHEMBL106227 | [3-Carboxy-2-(3-tetradecyl-ureido)-...)Show SMILES CCCCCCCCCCCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C Show InChI InChI=1S/C22H45N3O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-23-22(28)24-20(18-21(26)27)19-25(2,3)4/h20H,5-19H2,1-4H3,(H2-,23,24,26,27,28)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 380 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carnitine O-palmitoyltransferase 1, liver isoform
(Rattus norvegicus) | BDBM50403029

(CHEMBL2216792)Show SMILES CC(C)(COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)n1cnc2c(N)cnnc12)[C@@H](O)C(=O)NCCC(=O)NCCSC(=O)[C@@]1(CCCCCCOc2ccc(Cl)cc2)CO1 |r| Show InChI InChI=1S/C36H53ClN7O19P3S/c1-35(2,30(47)32(48)40-13-11-26(45)39-14-16-67-34(49)36(20-58-36)12-5-3-4-6-15-57-23-9-7-22(37)8-10-23)19-60-66(55,56)63-65(53,54)59-18-25-29(62-64(50,51)52)28(46)33(61-25)44-21-41-27-24(38)17-42-43-31(27)44/h7-10,17,21,25,28-30,33,46-47H,3-6,11-16,18-20H2,1-2H3,(H2,38,43)(H,39,45)(H,40,48)(H,53,54)(H,55,56)(H2,50,51,52)/t25-,28-,29-,30+,33-,36-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT1A expressed in intact Saccharomyces cerevisiae using [methyl-3H]carnitine by radiometric assay |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403040

(CHEMBL2216779)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CCc2ccc(cc12)C(=O)Nc1ccc(cc1)C(O)=O Show InChI InChI=1S/C23H19ClN2O6S/c1-32-20-9-6-17(24)13-21(20)33(30,31)26-11-10-14-2-3-16(12-19(14)26)22(27)25-18-7-4-15(5-8-18)23(28)29/h2-9,12-13H,10-11H2,1H3,(H,25,27)(H,28,29) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 740 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Rattus norvegicus) | BDBM50403034
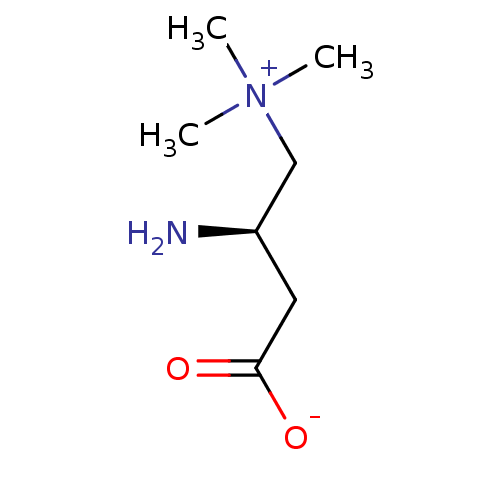
(CHEMBL2114397)Show InChI InChI=1S/C7H16N2O2/c1-9(2,3)5-6(8)4-7(10)11/h6H,4-5,8H2,1-3H3/t6-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CPT2 in 24 hrs fasted rat liver mitochondria assessed as palmitoylcarnitine oxidation by radiometry |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50122839

(CHEMBL106227 | [3-Carboxy-2-(3-tetradecyl-ureido)-...)Show SMILES CCCCCCCCCCCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C Show InChI InChI=1S/C22H45N3O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-23-22(28)24-20(18-21(26)27)19-25(2,3)4/h20H,5-19H2,1-4H3,(H2-,23,24,26,27,28)/t20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM80613
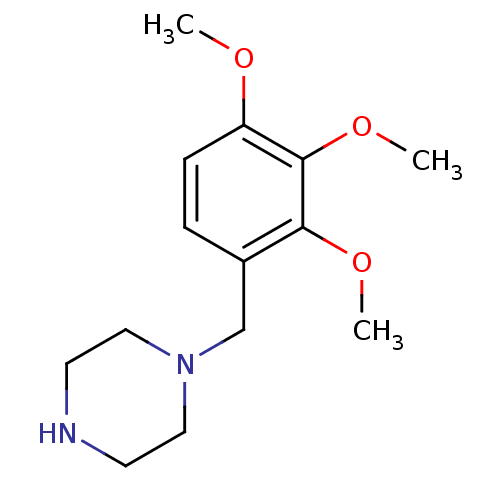
(1-(2,3,4-Trimethoxy-benzyl)-piperazine | 1-(2,3,4-...)Show InChI InChI=1S/C14H22N2O3/c1-17-12-5-4-11(13(18-2)14(12)19-3)10-16-8-6-15-7-9-16/h4-5,15H,6-10H2,1-3H3 | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CPT1A in rat myocardium |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50122839

(CHEMBL106227 | [3-Carboxy-2-(3-tetradecyl-ureido)-...)Show SMILES CCCCCCCCCCCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C Show InChI InChI=1S/C22H45N3O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-23-22(28)24-20(18-21(26)27)19-25(2,3)4/h20H,5-19H2,1-4H3,(H2-,23,24,26,27,28)/t20-/m1/s1 | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403032
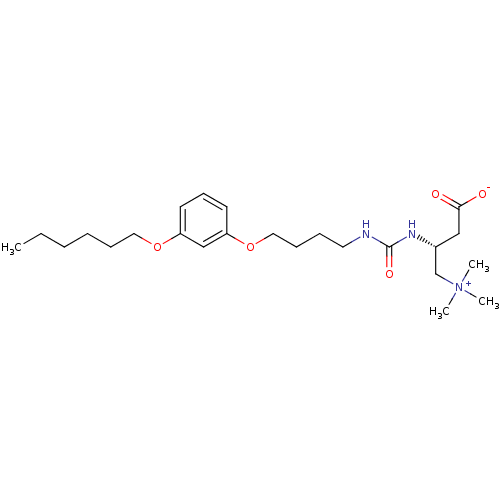
(CHEMBL2216785)Show SMILES CCCCCCOc1cccc(OCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C)c1 |r| Show InChI InChI=1S/C24H41N3O5/c1-5-6-7-9-15-31-21-12-11-13-22(18-21)32-16-10-8-14-25-24(30)26-20(17-23(28)29)19-27(2,3)4/h11-13,18,20H,5-10,14-17,19H2,1-4H3,(H2-,25,26,28,29,30)/t20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403031

(CHEMBL2216771)Show InChI InChI=1S/C18H14O5S/c19-17(18(20)21)15-7-8-16(24-15)23-10-9-22-14-6-5-12-3-1-2-4-13(12)11-14/h1-8,11H,9-10H2,(H,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Homo sapiens (Human)) | BDBM50403033

(CHEMBL1231507)Show SMILES CCCCCCCCCCCCCCCC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C |r| Show InChI InChI=1S/C23H46N2O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-18-22(26)24-21(19-23(27)28)20-25(2,3)4/h21H,5-20H2,1-4H3,(H-,24,26,27,28)/t21-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403038

(CHEMBL2216777)Show SMILES COc1ccc(C)cc1S(=O)(=O)N1CCCc2ccc(cc12)C(=O)Nc1ccc(CC(O)=O)cc1 Show InChI InChI=1S/C26H26N2O6S/c1-17-5-12-23(34-2)24(14-17)35(32,33)28-13-3-4-19-8-9-20(16-22(19)28)26(31)27-21-10-6-18(7-11-21)15-25(29)30/h5-12,14,16H,3-4,13,15H2,1-2H3,(H,27,31)(H,29,30) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Rattus norvegicus) | BDBM50403040

(CHEMBL2216779)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CCc2ccc(cc12)C(=O)Nc1ccc(cc1)C(O)=O Show InChI InChI=1S/C23H19ClN2O6S/c1-32-20-9-6-17(24)13-21(20)33(30,31)26-11-10-14-2-3-16(12-19(14)26)22(27)25-18-7-4-15(5-8-18)23(28)29/h2-9,12-13H,10-11H2,1H3,(H,25,27)(H,28,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50403039

(CHEMBL2216778)Show SMILES COc1ccc(Cl)cc1S(=O)(=O)N1CC(Oc2ccc(cc12)C(=O)Nc1cccc(F)c1)C(O)=O Show InChI InChI=1S/C23H18ClFN2O7S/c1-33-19-8-6-14(24)10-21(19)35(31,32)27-12-20(23(29)30)34-18-7-5-13(9-17(18)27)22(28)26-16-4-2-3-15(25)11-16/h2-11,20H,12H2,1H3,(H,26,28)(H,29,30) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50403038

(CHEMBL2216777)Show SMILES COc1ccc(C)cc1S(=O)(=O)N1CCCc2ccc(cc12)C(=O)Nc1ccc(CC(O)=O)cc1 Show InChI InChI=1S/C26H26N2O6S/c1-17-5-12-23(34-2)24(14-17)35(32,33)28-13-3-4-19-8-9-20(16-22(19)28)26(31)27-21-10-6-18(7-11-21)15-25(29)30/h5-12,14,16H,3-4,13,15H2,1-2H3,(H,27,31)(H,29,30) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403034

(CHEMBL2114397)Show InChI InChI=1S/C7H16N2O2/c1-9(2,3)5-6(8)4-7(10)11/h6H,4-5,8H2,1-3H3/t6-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403031

(CHEMBL2216771)Show InChI InChI=1S/C18H14O5S/c19-17(18(20)21)15-7-8-16(24-15)23-10-9-22-14-6-5-12-3-1-2-4-13(12)11-14/h1-8,11H,9-10H2,(H,20,21) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403037

(CHEMBL2216774)Show SMILES O=C(COc1ccccc1)N1CCCC[C@@H]1c1nc(cs1)-c1ccccn1 |r| Show InChI InChI=1S/C21H21N3O2S/c25-20(14-26-16-8-2-1-3-9-16)24-13-7-5-11-19(24)21-23-18(15-27-21)17-10-4-6-12-22-17/h1-4,6,8-10,12,15,19H,5,7,11,13-14H2/t19-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Mitochondrial carnitine/acylcarnitine carrier protein
(Homo sapiens (Human)) | BDBM50403026
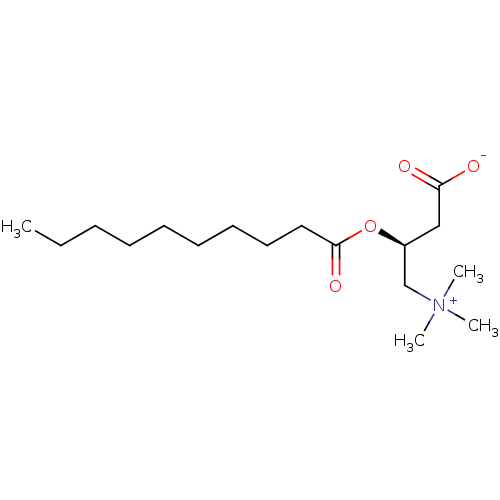
(CHEMBL2216788)Show SMILES CCCCCCCCCC(=O)O[C@@H](CC([O-])=O)C[N+](C)(C)C |r| Show InChI InChI=1S/C17H33NO4/c1-5-6-7-8-9-10-11-12-17(21)22-15(13-16(19)20)14-18(2,3)4/h15H,5-14H2,1-4H3/t15-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CACT |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Rattus norvegicus) | BDBM50403034

(CHEMBL2114397)Show InChI InChI=1S/C7H16N2O2/c1-9(2,3)5-6(8)4-7(10)11/h6H,4-5,8H2,1-3H3/t6-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Homo sapiens (Human)) | BDBM50403032

(CHEMBL2216785)Show SMILES CCCCCCOc1cccc(OCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C)c1 |r| Show InChI InChI=1S/C24H41N3O5/c1-5-6-7-9-15-31-21-12-11-13-22(18-21)32-16-10-8-14-25-24(30)26-20(17-23(28)29)19-27(2,3)4/h11-13,18,20H,5-10,14-17,19H2,1-4H3,(H2-,25,26,28,29,30)/t20-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50122839

(CHEMBL106227 | [3-Carboxy-2-(3-tetradecyl-ureido)-...)Show SMILES CCCCCCCCCCCCCCNC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C Show InChI InChI=1S/C22H45N3O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-23-22(28)24-20(18-21(26)27)19-25(2,3)4/h20H,5-19H2,1-4H3,(H2-,23,24,26,27,28)/t20-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50403043

(CHEMBL2216780)Show SMILES OC(=O)c1ccc(NC(=O)c2cccc(NS(=O)(=O)c3ccc(Cl)c(Cl)c3)c2)cc1 Show InChI InChI=1S/C20H14Cl2N2O5S/c21-17-9-8-16(11-18(17)22)30(28,29)24-15-3-1-2-13(10-15)19(25)23-14-6-4-12(5-7-14)20(26)27/h1-11,24H,(H,23,25)(H,26,27) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, liver isoform
(Rattus norvegicus) | BDBM50403046
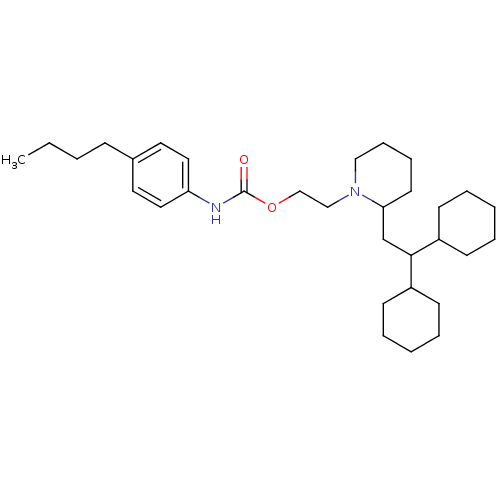
(CHEMBL2216772)Show SMILES CCCCc1ccc(NC(=O)OCCN2CCCCC2CC(C2CCCCC2)C2CCCCC2)cc1 Show InChI InChI=1S/C32H52N2O2/c1-2-3-12-26-18-20-29(21-19-26)33-32(35)36-24-23-34-22-11-10-17-30(34)25-31(27-13-6-4-7-14-27)28-15-8-5-9-16-28/h18-21,27-28,30-31H,2-17,22-25H2,1H3,(H,33,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Rattus norvegicus) | BDBM50403046

(CHEMBL2216772)Show SMILES CCCCc1ccc(NC(=O)OCCN2CCCCC2CC(C2CCCCC2)C2CCCCC2)cc1 Show InChI InChI=1S/C32H52N2O2/c1-2-3-12-26-18-20-29(21-19-26)33-32(35)36-24-23-34-22-11-10-17-30(34)25-31(27-13-6-4-7-14-27)28-15-8-5-9-16-28/h18-21,27-28,30-31H,2-17,22-25H2,1H3,(H,33,35) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50118093
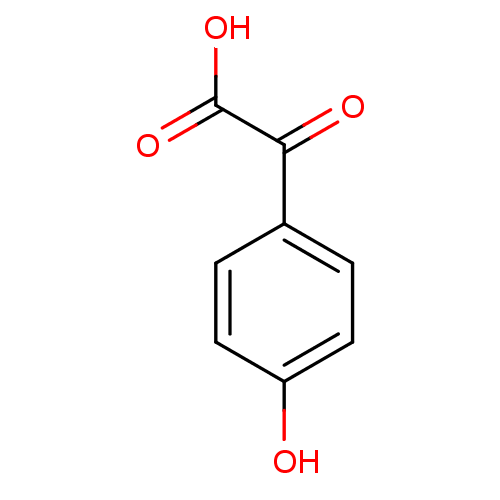
((4-Hydroxy-phenyl)-oxo-acetic acid | CHEMBL129918)Show InChI InChI=1S/C8H6O4/c9-6-3-1-5(2-4-6)7(10)8(11)12/h1-4,9H,(H,11,12) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of CPT1B in heart mitochondria |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 2, mitochondrial
(Rattus norvegicus) | BDBM50403038

(CHEMBL2216777)Show SMILES COc1ccc(C)cc1S(=O)(=O)N1CCCc2ccc(cc12)C(=O)Nc1ccc(CC(O)=O)cc1 Show InChI InChI=1S/C26H26N2O6S/c1-17-5-12-23(34-2)24(14-17)35(32,33)28-13-3-4-19-8-9-20(16-22(19)28)26(31)27-21-10-6-18(7-11-21)15-25(29)30/h5-12,14,16H,3-4,13,15H2,1-2H3,(H,27,31)(H,29,30) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.67E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of rat CPT2 |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Homo sapiens (Human)) | BDBM50403033

(CHEMBL1231507)Show SMILES CCCCCCCCCCCCCCCC(=O)N[C@H](CC([O-])=O)C[N+](C)(C)C |r| Show InChI InChI=1S/C23H46N2O3/c1-5-6-7-8-9-10-11-12-13-14-15-16-17-18-22(26)24-21(19-23(27)28)20-25(2,3)4/h21H,5-20H2,1-4H3,(H-,24,26,27,28)/t21-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.17E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human CPT1B |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |
Carnitine O-palmitoyltransferase 1, muscle isoform
(Rattus norvegicus) | BDBM50403034

(CHEMBL2114397)Show InChI InChI=1S/C7H16N2O2/c1-9(2,3)5-6(8)4-7(10)11/h6H,4-5,8H2,1-3H3/t6-/m1/s1 | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.73E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
F. Hoffmann-La Roche Ltd.
Curated by ChEMBL
| Assay Description
Maximal inhibition of rat CPT1A |
J Med Chem 54: 3109-52 (2011)
Article DOI: 10.1021/jm100809g
BindingDB Entry DOI: 10.7270/Q29K4CDQ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data