

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Muscarinic acetylcholine receptor M2 (Mus musculus) | BDBM50403547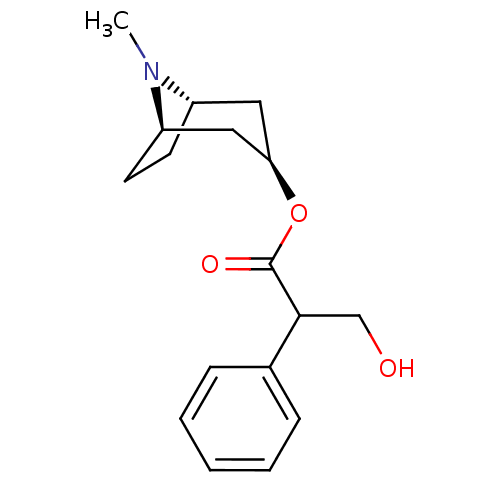 (ATROPEN | ATROPINE) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Similars | PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Carolina Curated by ChEMBL | Assay Description Ability of the compound to displace [3H]-methylscopolamine ([3H]-NMS) from mouse cerebral cortex | J Med Chem 35: 3141-7 (1992) BindingDB Entry DOI: 10.7270/Q200012X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Mus musculus) | BDBM50004665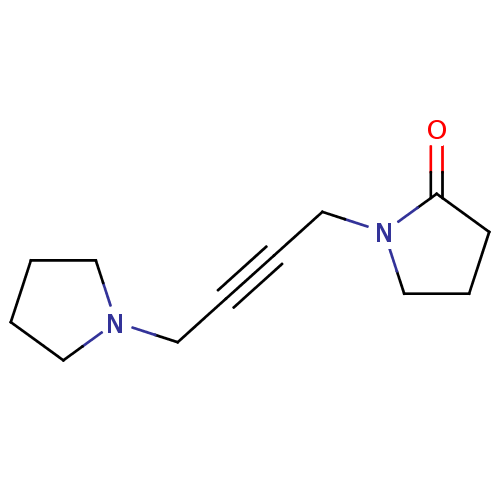 ((oxotremorine)1-(4-Pyrrolidin-1-yl-but-2-ynyl)-pyr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | PubMed | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Carolina Curated by ChEMBL | Assay Description Ability of the compound to displace [3H]-methylscopolamine ([3H]-NMS) from mouse cerebral cortex | J Med Chem 35: 3141-7 (1992) BindingDB Entry DOI: 10.7270/Q200012X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Mus musculus) | BDBM50004664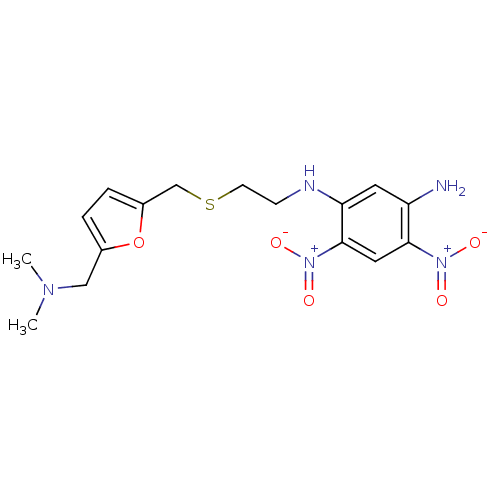 (CHEMBL107892 | N-[2-(5-Dimethylaminomethyl-furan-2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Carolina Curated by ChEMBL | Assay Description Ability of the compound to displace [3H]-methylscopolamine ([3H]-NMS) from mouse cerebral cortex | J Med Chem 35: 3141-7 (1992) BindingDB Entry DOI: 10.7270/Q200012X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Mus musculus) | BDBM50004652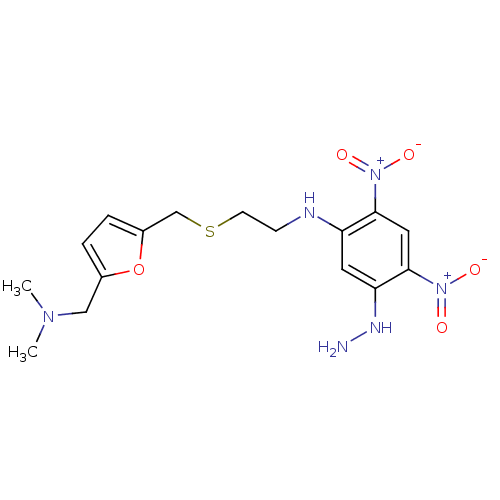 (CHEMBL321605 | [2-(5-Dimethylaminomethyl-furan-2-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Carolina Curated by ChEMBL | Assay Description Ability of the compound to displace [3H]-methylscopolamine ([3H]-NMS) from mouse cerebral cortex | J Med Chem 35: 3141-7 (1992) BindingDB Entry DOI: 10.7270/Q200012X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (Mus musculus) | BDBM50004661 (CHEMBL316973 | [2-(5-Dimethylaminomethyl-furan-2-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Carolina Curated by ChEMBL | Assay Description Ability of the compound to displace [3H]-oxotremorine ([3H]-OXO-M) from mouse cerebral cortex | J Med Chem 35: 3141-7 (1992) BindingDB Entry DOI: 10.7270/Q200012X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||