

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Procathepsin L (Homo sapiens (Human)) | BDBM19855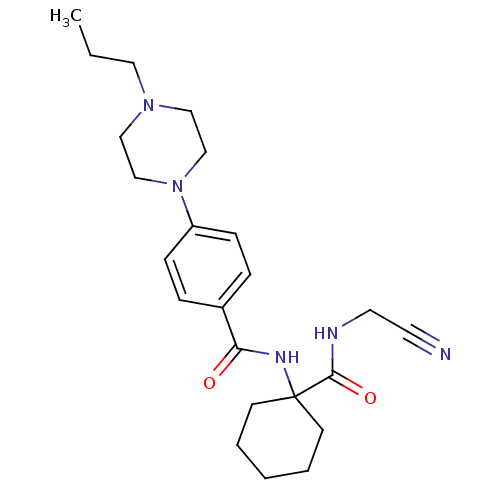 (Balicatib | CHEMBL371064 | N-[1-(cyanomethylcarbam...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 503 | n/a | 48 | n/a | n/a | 5.5 | 22 |
Merck Frosst Centre for Therapeutic Research | Assay Description Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ... | J Med Chem 48: 7535-43 (2005) Article DOI: 10.1021/jm0504961 BindingDB Entry DOI: 10.7270/Q28S4N63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM23866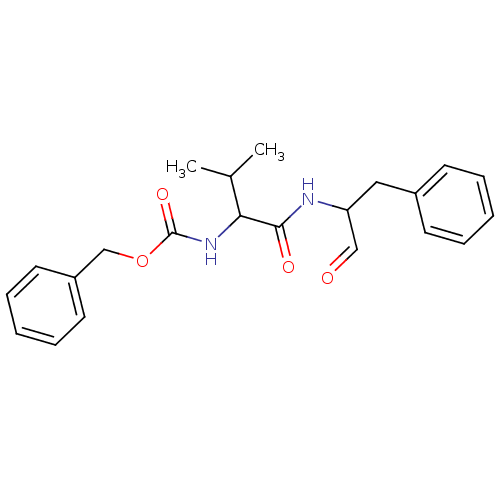 (MDL28170 | Z-Val-Phe-CHO | benzyl N-[(1S)-2-methyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM429284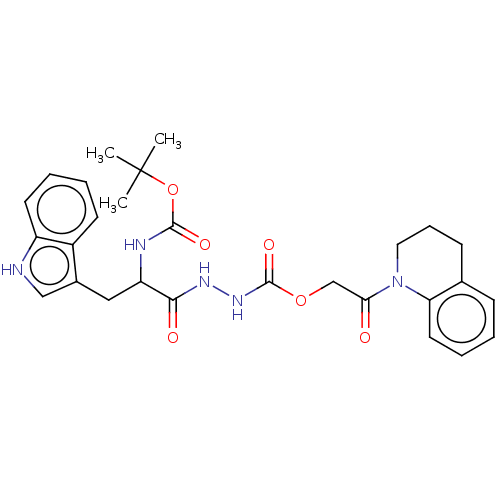 (med.21724, Compound 22) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 193 | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM19859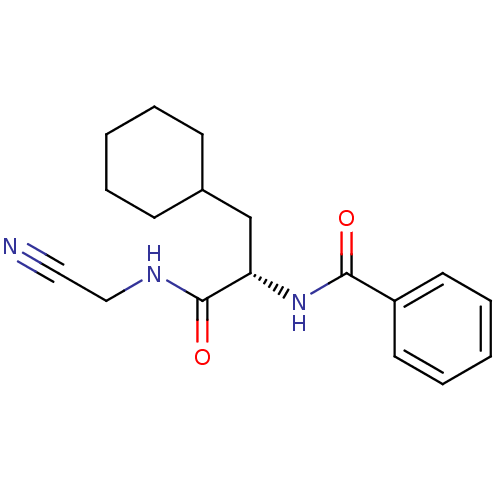 ((2S)-N-(cyanomethyl)-3-cyclohexyl-2-(phenylformami...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 55 | n/a | 230 | n/a | n/a | 5.5 | 22 |
Merck Frosst Centre for Therapeutic Research | Assay Description Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ... | J Med Chem 48: 7535-43 (2005) Article DOI: 10.1021/jm0504961 BindingDB Entry DOI: 10.7270/Q28S4N63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM19856 ((2R)-N-(cyanomethyl)-4-methyl-2-{3-[4-(piperazin-1...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60E+3 | n/a | 320 | n/a | n/a | 5.5 | 22 |
Merck Frosst Centre for Therapeutic Research | Assay Description Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ... | J Med Chem 48: 7535-43 (2005) Article DOI: 10.1021/jm0504961 BindingDB Entry DOI: 10.7270/Q28S4N63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM19854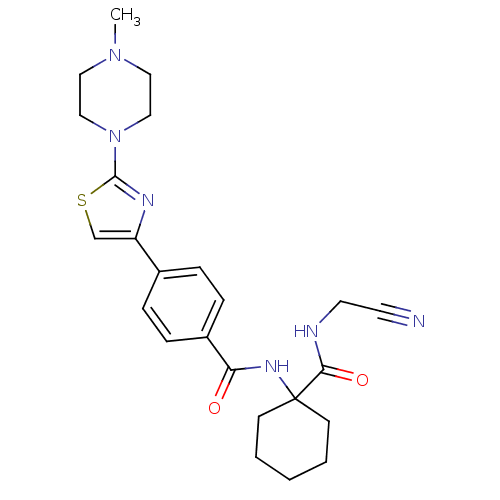 (CHEMBL426819 | CRA-013783/L-006235 | N-{1-[(cyanom...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.30E+3 | n/a | 340 | n/a | n/a | 5.5 | 22 |
Merck Frosst Centre for Therapeutic Research | Assay Description Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ... | J Med Chem 48: 7535-43 (2005) Article DOI: 10.1021/jm0504961 BindingDB Entry DOI: 10.7270/Q28S4N63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM429290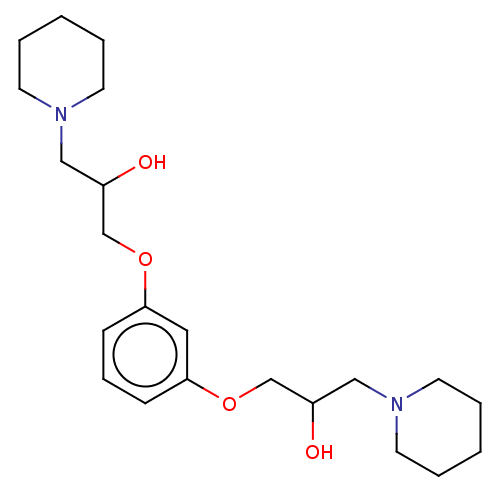 (med.21724, Compound 26) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM50176994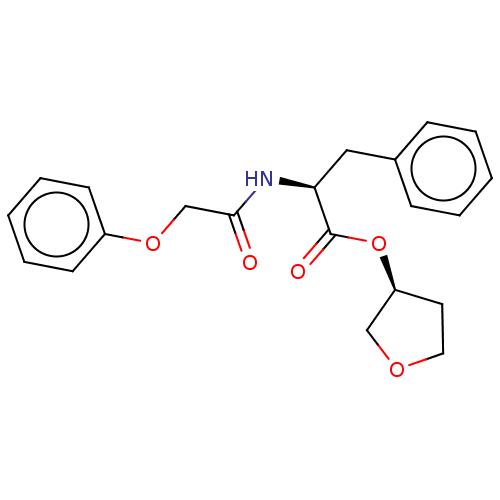 (CHEMBL3813784) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a |
Swiss Federal Institute of Technology (ETH) Curated by ChEMBL | Assay Description Activation of human liver cathepsin L using Z-FR-AMC as substrate measured for 120 mins by fluorescence assay | J Med Chem 59: 4077-86 (2016) Article DOI: 10.1021/acs.jmedchem.5b01849 BindingDB Entry DOI: 10.7270/Q2FN184P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM50376204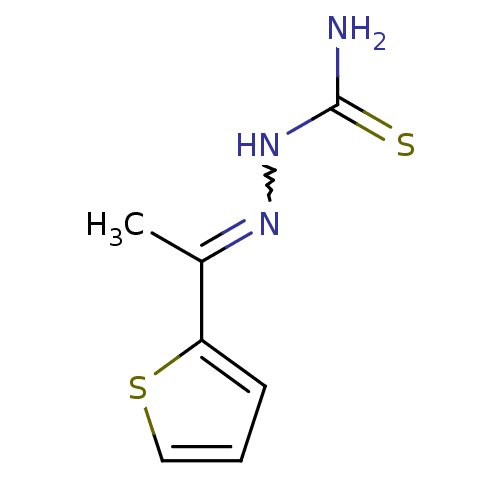 (CHEMBL401925 | med.21724, Compound 23) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM429289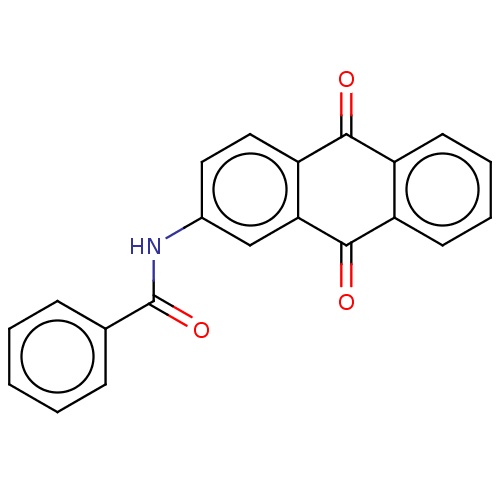 (med.21724, Compound 25) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 9.70E+3 | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM19857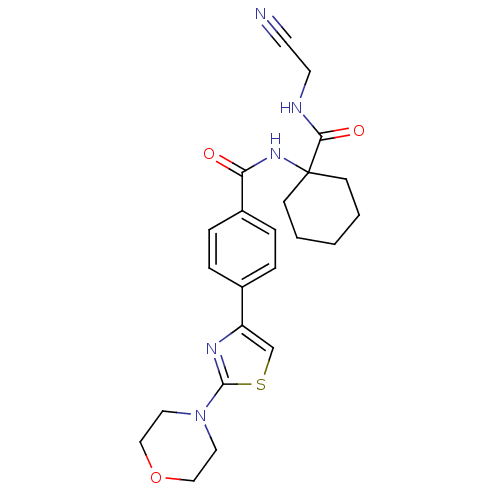 (N-{1-[(cyanomethyl)carbamoyl]cyclohexyl}-4-[2-(mor...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.40E+4 | n/a | >1.00E+4 | n/a | n/a | 5.5 | 22 |
Merck Frosst Centre for Therapeutic Research | Assay Description Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ... | J Med Chem 48: 7535-43 (2005) Article DOI: 10.1021/jm0504961 BindingDB Entry DOI: 10.7270/Q28S4N63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM19858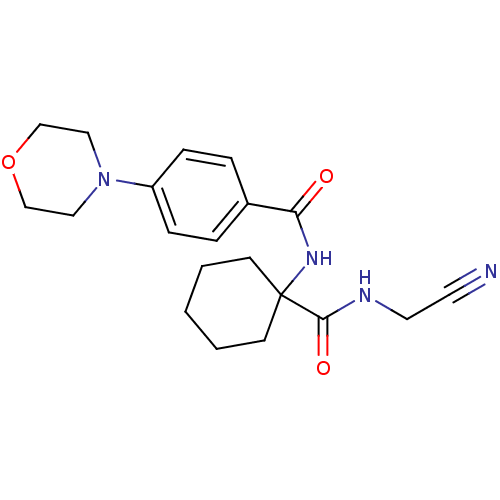 (N-{1-[(cyanomethyl)carbamoyl]cyclohexyl}-4-(morpho...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.20E+4 | n/a | >1.00E+4 | n/a | n/a | 5.5 | 22 |
Merck Frosst Centre for Therapeutic Research | Assay Description Prior to the addition of substrate, different concentrations of the inhibitor ranging from 100 uM to 0.2 nM were preincubated for 15 min with enzyme ... | J Med Chem 48: 7535-43 (2005) Article DOI: 10.1021/jm0504961 BindingDB Entry DOI: 10.7270/Q28S4N63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Procathepsin L (Homo sapiens (Human)) | BDBM11318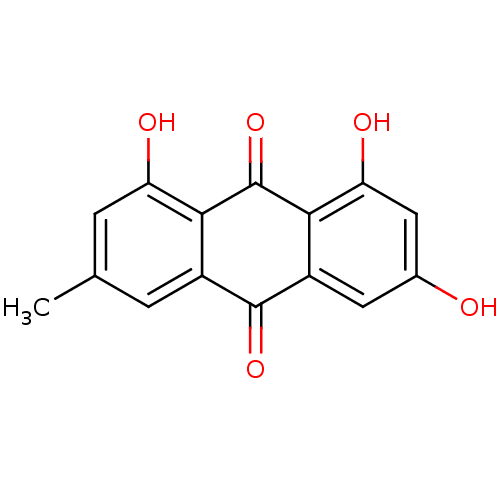 (1,3,8-trihydroxy-6-methyl-9,10-dihydroanthracene-9...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.00E+5 | n/a | n/a | n/a | n/a |
University of Bonn | Assay Description This is a review article. | Med Res Rev (2020) Article DOI: 10.1002/med.21724 BindingDB Entry DOI: 10.7270/Q2JS9ST6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||