

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neurotensin receptor type 1 (Rattus norvegicus) | BDBM50240845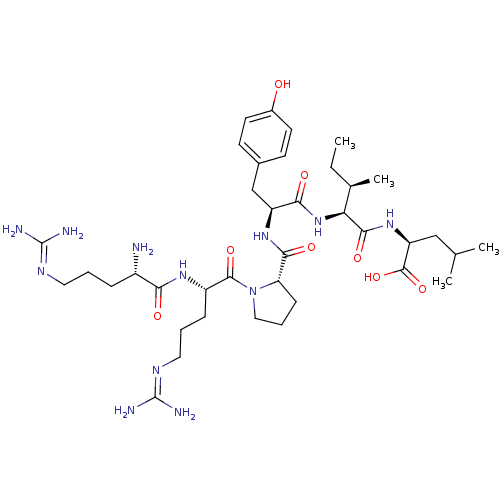 ((S)-2-{(2S,3R)-2-[(S)-2-({(S)-1-[(S)-2-((S)-2-Amin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Agonist activity at rat NTS1 stably expressed in CHOK1 cells assessed as induction of calcium release by FLIPR assay | J Med Chem 57: 5318-32 (2014) Article DOI: 10.1021/jm5003843 BindingDB Entry DOI: 10.7270/Q2PR7XJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Rattus norvegicus) | BDBM50130880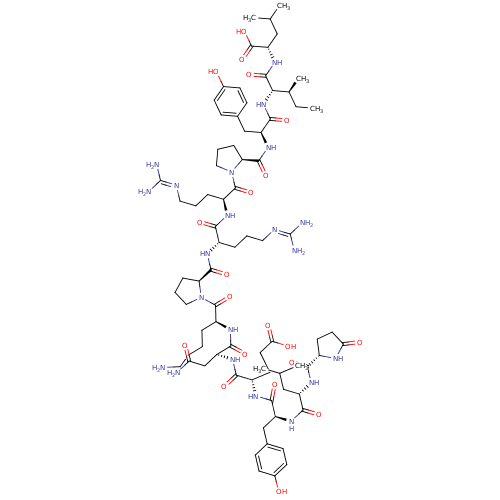 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Agonist activity at rat NTS1 stably expressed in CHOK1 cells assessed as induction of calcium release by FLIPR assay | J Med Chem 57: 5318-32 (2014) Article DOI: 10.1021/jm5003843 BindingDB Entry DOI: 10.7270/Q2PR7XJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.0420 | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Agonist activity at NTSR1 (unknown origin) expressed in CHO cells assessed as potentiation of NT(8-13) peptide-induced change in intracellular Ca2+ l... | Bioorg Med Chem Lett 24: 3974-8 (2014) Article DOI: 10.1016/j.bmcl.2014.06.033 BindingDB Entry DOI: 10.7270/Q2ZP47RG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.0692 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at human neurotensin receptor 1 expressed in CHO cells assessed as increase in intracellular calcium by Fluo-4 dye based fluorescenc... | ACS Med Chem Lett 11: 16-22 (2020) Article DOI: 10.1021/acsmedchemlett.9b00462 BindingDB Entry DOI: 10.7270/Q2SF30GN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.150 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Agonist activity at NTR1 (unknown origin) expressed in CHOK1 cells by calcium mobilization assay | Bioorg Med Chem Lett 19: 1438-41 (2009) Article DOI: 10.1016/j.bmcl.2009.01.024 BindingDB Entry DOI: 10.7270/Q2S46RVQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553767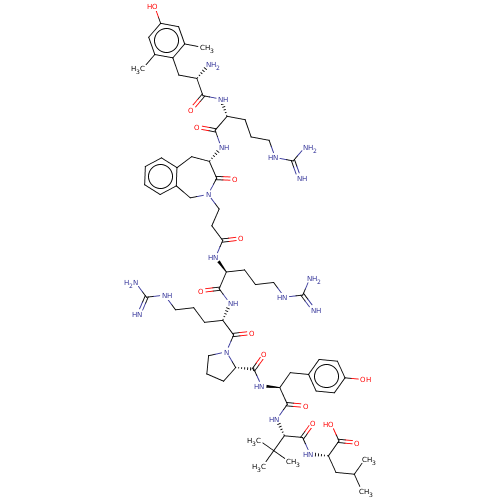 (CHEMBL4789659) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.240 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553768 (CHEMBL4744531) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.260 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50124135 (CHEMBL3622803) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.340 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in CHOK1 cells co-expressing hNTS1-GFP10/RlucII-beta-arrestin 2 assessed as beta-arrestin2 recruitm... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50509513 (CHEMBL4537266) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.372 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at human neurotensin receptor 1 expressed in CHO cells assessed as increase in intracellular calcium by Fluo-4 dye based fluorescenc... | ACS Med Chem Lett 11: 16-22 (2020) Article DOI: 10.1021/acsmedchemlett.9b00462 BindingDB Entry DOI: 10.7270/Q2SF30GN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at Galpha13-Rluc2/Gbeta1/GFP10-Ggamma1 fused human NTS1 expressed in CHO-K1 cell membranes assessed as Galpha13 activation after 5 m... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50509511 (CHEMBL4457798) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.417 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at human neurotensin receptor 1 expressed in CHO cells assessed as increase in intracellular calcium by Fluo-4 dye based fluorescenc... | ACS Med Chem Lett 11: 16-22 (2020) Article DOI: 10.1021/acsmedchemlett.9b00462 BindingDB Entry DOI: 10.7270/Q2SF30GN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at GFP10/beta-arrestin1-Rluc2 fused human NTS1 expressed in CHO-K1 cell membranes assessed as beta-arrestin1 recruitment after 20 mi... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at GFP10/beta-arrestin2-Rluc2 fused human NTS1 expressed in CHO-K1 cell membranes assessed as beta-arrestin2 recruitment after 20 mi... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at Galphaq-Rluc2/Gbeta1/GFP10-Ggamma1 fused human NTS1 expressed in CHO-K1 cell membranes assessed as Galphaq activation after 5 min... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50257186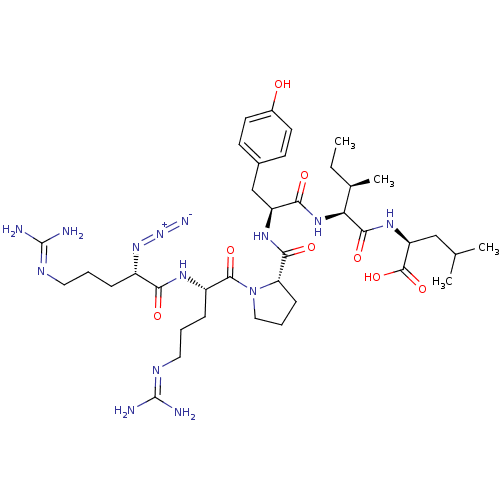 ((S)-2-((2S,3R)-2-((S)-2-((S)-1-((S)-2-((S)-2-azido...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.780 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in human HT-29 cells assessed as increase in intracellular calcium concentration by FLIPR assay | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50257185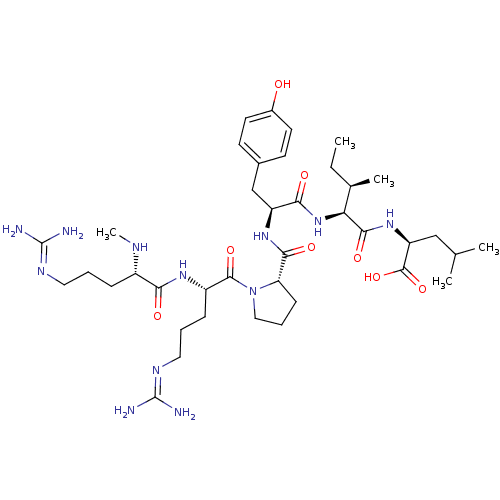 ((S)-2-((2S,3R)-2-((S)-2-((S)-1-((S)-5-guanidino-2-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.853 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in human HT-29 cells assessed as increase in intracellular calcium concentration by FLIPR assay | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553771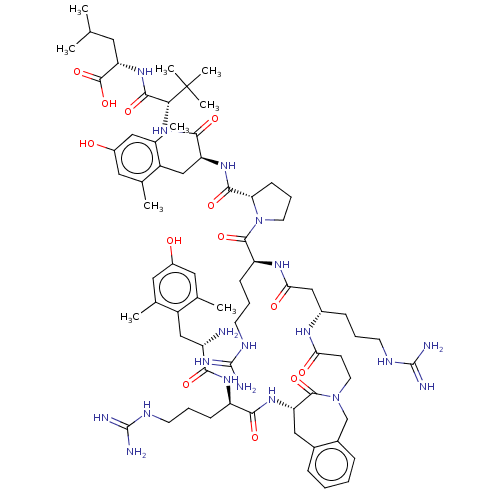 (CHEMBL4776986) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 0.860 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50240845 ((S)-2-{(2S,3R)-2-[(S)-2-({(S)-1-[(S)-2-((S)-2-Amin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in human HT-29 cells assessed as increase in intracellular calcium concentration by FLIPR assay | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50509510 (CHEMBL4564746) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at human neurotensin receptor 1 in HT-29 cells assessed as intracellular calcium mobilization by Fura-2 dye based spectrofluorimetry... | ACS Med Chem Lett 11: 16-22 (2020) Article DOI: 10.1021/acsmedchemlett.9b00462 BindingDB Entry DOI: 10.7270/Q2SF30GN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM297513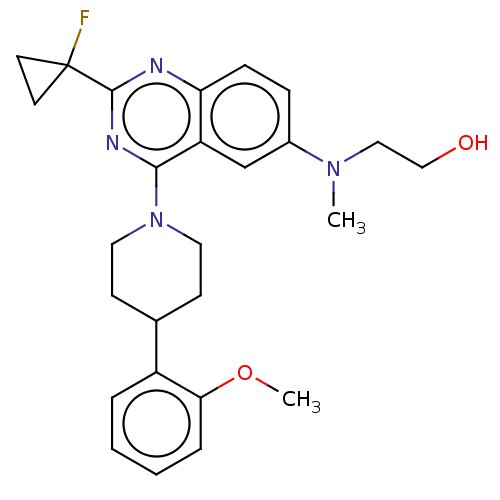 (2-({2-(1-Fluoro-cyclopropyl)-4-[4- (2-methoxy-phen...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a |
Sanford Burnham Prebys Medical Discovery Institute Curated by ChEMBL | Assay Description Positive allosteric modulation of human GFP-fused PK-tagged Gq-coupled NTR1 expressed in CHOK1 cells assessed as effect on NT (8 to 13) peptide-media... | J Med Chem 62: 8357-8363 (2019) Article DOI: 10.1021/acs.jmedchem.9b00340 BindingDB Entry DOI: 10.7270/Q2HQ438V | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at human neurotensin receptor 1 in HT-29 cells assessed as intracellular calcium mobilization by Fura-2 dye based spectrofluorimetry... | ACS Med Chem Lett 11: 16-22 (2020) Article DOI: 10.1021/acsmedchemlett.9b00462 BindingDB Entry DOI: 10.7270/Q2SF30GN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553769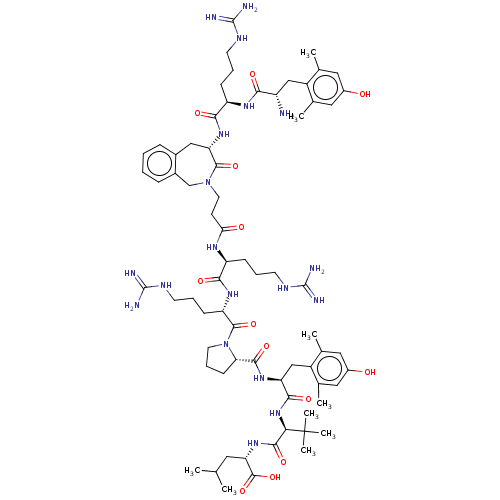 (CHEMBL4798887) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at NTSR1 in human HT-29 cells assessed as induction of intracellular Ca2+ mobilization by fura-2 dye-based spectrofluorimetric analy... | J Med Chem 59: 1925-45 (2016) Article DOI: 10.1021/acs.jmedchem.5b01495 BindingDB Entry DOI: 10.7270/Q2V69MGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Rattus norvegicus) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 in Sprague-Dawley rat ileum assessed as inhibition of carbachol-induced contraction | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Rattus norvegicus) | BDBM50124132 (CHEMBL3622802) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 in Sprague-Dawley rat ileum assessed as inhibition of carbachol-induced contraction | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in CHOK1 cells co-expressing Galphaq-RlucII(121)/Gbeta1/GFP10-Ggamma1 assessed as Galpha-q stimulat... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at NTSR1 in human HT-29 cells assessed as induction of intracellular Ca2+ mobilization by fura-2 dye-based spectrofluorimetric analy... | J Med Chem 59: 1925-45 (2016) Article DOI: 10.1021/acs.jmedchem.5b01495 BindingDB Entry DOI: 10.7270/Q2V69MGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50276349 (CHEMBL450740 | Polyamine-NT) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Agonist activity at human recombinant NTS1 receptor expressed in HEK293 cells assessed as stimulation of Ca2+ mobilization | J Med Chem 52: 1514-7 (2009) Article DOI: 10.1021/jm801481y BindingDB Entry DOI: 10.7270/Q2M908JS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50124132 (CHEMBL3622802) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in CHOK1 cells co-expressing Galphaq-RlucII(121)/Gbeta1/GFP10-Ggamma1 assessed as Galpha-q stimulat... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50124132 (CHEMBL3622802) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in CHOK1 cells co-expressing hNTS1-GFP10/RlucII-beta-arrestin 2 assessed as beta-arrestin2 recruitm... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a |
Friedrich Alexander University Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in HEK293 cells assessed as inhibition of constitutive activity on MAPK-mediated luciferase activit... | J Med Chem 54: 2915-23 (2011) Article DOI: 10.1021/jm200006c BindingDB Entry DOI: 10.7270/Q2XK8FVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50159155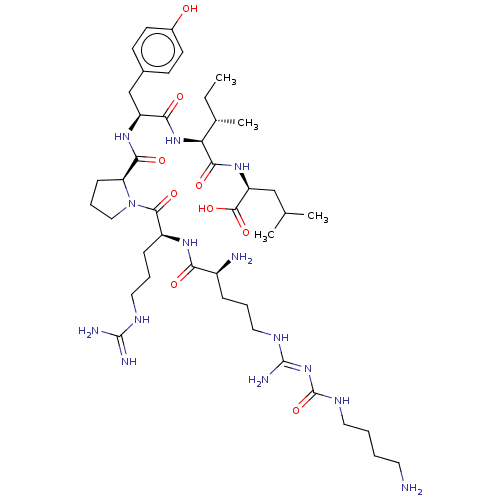 (CHEMBL3786852) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at NTSR1 in human HT-29 cells assessed as induction of intracellular Ca2+ mobilization by fura-2 dye-based spectrofluorimetric analy... | J Med Chem 59: 1925-45 (2016) Article DOI: 10.1021/acs.jmedchem.5b01495 BindingDB Entry DOI: 10.7270/Q2V69MGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553777 (CHEMBL4597094) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50159155 (CHEMBL3786852) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at NTSR1 in human HT-29 cells assessed as induction of intracellular Ca2+ mobilization by fura-2 dye-based spectrofluorimetric analy... | J Med Chem 59: 1925-45 (2016) Article DOI: 10.1021/acs.jmedchem.5b01495 BindingDB Entry DOI: 10.7270/Q2V69MGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553772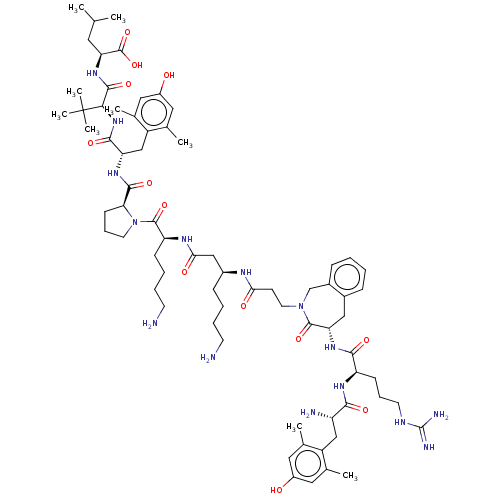 (CHEMBL4749016) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50257188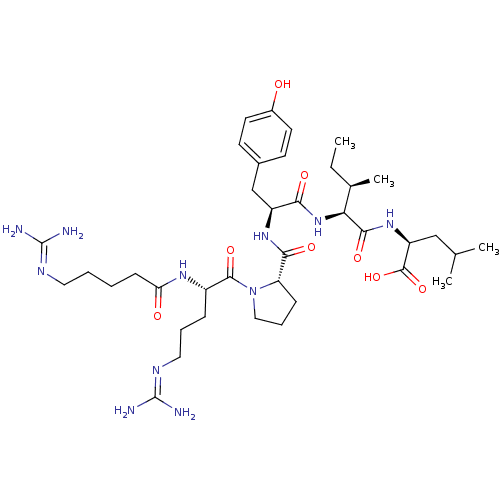 ((S)-2-((2S,3R)-2-((S)-2-((S)-1-((S)-5-guanidino-2-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.85 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in human HT-29 cells assessed as increase in intracellular calcium concentration by FLIPR assay | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50240844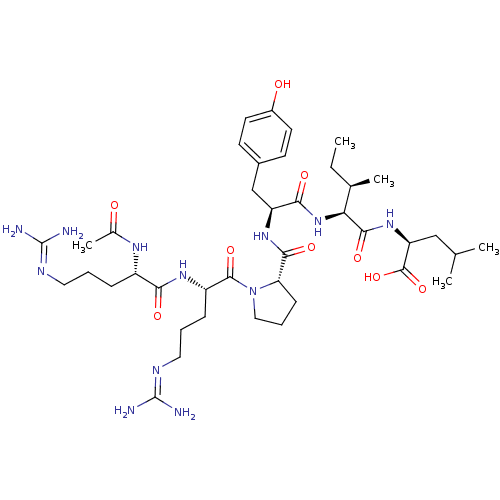 ((S)-2-((2S,3R)-2-((S)-2-((S)-1-((S)-2-((S)-2-aceta...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.89 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in human HT-29 cells assessed as increase in intracellular calcium concentration by FLIPR assay | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in CHOK1 cells co-expressing hNTS1-GFP10/RlucII-beta-arrestin 2 assessed as beta-arrestin2 recruitm... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.98 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in human HT-29 cells assessed as increase in intracellular calcium concentration by FLIPR assay | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at human NTR1 assessed as increase in intracellular calcium concentration | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50159156 (CHEMBL3786291) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at NTSR1 in human HT-29 cells assessed as induction of intracellular Ca2+ mobilization by fura-2 dye-based spectrofluorimetric analy... | J Med Chem 59: 1925-45 (2016) Article DOI: 10.1021/acs.jmedchem.5b01495 BindingDB Entry DOI: 10.7270/Q2V69MGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50159156 (CHEMBL3786291) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at NTSR1 in human HT-29 cells assessed as induction of intracellular Ca2+ mobilization by fura-2 dye-based spectrofluorimetric analy... | J Med Chem 59: 1925-45 (2016) Article DOI: 10.1021/acs.jmedchem.5b01495 BindingDB Entry DOI: 10.7270/Q2V69MGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (MOUSE) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Agonist activity at NTR1 in mouse N1E-115 cells assessed as increase in intracellular calcium concentration | J Med Chem 52: 1803-13 (2009) Article DOI: 10.1021/jm801072v BindingDB Entry DOI: 10.7270/Q2VD6Z9D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50458820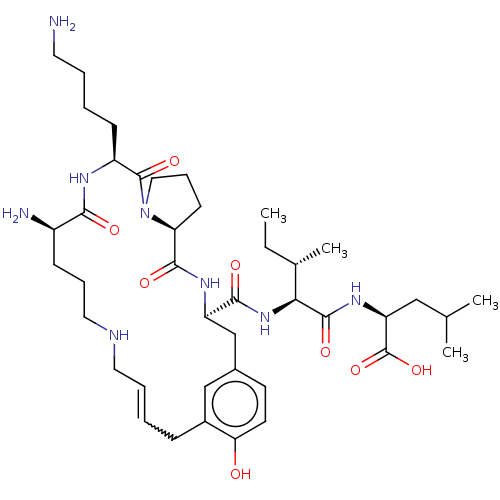 (CHEMBL4216311) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at Galphaq-Rluc2/Gbeta1/GFP10-Ggamma1 fused human NTS1 expressed in CHO-K1 cell membranes assessed as Galphaq activation after 5 min... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50458820 (CHEMBL4216311) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at GFP10/beta-arrestin2-Rluc2 fused human NTS1 expressed in CHO-K1 cell membranes assessed as beta-arrestin2 recruitment after 20 mi... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50553770 (CHEMBL4758754) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 3.90 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human NTS1 expressed in HEK293 cells co-expressing Galphaq/RlucII/GFP10-Ggamma1/Gbeta1 assessed as increase in Galphaq activation... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01376 BindingDB Entry DOI: 10.7270/Q2H41W2P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50458820 (CHEMBL4216311) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Agonist activity at GFP10/beta-arrestin1-Rluc2 fused human NTS1 expressed in CHO-K1 cell membranes assessed as beta-arrestin1 recruitment after 20 mi... | J Med Chem 61: 7103-7115 (2018) Article DOI: 10.1021/acs.jmedchem.8b00175 BindingDB Entry DOI: 10.7270/Q2D50QMT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50124141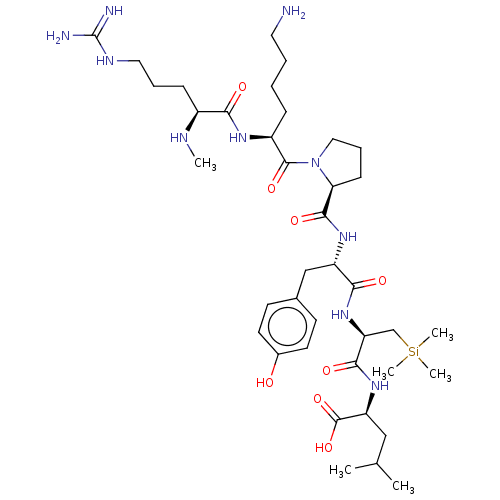 (CHEMBL3622805) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.20 | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Agonist activity at human NTS1 receptor expressed in CHOK1 cells co-expressing hNTS1-GFP10/RlucII-beta-arrestin 2 assessed as beta-arrestin2 recruitm... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 1 (Homo sapiens (Human)) | BDBM50509511 (CHEMBL4457798) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 5.90 | n/a | n/a | n/a | n/a |
University of Regensburg Curated by ChEMBL | Assay Description Agonist activity at human neurotensin receptor 1 in HT-29 cells assessed as intracellular calcium mobilization by Fura-2 dye based spectrofluorimetry... | ACS Med Chem Lett 11: 16-22 (2020) Article DOI: 10.1021/acsmedchemlett.9b00462 BindingDB Entry DOI: 10.7270/Q2SF30GN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 919 total ) | Next | Last >> |