 Found 131 hits of ic50 data for polymerid = 50001306
Found 131 hits of ic50 data for polymerid = 50001306 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM4552
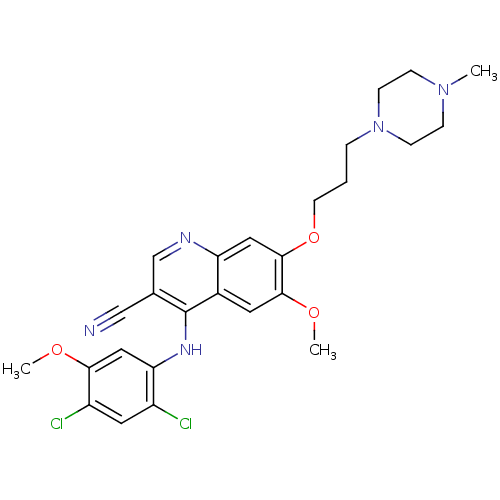
(4-[(2,4-Dichloro-5-methoxyphenyl)amino]-6-methoxy-...)Show SMILES COc1cc(Nc2c(cnc3cc(OCCCN4CCN(C)CC4)c(OC)cc23)C#N)c(Cl)cc1Cl Show InChI InChI=1S/C26H29Cl2N5O3/c1-32-6-8-33(9-7-32)5-4-10-36-25-13-21-18(11-24(25)35-3)26(17(15-29)16-30-21)31-22-14-23(34-2)20(28)12-19(22)27/h11-14,16H,4-10H2,1-3H3,(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Center for Molecular Medicine of the Austrian Academy of Sciences
Curated by ChEMBL
| Assay Description
Inhibition of ABL2 |
Leukemia 23: 477-85 (2009)
Article DOI: 10.1038/leu.2008.334
BindingDB Entry DOI: 10.7270/Q22Z15R6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427749
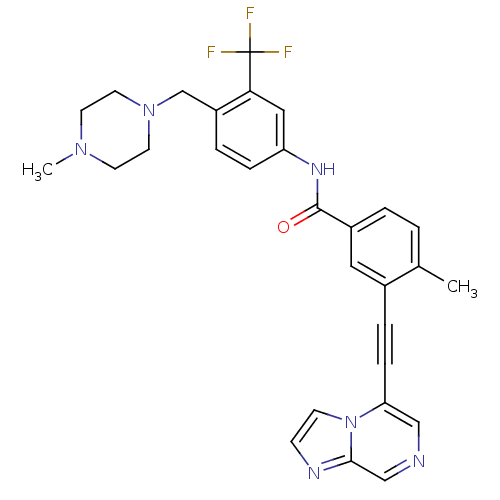
(CHEMBL2324924)Show SMILES CN1CCN(Cc2ccc(NC(=O)c3ccc(C)c(c3)C#Cc3cncc4nccn34)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C29H27F3N6O/c1-20-3-4-22(15-21(20)6-8-25-17-33-18-27-34-9-10-38(25)27)28(39)35-24-7-5-23(26(16-24)29(30,31)32)19-37-13-11-36(2)12-14-37/h3-5,7,9-10,15-18H,11-14,19H2,1-2H3,(H,35,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427749

(CHEMBL2324924)Show SMILES CN1CCN(Cc2ccc(NC(=O)c3ccc(C)c(c3)C#Cc3cncc4nccn34)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C29H27F3N6O/c1-20-3-4-22(15-21(20)6-8-25-17-33-18-27-34-9-10-38(25)27)28(39)35-24-7-5-23(26(16-24)29(30,31)32)19-37-13-11-36(2)12-14-37/h3-5,7,9-10,15-18H,11-14,19H2,1-2H3,(H,35,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427748
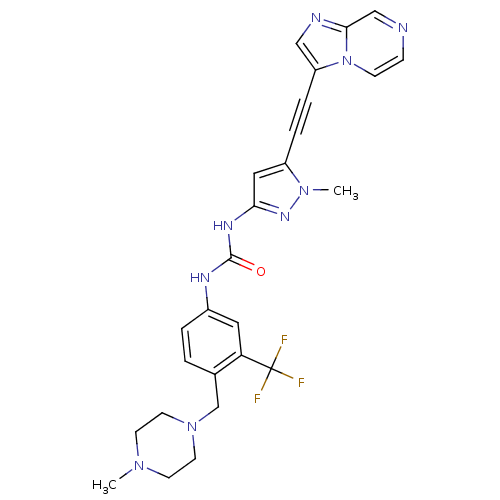
(CHEMBL2324925)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cnc5cnccn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H26F3N9O/c1-35-9-11-37(12-10-35)17-18-3-4-19(13-22(18)26(27,28)29)32-25(39)33-23-14-20(36(2)34-23)5-6-21-15-31-24-16-30-7-8-38(21)24/h3-4,7-8,13-16H,9-12,17H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM378885
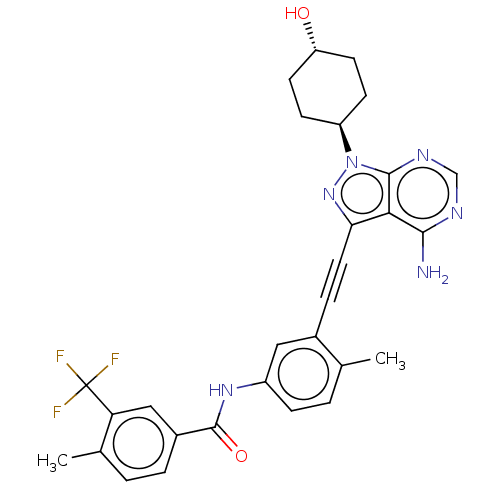
(US10266537, Compound 93)Show SMILES Cc1ccc(NC(=O)c2ccc(C)c(c2)C(F)(F)F)cc1C#Cc1nn([C@H]2CC[C@H](O)CC2)c2ncnc(N)c12 |r,wU:26.27,wD:29.31,(-5.46,5.4,;-3.98,5.8,;-3.58,7.29,;-2.09,7.69,;-1,6.6,;.49,7,;1.57,5.91,;1.18,4.42,;3.06,6.31,;3.46,7.79,;4.95,8.19,;6.04,7.1,;7.53,7.5,;5.64,5.62,;4.15,5.22,;6.73,4.53,;8.22,4.93,;6.33,3.04,;7.82,3.44,;-1.4,5.11,;-2.89,4.71,;-3.29,3.22,;-3.68,1.74,;-4.08,.25,;-3.18,-1,;-4.08,-2.24,;-3.68,-3.73,;-2.2,-4.13,;-1.8,-5.62,;-2.89,-6.7,;-2.49,-8.19,;-4.38,-6.31,;-4.77,-4.82,;-5.55,-1.77,;-6.88,-2.54,;-8.22,-1.77,;-8.22,-.23,;-6.88,.54,;-6.88,2.08,;-5.55,-.23,)| Show InChI InChI=1S/C29H27F3N6O2/c1-16-4-7-20(36-28(40)19-5-3-17(2)23(14-19)29(30,31)32)13-18(16)6-12-24-25-26(33)34-15-35-27(25)38(37-24)21-8-10-22(39)11-9-21/h3-5,7,13-15,21-22,39H,8-11H2,1-2H3,(H,36,40)(H2,33,34,35)/t21-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Kalypsys, Inc.
| Assay Description
The aim of this experiment is to detect the inhibitory activity of the compounds of the present invention against in vitro protein kinases using isot... |
Bioorg Med Chem Lett 17: 3562-9 (2007)
BindingDB Entry DOI: 10.7270/Q21C2065 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM378885

(US10266537, Compound 93)Show SMILES Cc1ccc(NC(=O)c2ccc(C)c(c2)C(F)(F)F)cc1C#Cc1nn([C@H]2CC[C@H](O)CC2)c2ncnc(N)c12 |r,wU:26.27,wD:29.31,(-5.46,5.4,;-3.98,5.8,;-3.58,7.29,;-2.09,7.69,;-1,6.6,;.49,7,;1.57,5.91,;1.18,4.42,;3.06,6.31,;3.46,7.79,;4.95,8.19,;6.04,7.1,;7.53,7.5,;5.64,5.62,;4.15,5.22,;6.73,4.53,;8.22,4.93,;6.33,3.04,;7.82,3.44,;-1.4,5.11,;-2.89,4.71,;-3.29,3.22,;-3.68,1.74,;-4.08,.25,;-3.18,-1,;-4.08,-2.24,;-3.68,-3.73,;-2.2,-4.13,;-1.8,-5.62,;-2.89,-6.7,;-2.49,-8.19,;-4.38,-6.31,;-4.77,-4.82,;-5.55,-1.77,;-6.88,-2.54,;-8.22,-1.77,;-8.22,-.23,;-6.88,.54,;-6.88,2.08,;-5.55,-.23,)| Show InChI InChI=1S/C29H27F3N6O2/c1-16-4-7-20(36-28(40)19-5-3-17(2)23(14-19)29(30,31)32)13-18(16)6-12-24-25-26(33)34-15-35-27(25)38(37-24)21-8-10-22(39)11-9-21/h3-5,7,13-15,21-22,39H,8-11H2,1-2H3,(H,36,40)(H2,33,34,35)/t21-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human recombinant ARG (38 to end residues) using EAIYAAPFAKKK as substrate incubated for 40 mins in presence of [gamma33P-ATP] by radio... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2M90DC6 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427747
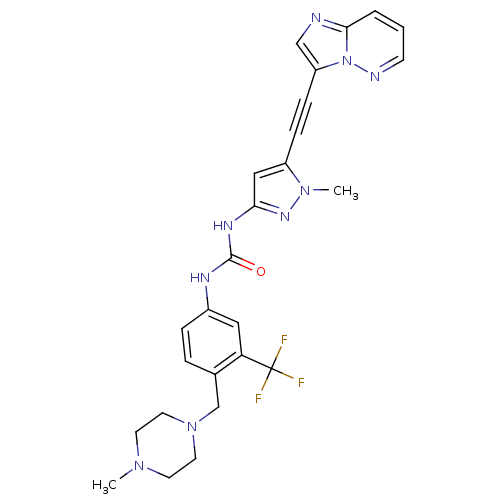
(CHEMBL2324926)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cnc5cccnn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H26F3N9O/c1-35-10-12-37(13-11-35)17-18-5-6-19(14-22(18)26(27,28)29)32-25(39)33-23-15-20(36(2)34-23)7-8-21-16-30-24-4-3-9-31-38(21)24/h3-6,9,14-16H,10-13,17H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427742
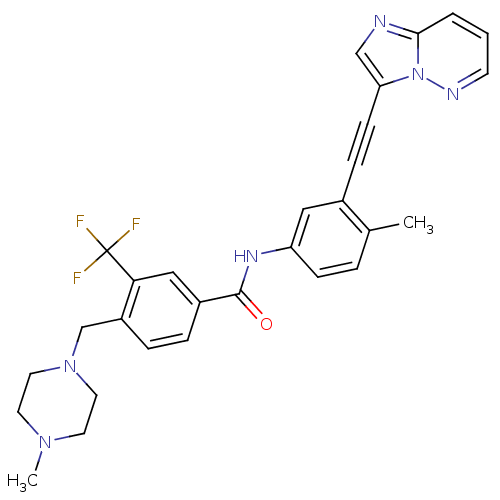
(CHEMBL2324930)Show SMILES CN1CCN(Cc2ccc(cc2C(F)(F)F)C(=O)Nc2ccc(C)c(c2)C#Cc2cnc3cccnn23)CC1 Show InChI InChI=1S/C29H27F3N6O/c1-20-5-9-24(16-21(20)8-10-25-18-33-27-4-3-11-34-38(25)27)35-28(39)22-6-7-23(26(17-22)29(30,31)32)19-37-14-12-36(2)13-15-37/h3-7,9,11,16-18H,12-15,19H2,1-2H3,(H,35,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427748

(CHEMBL2324925)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cnc5cnccn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H26F3N9O/c1-35-9-11-37(12-10-35)17-18-3-4-19(13-22(18)26(27,28)29)32-25(39)33-23-14-20(36(2)34-23)5-6-21-15-31-24-16-30-7-8-38(21)24/h3-4,7-8,13-16H,9-12,17H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10.1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human ABL2 using EAIYAAPFAKKK as substrate by [gamma-33P]-ATP assay |
Citation and Details
Article DOI: 10.1016/j.bmc.2018.02.022
BindingDB Entry DOI: 10.7270/Q2DJ5KB8 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427750
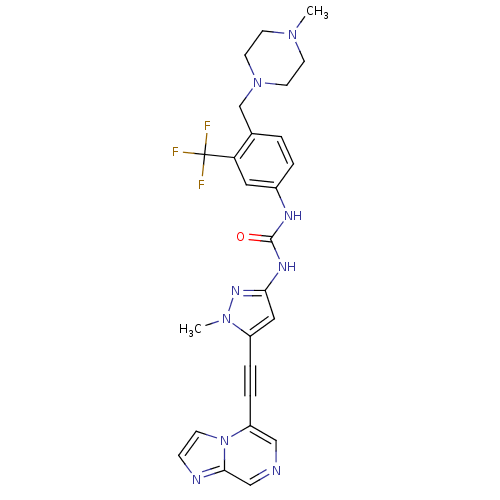
(CHEMBL2324923)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cncc5nccn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H26F3N9O/c1-35-9-11-37(12-10-35)17-18-3-4-19(13-22(18)26(27,28)29)32-25(39)33-23-14-20(36(2)34-23)5-6-21-15-30-16-24-31-7-8-38(21)24/h3-4,7-8,13-16H,9-12,17H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427745
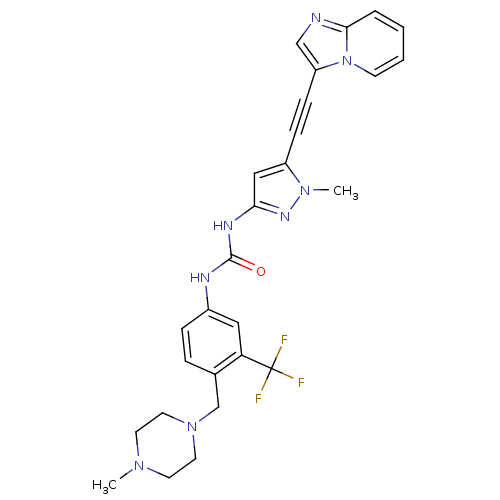
(CHEMBL2324927)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cnc5ccccn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C27H27F3N8O/c1-35-11-13-37(14-12-35)18-19-6-7-20(15-23(19)27(28,29)30)32-26(39)33-24-16-21(36(2)34-24)8-9-22-17-31-25-5-3-4-10-38(22)25/h3-7,10,15-17H,11-14,18H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427744
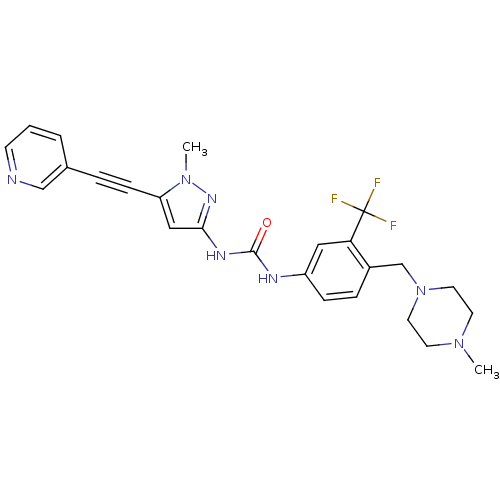
(CHEMBL2324928)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cccnc4)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C25H26F3N7O/c1-33-10-12-35(13-11-33)17-19-6-7-20(14-22(19)25(26,27)28)30-24(36)31-23-15-21(34(2)32-23)8-5-18-4-3-9-29-16-18/h3-4,6-7,9,14-16H,10-13,17H2,1-2H3,(H2,30,31,32,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427750

(CHEMBL2324923)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cncc5nccn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H26F3N9O/c1-35-9-11-37(12-10-35)17-18-3-4-19(13-22(18)26(27,28)29)32-25(39)33-23-14-20(36(2)34-23)5-6-21-15-30-16-24-31-7-8-38(21)24/h3-4,7-8,13-16H,9-12,17H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 15.8 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florida
Curated by ChEMBL
| Assay Description
Inhibition of human ABL2 using EAIYAAPFAKKK as substrate by [gamma-33P]-ATP assay |
Eur J Med Chem 161: 456-467 (2019)
Article DOI: 10.1016/j.ejmech.2018.10.052
BindingDB Entry DOI: 10.7270/Q2W380MT |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427741
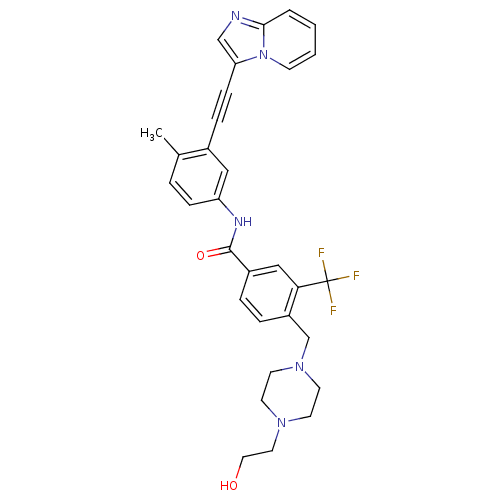
(CHEMBL2324931)Show SMILES Cc1ccc(NC(=O)c2ccc(CN3CCN(CCO)CC3)c(c2)C(F)(F)F)cc1C#Cc1cnc2ccccn12 Show InChI InChI=1S/C31H30F3N5O2/c1-22-5-9-26(18-23(22)8-10-27-20-35-29-4-2-3-11-39(27)29)36-30(41)24-6-7-25(28(19-24)31(32,33)34)21-38-14-12-37(13-15-38)16-17-40/h2-7,9,11,18-20,40H,12-17,21H2,1H3,(H,36,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481113
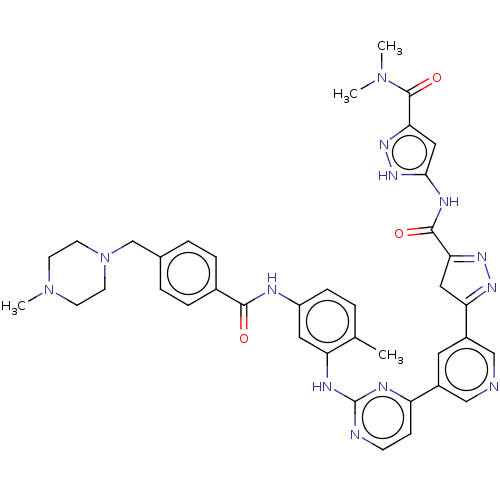
(US10906896, Cpd 117)Show SMILES CN(C)C(=O)c1cc(NC(=O)C2=NN=C(C2)c2cncc(c2)-c2ccnc(Nc3cc(NC(=O)c4ccc(CN5CCN(C)CC5)cc4)ccc3C)n2)[nH]n1 |c:13,t:11| Show InChI InChI=1S/C39H41N13O3/c1-24-5-10-29(42-36(53)26-8-6-25(7-9-26)23-52-15-13-51(4)14-16-52)18-31(24)44-39-41-12-11-30(43-39)27-17-28(22-40-21-27)32-19-33(47-46-32)37(54)45-35-20-34(48-49-35)38(55)50(2)3/h5-12,17-18,20-22H,13-16,19,23H2,1-4H3,(H,42,53)(H,41,43,44)(H2,45,48,49,54) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612783
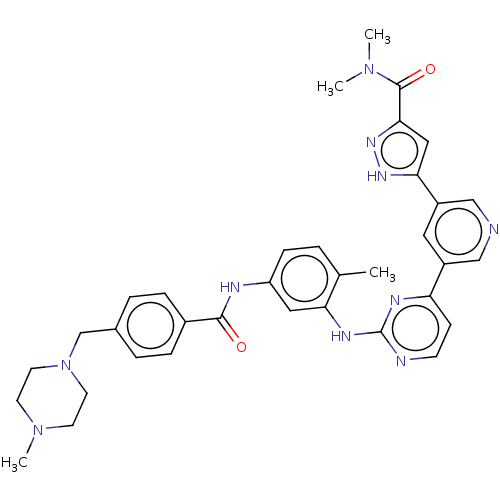
(US11725005, Compound 117)Show SMILES CN(C)C(=O)c1cc([nH]n1)-c1cncc(c1)-c1ccnc(Nc2cc(NC(=O)c3ccc(CN4CCN(C)CC4)cc3)ccc2C)n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427743
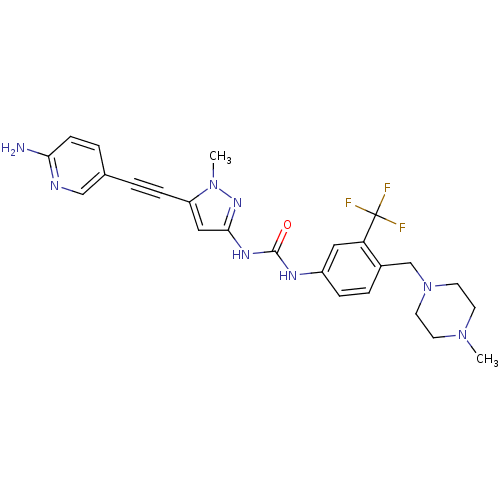
(CHEMBL2324929)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4ccc(N)nc4)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C25H27F3N8O/c1-34-9-11-36(12-10-34)16-18-5-6-19(13-21(18)25(26,27)28)31-24(37)32-23-14-20(35(2)33-23)7-3-17-4-8-22(29)30-15-17/h4-6,8,13-15H,9-12,16H2,1-2H3,(H2,29,30)(H2,31,32,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612893
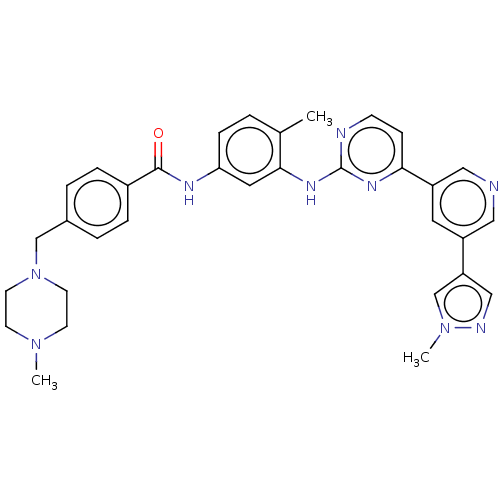
(US11725005, Compound 8300)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cnn(C)c3)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427742

(CHEMBL2324930)Show SMILES CN1CCN(Cc2ccc(cc2C(F)(F)F)C(=O)Nc2ccc(C)c(c2)C#Cc2cnc3cccnn23)CC1 Show InChI InChI=1S/C29H27F3N6O/c1-20-5-9-24(16-21(20)8-10-25-18-33-27-4-3-11-34-38(25)27)35-28(39)22-6-7-23(26(17-22)29(30,31)32)19-37-14-12-36(2)13-15-37/h3-7,9,11,16-18H,12-15,19H2,1-2H3,(H,35,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481131
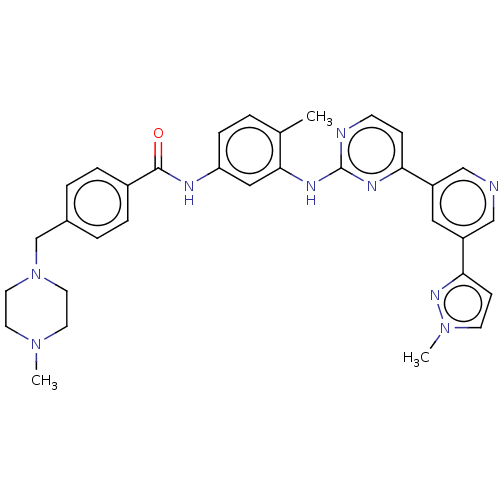
(US10906896, Cpd 8300 | US10906896, Cpd 832 | US117...)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3ccn(C)n3)c2)CC1 Show InChI InChI=1S/C33H35N9O/c1-23-4-9-28(36-32(43)25-7-5-24(6-8-25)22-42-16-14-40(2)15-17-42)19-31(23)38-33-35-12-10-29(37-33)26-18-27(21-34-20-26)30-11-13-41(3)39-30/h4-13,18-21H,14-17,22H2,1-3H3,(H,36,43)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427740
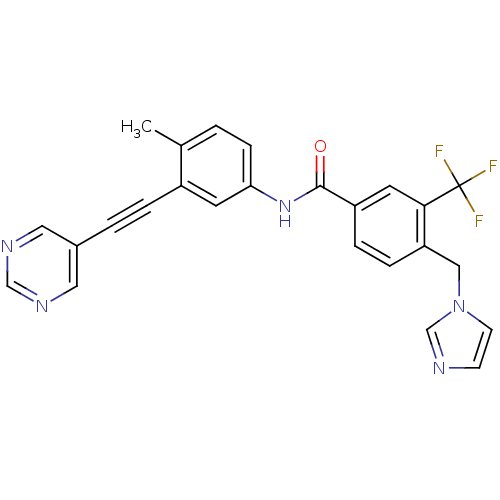
(CHEMBL2324932)Show SMILES Cc1ccc(NC(=O)c2ccc(Cn3ccnc3)c(c2)C(F)(F)F)cc1C#Cc1cncnc1 Show InChI InChI=1S/C25H18F3N5O/c1-17-2-7-22(10-19(17)4-3-18-12-30-15-31-13-18)32-24(34)20-5-6-21(14-33-9-8-29-16-33)23(11-20)25(26,27)28/h2,5-13,15-16H,14H2,1H3,(H,32,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427741

(CHEMBL2324931)Show SMILES Cc1ccc(NC(=O)c2ccc(CN3CCN(CCO)CC3)c(c2)C(F)(F)F)cc1C#Cc1cnc2ccccn12 Show InChI InChI=1S/C31H30F3N5O2/c1-22-5-9-26(18-23(22)8-10-27-20-35-29-4-2-3-11-39(27)29)36-30(41)24-6-7-25(28(19-24)31(32,33)34)21-38-14-12-37(13-15-38)16-17-40/h2-7,9,11,18-20,40H,12-17,21H2,1H3,(H,36,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481115
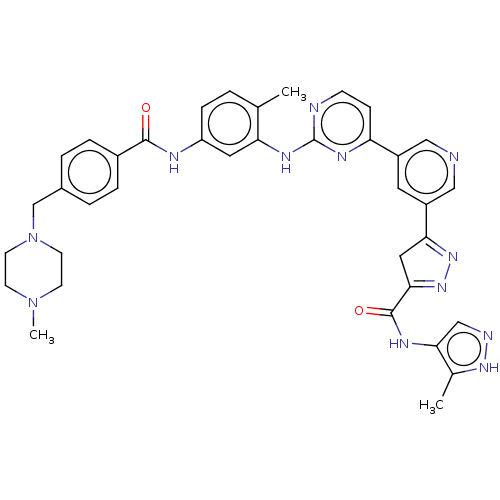
(US10906896, Cpd 119)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)C3=NN=C(C3)C(=O)Nc3cn[nH]c3C)c2)CC1 |c:39,t:37| Show InChI InChI=1S/C37H38N12O2/c1-23-4-9-29(41-35(50)26-7-5-25(6-8-26)22-49-14-12-48(3)13-15-49)17-31(23)44-37-39-11-10-30(43-37)27-16-28(20-38-19-27)32-18-33(47-46-32)36(51)42-34-21-40-45-24(34)2/h4-11,16-17,19-21H,12-15,18,22H2,1-3H3,(H,40,45)(H,41,50)(H,42,51)(H,39,43,44) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 44.7 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
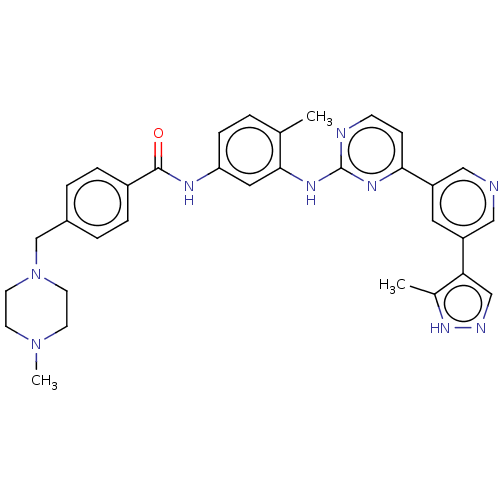
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612785
(US11725005, Compound 119)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cn[nH]c3C)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 44.7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427747

(CHEMBL2324926)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cnc5cccnn45)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H26F3N9O/c1-35-10-12-37(13-11-35)17-18-5-6-19(14-22(18)26(27,28)29)32-25(39)33-23-15-20(36(2)34-23)7-8-21-16-30-24-4-3-9-31-38(21)24/h3-6,9,14-16H,10-13,17H2,1-2H3,(H2,32,33,34,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
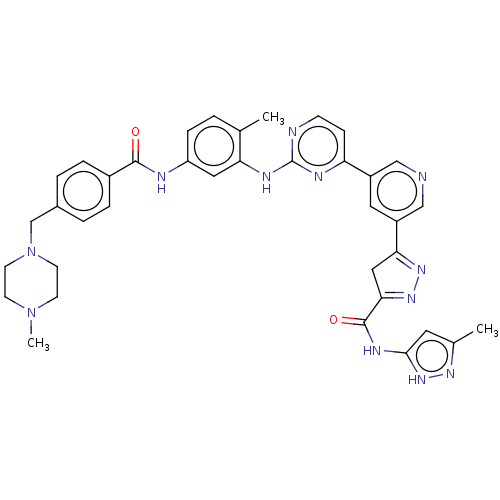
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481109
(US10906896, Cpd 113)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)C3=NN=C(C3)C(=O)Nc3cc(C)n[nH]3)c2)CC1 |c:39,t:37| Show InChI InChI=1S/C37H38N12O2/c1-23-4-9-29(40-35(50)26-7-5-25(6-8-26)22-49-14-12-48(3)13-15-49)18-31(23)42-37-39-11-10-30(41-37)27-17-28(21-38-20-27)32-19-33(46-45-32)36(51)43-34-16-24(2)44-47-34/h4-11,16-18,20-21H,12-15,19,22H2,1-3H3,(H,40,50)(H,39,41,42)(H2,43,44,47,51) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 50.1 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612772
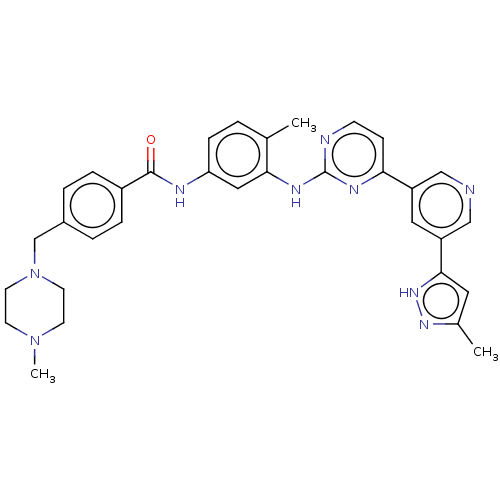
(US11725005, Compound 113)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cc(C)n[nH]3)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 50.1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481114
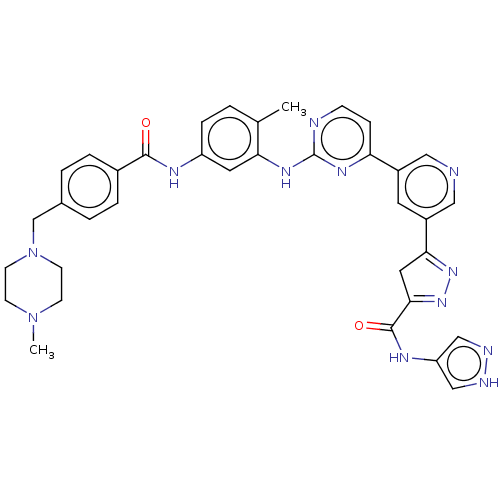
(US10906896, Cpd 118)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)C3=NN=C(C3)C(=O)Nc3cn[nH]c3)c2)CC1 |c:39,t:37| Show InChI InChI=1S/C36H36N12O2/c1-23-3-8-28(41-34(49)25-6-4-24(5-7-25)22-48-13-11-47(2)12-14-48)16-31(23)44-36-38-10-9-30(43-36)26-15-27(19-37-18-26)32-17-33(46-45-32)35(50)42-29-20-39-40-21-29/h3-10,15-16,18-21H,11-14,17,22H2,1-2H3,(H,39,40)(H,41,49)(H,42,50)(H,38,43,44) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481130
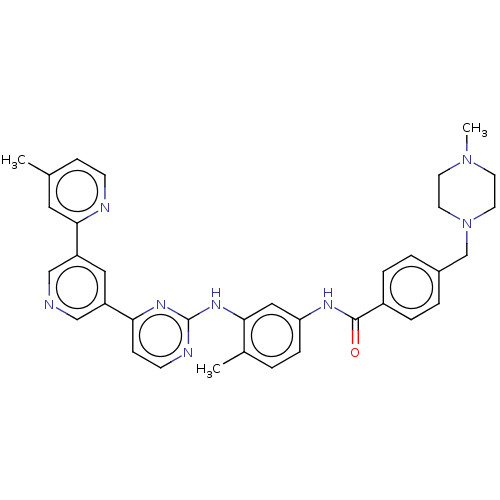
(US10906896, Cpd 830 | US11725005, Compound 830)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cc(C)ccn3)c2)CC1 Show InChI InChI=1S/C35H36N8O/c1-24-10-12-37-33(18-24)29-19-28(21-36-22-29)31-11-13-38-35(40-31)41-32-20-30(9-4-25(32)2)39-34(44)27-7-5-26(6-8-27)23-43-16-14-42(3)15-17-43/h4-13,18-22H,14-17,23H2,1-3H3,(H,39,44)(H,38,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481130

(US10906896, Cpd 830 | US11725005, Compound 830)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cc(C)ccn3)c2)CC1 Show InChI InChI=1S/C35H36N8O/c1-24-10-12-37-33(18-24)29-19-28(21-36-22-29)31-11-13-38-35(40-31)41-32-20-30(9-4-25(32)2)39-34(44)27-7-5-26(6-8-27)23-43-16-14-42(3)15-17-43/h4-13,18-22H,14-17,23H2,1-3H3,(H,39,44)(H,38,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612784
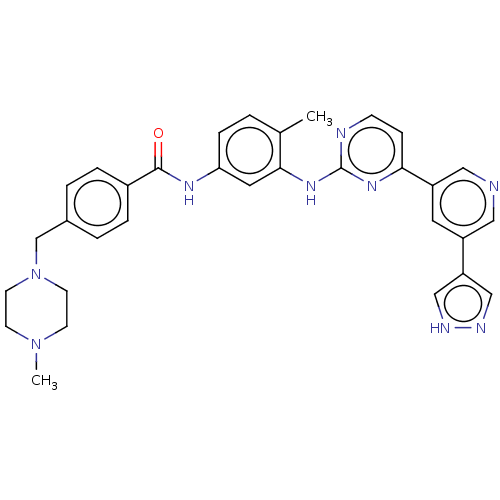
(US11725005, Compound 118)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cn[nH]c3)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427744

(CHEMBL2324928)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4cccnc4)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C25H26F3N7O/c1-33-10-12-35(13-11-33)17-19-6-7-20(14-22(19)25(26,27)28)30-24(36)31-23-15-21(34(2)32-23)8-5-18-4-3-9-29-16-18/h3-4,6-7,9,14-16H,10-13,17H2,1-2H3,(H2,30,31,32,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 66 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM50427743

(CHEMBL2324929)Show SMILES CN1CCN(Cc2ccc(NC(=O)Nc3cc(C#Cc4ccc(N)nc4)n(C)n3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C25H27F3N8O/c1-34-9-11-36(12-10-34)16-18-5-6-19(13-21(18)25(26,27)28)31-24(37)32-23-14-20(35(2)33-23)7-3-17-4-8-22(29)30-15-17/h4-6,8,13-15H,9-12,16H2,1-2H3,(H2,29,30)(H2,31,32,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Cyclofluidic Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Abl2 kinase (unknown origin) using Tyr 6 peptide as substrate assessed as residual enzyme activity after every 10 secs measured for 10 ... |
J Med Chem 56: 3033-47 (2013)
Article DOI: 10.1021/jm400099d
BindingDB Entry DOI: 10.7270/Q24T6KPD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481107
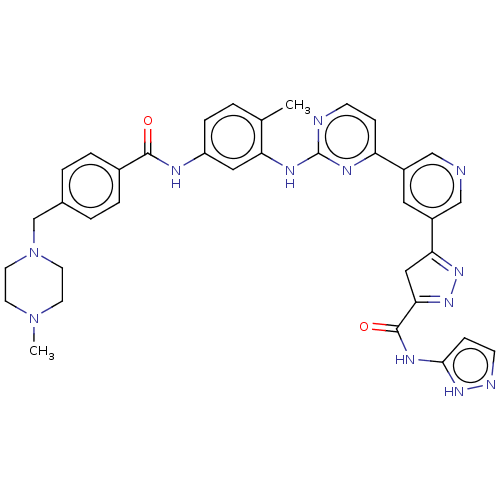
(US10906896, Cpd 107)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)C3=NN=C(C3)C(=O)Nc3ccn[nH]3)c2)CC1 |c:39,t:37| Show InChI InChI=1S/C36H36N12O2/c1-23-3-8-28(40-34(49)25-6-4-24(5-7-25)22-48-15-13-47(2)14-16-48)18-30(23)42-36-38-11-9-29(41-36)26-17-27(21-37-20-26)31-19-32(45-44-31)35(50)43-33-10-12-39-46-33/h3-12,17-18,20-21H,13-16,19,22H2,1-2H3,(H,40,49)(H,38,41,42)(H2,39,43,46,50) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612764
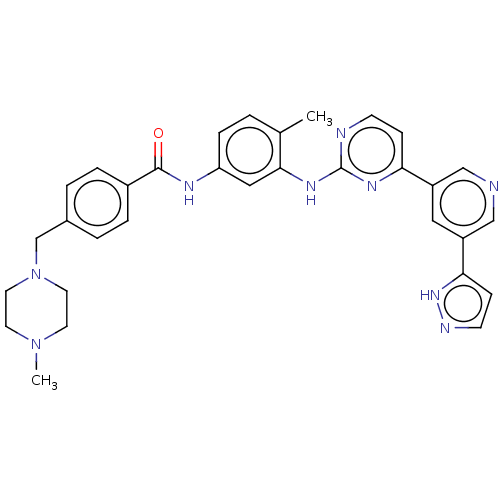
(US11725005, Compound 107)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3ccn[nH]3)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481119
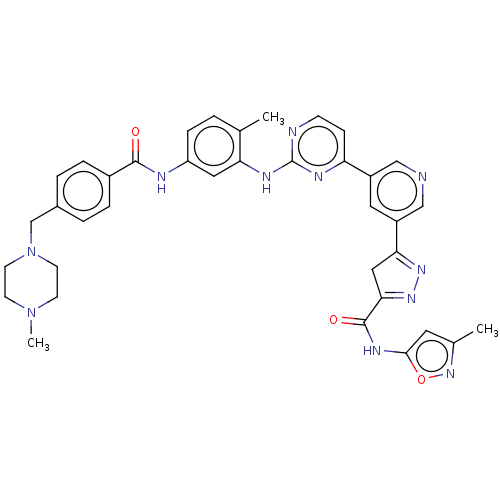
(US10906896, Cpd 207)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)C3=NN=C(C3)C(=O)Nc3cc(C)no3)c2)CC1 |c:39,t:37| Show InChI InChI=1S/C37H37N11O3/c1-23-4-9-29(40-35(49)26-7-5-25(6-8-26)22-48-14-12-47(3)13-15-48)18-31(23)42-37-39-11-10-30(41-37)27-17-28(21-38-20-27)32-19-33(45-44-32)36(50)43-34-16-24(2)46-51-34/h4-11,16-18,20-21H,12-15,19,22H2,1-3H3,(H,40,49)(H,43,50)(H,39,41,42) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 73.7 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
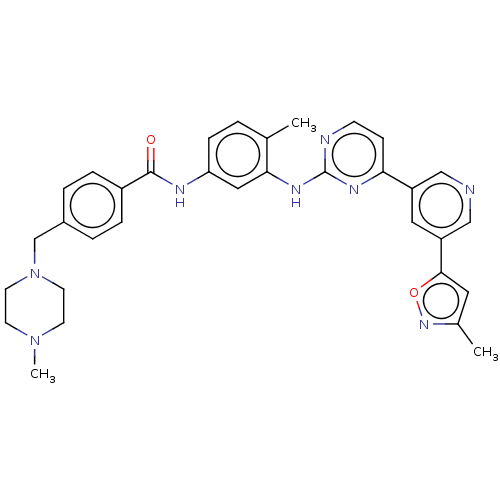
(Homo sapiens (Human)) | BDBM612791
(US11725005, Compound 207)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cc(C)no3)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 73.7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a | n/a |
Southeast University
Curated by ChEMBL
| Assay Description
Inhibition of ARG (unknown origin) incubated for 1 hr by spectrophotometric analysis |
Bioorg Med Chem 24: 3483-93 (2016)
Article DOI: 10.1016/j.bmc.2016.05.057
BindingDB Entry DOI: 10.7270/Q29G5PQT |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481128
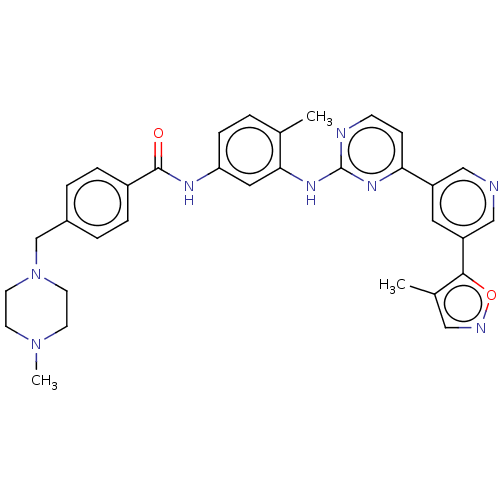
(US10906896, Cpd 809 | US11725005, Compound 809)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3oncc3C)c2)CC1 Show InChI InChI=1S/C33H34N8O2/c1-22-4-9-28(37-32(42)25-7-5-24(6-8-25)21-41-14-12-40(3)13-15-41)17-30(22)39-33-35-11-10-29(38-33)26-16-27(20-34-19-26)31-23(2)18-36-43-31/h4-11,16-20H,12-15,21H2,1-3H3,(H,37,42)(H,35,38,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| | n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481131

(US10906896, Cpd 8300 | US10906896, Cpd 832 | US117...)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3ccn(C)n3)c2)CC1 Show InChI InChI=1S/C33H35N9O/c1-23-4-9-28(36-32(43)25-7-5-24(6-8-25)22-42-16-14-40(2)15-17-42)19-31(23)38-33-35-12-10-29(37-33)26-18-27(21-34-20-26)30-11-13-41(3)39-30/h4-13,18-21H,14-17,22H2,1-3H3,(H,36,43)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481131

(US10906896, Cpd 8300 | US10906896, Cpd 832 | US117...)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3ccn(C)n3)c2)CC1 Show InChI InChI=1S/C33H35N9O/c1-23-4-9-28(36-32(43)25-7-5-24(6-8-25)22-42-16-14-40(2)15-17-42)19-31(23)38-33-35-12-10-29(37-33)26-18-27(21-34-20-26)30-11-13-41(3)39-30/h4-13,18-21H,14-17,22H2,1-3H3,(H,36,43)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481128

(US10906896, Cpd 809 | US11725005, Compound 809)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3oncc3C)c2)CC1 Show InChI InChI=1S/C33H34N8O2/c1-22-4-9-28(37-32(42)25-7-5-24(6-8-25)21-41-14-12-40(3)13-15-41)17-30(22)39-33-35-11-10-29(38-33)26-16-27(20-34-19-26)31-23(2)18-36-43-31/h4-11,16-20H,12-15,21H2,1-3H3,(H,37,42)(H,35,38,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM612767
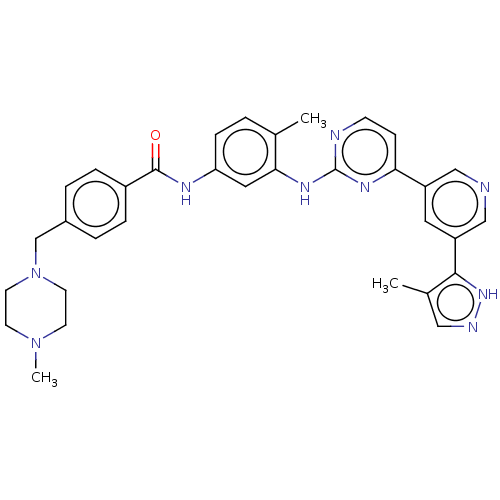
(US11725005, Compound 108)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3[nH]ncc3C)c2)CC1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 85.9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
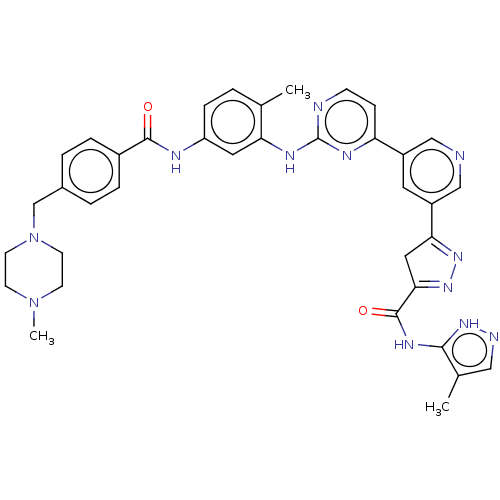
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481108
(US10906896, Cpd 108)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)C3=NN=C(C3)C(=O)Nc3[nH]ncc3C)c2)CC1 |c:39,t:37| Show InChI InChI=1S/C37H38N12O2/c1-23-4-9-29(41-35(50)26-7-5-25(6-8-26)22-49-14-12-48(3)13-15-49)17-31(23)43-37-39-11-10-30(42-37)27-16-28(21-38-20-27)32-18-33(46-45-32)36(51)44-34-24(2)19-40-47-34/h4-11,16-17,19-21H,12-15,18,22H2,1-3H3,(H,41,50)(H,39,42,43)(H2,40,44,47,51) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 85.9 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
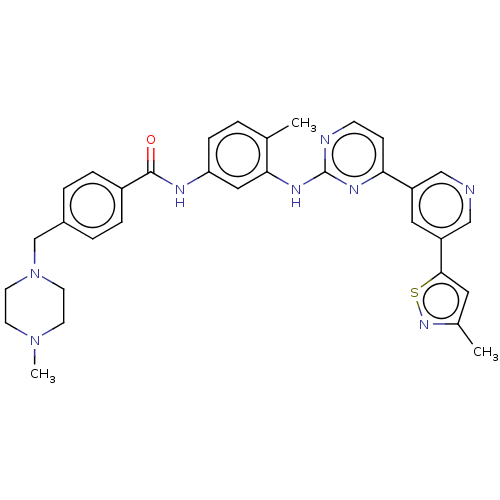
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481137
(US10906896, Cpd 8190 | US11725005, Compound 8190)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cc(C)ns3)c2)CC1 Show InChI InChI=1S/C33H34N8OS/c1-22-4-9-28(36-32(42)25-7-5-24(6-8-25)21-41-14-12-40(3)13-15-41)18-30(22)38-33-35-11-10-29(37-33)26-17-27(20-34-19-26)31-16-23(2)39-43-31/h4-11,16-20H,12-15,21H2,1-3H3,(H,36,42)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 91 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481137

(US10906896, Cpd 8190 | US11725005, Compound 8190)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3cc(C)ns3)c2)CC1 Show InChI InChI=1S/C33H34N8OS/c1-22-4-9-28(36-32(42)25-7-5-24(6-8-25)21-41-14-12-40(3)13-15-41)18-30(22)38-33-35-11-10-29(37-33)26-17-27(20-34-19-26)31-16-23(2)39-43-31/h4-11,16-20H,12-15,21H2,1-3H3,(H,36,42)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 91 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |
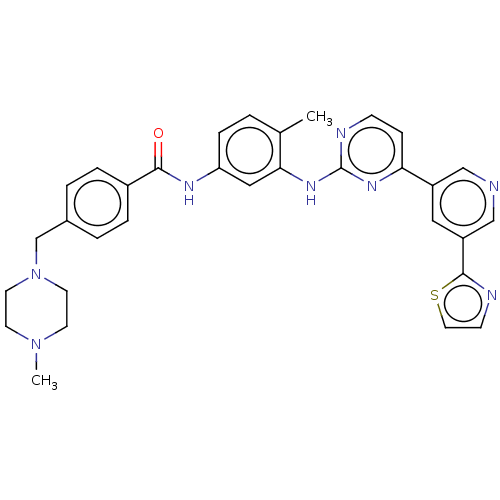
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481143
(US10906896, Cpd 8270 | US11725005, Compound 8270)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3nccs3)c2)CC1 Show InChI InChI=1S/C32H32N8OS/c1-22-3-8-27(36-30(41)24-6-4-23(5-7-24)21-40-14-12-39(2)13-15-40)18-29(22)38-32-35-10-9-28(37-32)25-17-26(20-33-19-25)31-34-11-16-42-31/h3-11,16-20H,12-15,21H2,1-2H3,(H,36,41)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2R215H9 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL2
(Homo sapiens (Human)) | BDBM481143

(US10906896, Cpd 8270 | US11725005, Compound 8270)Show SMILES CN1CCN(Cc2ccc(cc2)C(=O)Nc2ccc(C)c(Nc3nccc(n3)-c3cncc(c3)-c3nccs3)c2)CC1 Show InChI InChI=1S/C32H32N8OS/c1-22-3-8-27(36-30(41)24-6-4-23(5-7-24)21-40-14-12-39(2)13-15-40)18-29(22)38-32-35-10-9-28(37-32)25-17-26(20-33-19-25)31-34-11-16-42-31/h3-11,16-20H,12-15,21H2,1-2H3,(H,36,41)(H,35,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Inhibikase Therapeutics, Inc.
US Patent
| Assay Description
Kinase base buffer (50 mM HEPES, pH 7.5 0.0015% Brij-35; 10 mM MgCl2 2 mM DTT) and Stop buffer, (100 mM HEPES, pH 7.5 0.015% Brij-35; 0.2% Coating Re... |
US Patent US10906896 (2021)
BindingDB Entry DOI: 10.7270/Q2G44TCQ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data