 Found 233 hits of ic50 data for polymerid = 50004595
Found 233 hits of ic50 data for polymerid = 50004595 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364407
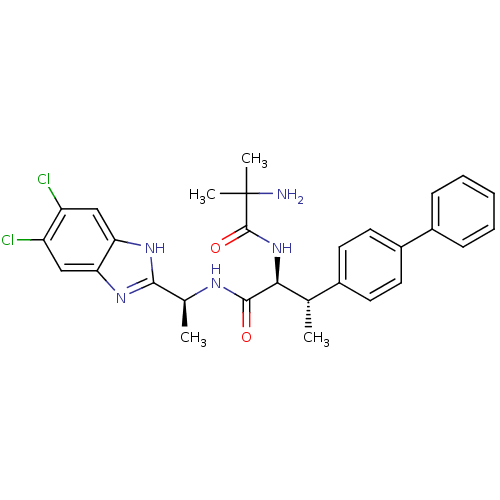
(CHEMBL1950444)Show SMILES C[C@H](NC(=O)[C@@H](NC(=O)C(C)(C)N)[C@@H](C)c1ccc(cc1)-c1ccccc1)c1nc2cc(Cl)c(Cl)cc2[nH]1 |r| Show InChI InChI=1S/C29H31Cl2N5O2/c1-16(18-10-12-20(13-11-18)19-8-6-5-7-9-19)25(36-28(38)29(3,4)32)27(37)33-17(2)26-34-23-14-21(30)22(31)15-24(23)35-26/h5-17,25H,32H2,1-4H3,(H,33,37)(H,34,35)(H,36,38)/t16-,17-,25-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0390 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse recombinant PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate for 30 mins by continuous fluorimetric assay |
Bioorg Med Chem Lett 22: 1774-8 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.064
BindingDB Entry DOI: 10.7270/Q27H1K28 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364689

(CHEMBL1951478)Show SMILES [O-][n+]1ccc(Cl)cc1C1OC2(CCN(CC2)S(=O)(=O)c2ccc(cn2)C(F)(F)F)c2ccccc12 Show InChI InChI=1S/C23H19ClF3N3O4S/c24-16-7-10-30(31)19(13-16)21-17-3-1-2-4-18(17)22(34-21)8-11-29(12-9-22)35(32,33)20-6-5-15(14-28-20)23(25,26)27/h1-7,10,13-14,21H,8-9,11-12H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364676
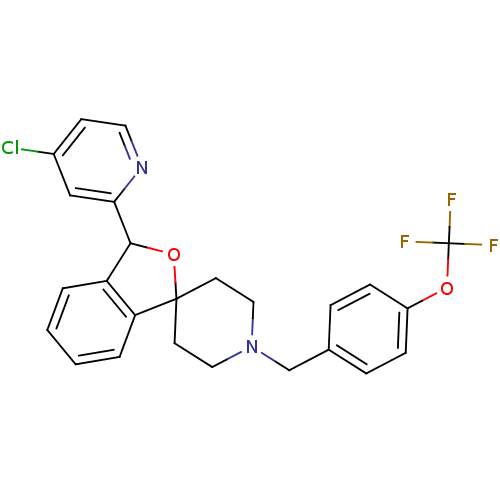
(CHEMBL1951465 | US8785634, 1)Show SMILES FC(F)(F)Oc1ccc(CN2CCC3(CC2)OC(c2ccccc32)c2cc(Cl)ccn2)cc1 Show InChI InChI=1S/C25H22ClF3N2O2/c26-18-9-12-30-22(15-18)23-20-3-1-2-4-21(20)24(33-23)10-13-31(14-11-24)16-17-5-7-19(8-6-17)32-25(27,28)29/h1-9,12,15,23H,10-11,13-14,16H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383421
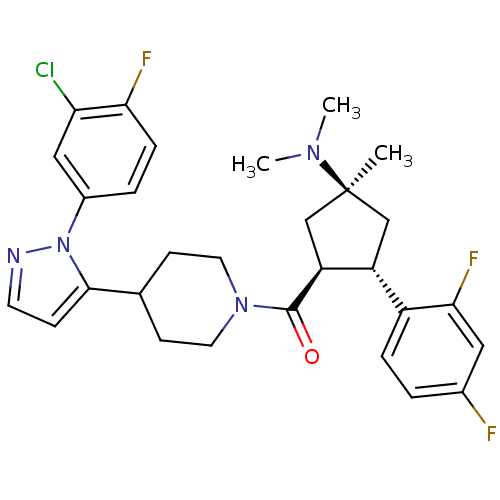
(CHEMBL2031595)Show SMILES CN(C)[C@]1(C)C[C@@H]([C@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1ccnn1-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C29H32ClF3N4O/c1-29(35(2)3)16-22(21-6-4-19(31)14-26(21)33)23(17-29)28(38)36-12-9-18(10-13-36)27-8-11-34-37(27)20-5-7-25(32)24(30)15-20/h4-8,11,14-15,18,22-23H,9-10,12-13,16-17H2,1-3H3/t22-,23+,29+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50443348
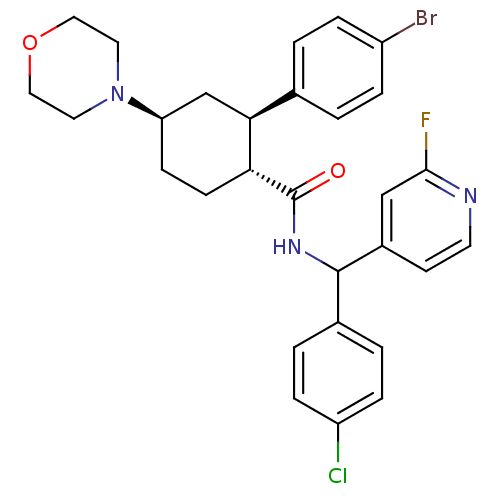
(CHEMBL3086040 | US8669252, 12)Show SMILES Fc1cc(ccn1)C(NC(=O)[C@@H]1CC[C@H](C[C@H]1c1ccc(Br)cc1)N1CCOCC1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H30BrClFN3O2/c30-22-5-1-19(2-6-22)26-18-24(35-13-15-37-16-14-35)9-10-25(26)29(36)34-28(20-3-7-23(31)8-4-20)21-11-12-33-27(32)17-21/h1-8,11-12,17,24-26,28H,9-10,13-16,18H2,(H,34,36)/t24-,25-,26+,28?/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 6228-33 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.094
BindingDB Entry DOI: 10.7270/Q2DZ09Q4 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382913
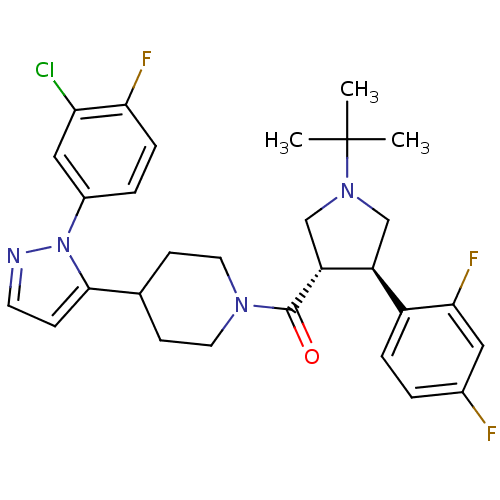
(CHEMBL2023210)Show SMILES CC(C)(C)N1C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1ccnn1-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C29H32ClF3N4O/c1-29(2,3)36-16-22(21-6-4-19(31)14-26(21)33)23(17-36)28(38)35-12-9-18(10-13-35)27-8-11-34-37(27)20-5-7-25(32)24(30)15-20/h4-8,11,14-15,18,22-23H,9-10,12-13,16-17H2,1-3H3/t22-,23+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364685
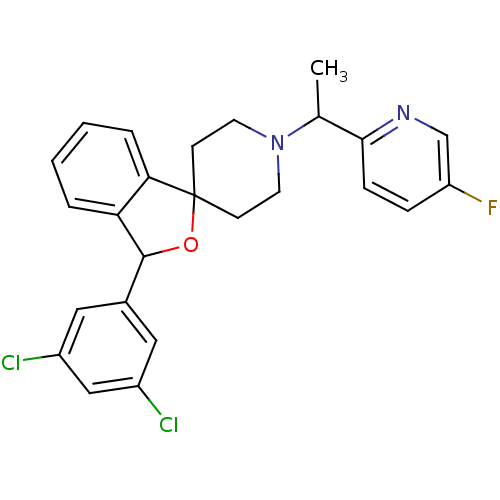
(CHEMBL1951474)Show SMILES CC(N1CCC2(CC1)OC(c1ccccc21)c1cc(Cl)cc(Cl)c1)c1ccc(F)cn1 Show InChI InChI=1S/C25H23Cl2FN2O/c1-16(23-7-6-20(28)15-29-23)30-10-8-25(9-11-30)22-5-3-2-4-21(22)24(31-25)17-12-18(26)14-19(27)13-17/h2-7,12-16,24H,8-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364679
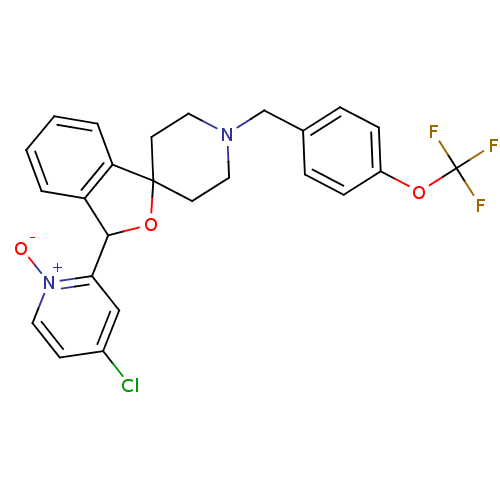
(CHEMBL1951468 | US8785634, 2)Show SMILES [O-][n+]1ccc(Cl)cc1C1OC2(CCN(Cc3ccc(OC(F)(F)F)cc3)CC2)c2ccccc12 Show InChI InChI=1S/C25H22ClF3N2O3/c26-18-9-12-31(32)22(15-18)23-20-3-1-2-4-21(20)24(34-23)10-13-30(14-11-24)16-17-5-7-19(8-6-17)33-25(27,28)29/h1-9,12,15,23H,10-11,13-14,16H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364687
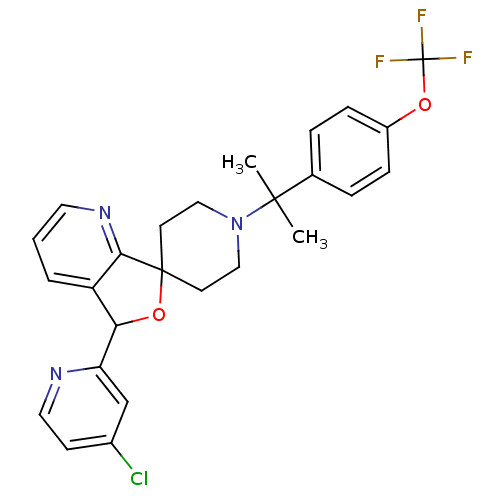
(CHEMBL1951476 | US8785634, 8)Show SMILES CC(C)(N1CCC2(CC1)OC(c1cccnc21)c1cc(Cl)ccn1)c1ccc(OC(F)(F)F)cc1 Show InChI InChI=1S/C26H25ClF3N3O2/c1-24(2,17-5-7-19(8-6-17)34-26(28,29)30)33-14-10-25(11-15-33)23-20(4-3-12-32-23)22(35-25)21-16-18(27)9-13-31-21/h3-9,12-13,16,22H,10-11,14-15H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382906

(CHEMBL2023202)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1cc(Cl)ccc1Cl |r| Show InChI InChI=1S/C30H34Cl2F2N4O/c1-18-13-27(38(35-18)28-14-20(31)5-8-25(28)32)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-6-21(33)15-26(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50361774
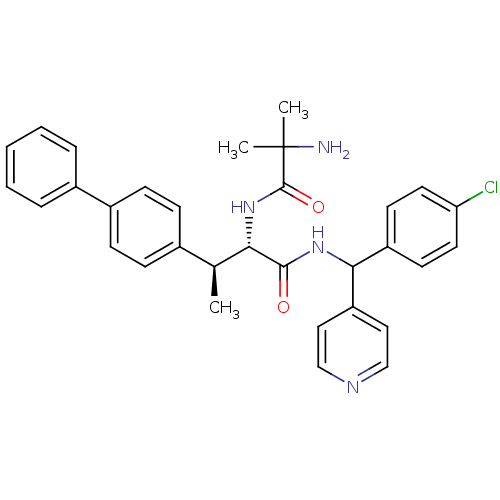
(CHEMBL1938510)Show SMILES C[C@H]([C@H](NC(=O)C(C)(C)N)C(=O)NC(c1ccncc1)c1ccc(Cl)cc1)c1ccc(cc1)-c1ccccc1 |r| Show InChI InChI=1S/C32H33ClN4O2/c1-21(22-9-11-24(12-10-22)23-7-5-4-6-8-23)28(37-31(39)32(2,3)34)30(38)36-29(26-17-19-35-20-18-26)25-13-15-27(33)16-14-25/h4-21,28-29H,34H2,1-3H3,(H,36,38)(H,37,39)/t21-,28-,29?/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 22: 658-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.10.060
BindingDB Entry DOI: 10.7270/Q2B56K50 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383427
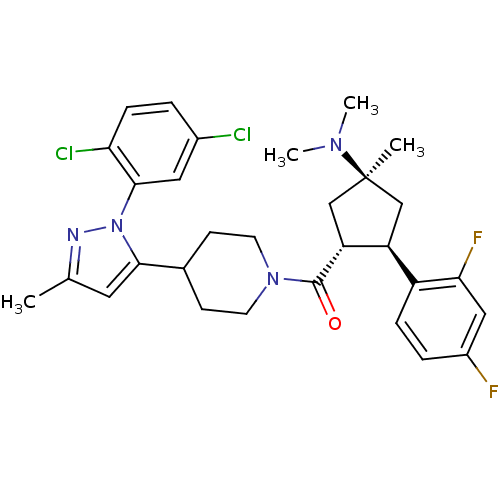
(CHEMBL2031588)Show SMILES CN(C)[C@]1(C)C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1cc(C)nn1-c1cc(Cl)ccc1Cl |r| Show InChI InChI=1S/C30H34Cl2F2N4O/c1-18-13-27(38(35-18)28-14-20(31)5-8-25(28)32)19-9-11-37(12-10-19)29(39)24-17-30(2,36(3)4)16-23(24)22-7-6-21(33)15-26(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+,30-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382906

(CHEMBL2023202)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1cc(Cl)ccc1Cl |r| Show InChI InChI=1S/C30H34Cl2F2N4O/c1-18-13-27(38(35-18)28-14-20(31)5-8-25(28)32)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-6-21(33)15-26(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50338033
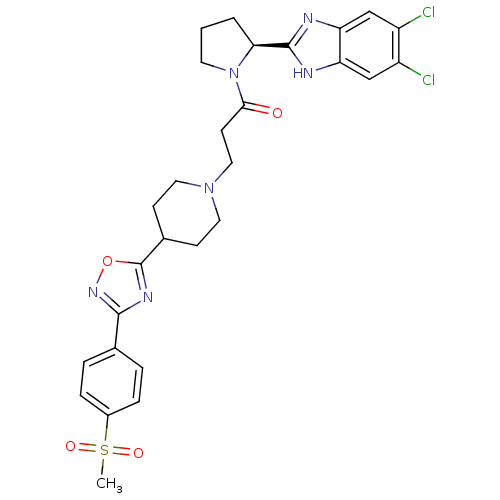
((S)-1-(2-(5,6-dichloro-1H-benzo[d]imidazol-2-yl)py...)Show SMILES CS(=O)(=O)c1ccc(cc1)-c1noc(n1)C1CCN(CCC(=O)N2CCC[C@H]2c2nc3cc(Cl)c(Cl)cc3[nH]2)CC1 |r| Show InChI InChI=1S/C28H30Cl2N6O4S/c1-41(38,39)19-6-4-17(5-7-19)26-33-28(40-34-26)18-8-12-35(13-9-18)14-10-25(37)36-11-2-3-24(36)27-31-22-15-20(29)21(30)16-23(22)32-27/h4-7,15-16,18,24H,2-3,8-14H2,1H3,(H,31,32)/t24-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.410 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 21: 1299-305 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.090
BindingDB Entry DOI: 10.7270/Q2VX0GSS |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383425
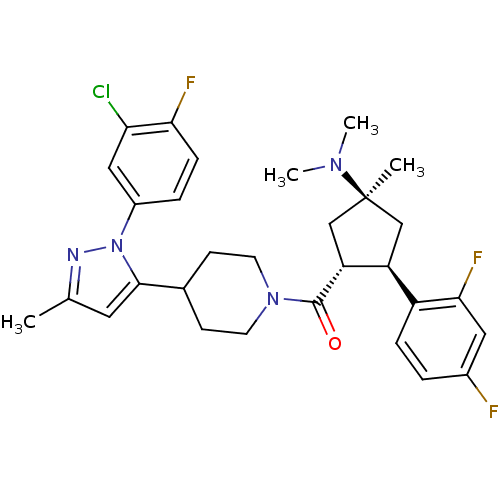
(CHEMBL2031590)Show SMILES CN(C)[C@]1(C)C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1cc(C)nn1-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C30H34ClF3N4O/c1-18-13-28(38(35-18)21-6-8-26(33)25(31)15-21)19-9-11-37(12-10-19)29(39)24-17-30(2,36(3)4)16-23(24)22-7-5-20(32)14-27(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+,30-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50338029
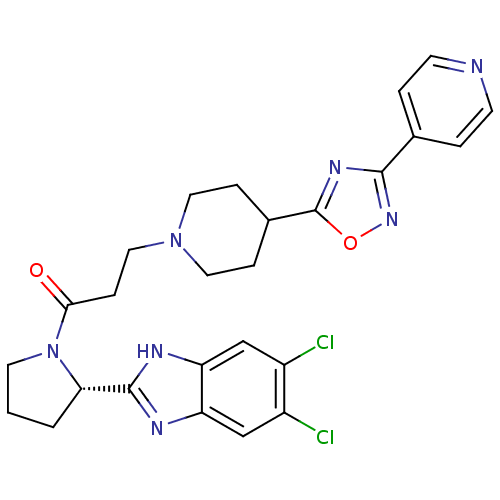
((S)-1-(2-(5,6-dichloro-1H-benzo[d]imidazol-2-yl)py...)Show SMILES Clc1cc2nc([nH]c2cc1Cl)[C@@H]1CCCN1C(=O)CCN1CCC(CC1)c1nc(no1)-c1ccncc1 |r| Show InChI InChI=1S/C26H27Cl2N7O2/c27-18-14-20-21(15-19(18)28)31-25(30-20)22-2-1-10-35(22)23(36)7-13-34-11-5-17(6-12-34)26-32-24(33-37-26)16-3-8-29-9-4-16/h3-4,8-9,14-15,17,22H,1-2,5-7,10-13H2,(H,30,31)/t22-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.530 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 21: 1299-305 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.090
BindingDB Entry DOI: 10.7270/Q2VX0GSS |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50001308
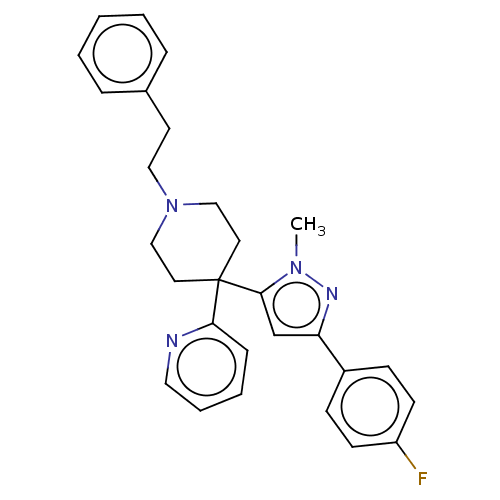
(CHEMBL3237393)Show SMILES OC(=O)C(F)(F)F.Cn1nc(cc1C1(CCN(CCc2ccccc2)CC1)c1ccccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C28H29FN4.C2HF3O2/c1-32-27(21-25(31-32)23-10-12-24(29)13-11-23)28(26-9-5-6-17-30-26)15-19-33(20-16-28)18-14-22-7-3-2-4-8-22;3-2(4,5)1(6)7/h2-13,17,21H,14-16,18-20H2,1H3;(H,6,7) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate after 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 24: 1657-60 (2014)
Article DOI: 10.1016/j.bmcl.2014.02.070
BindingDB Entry DOI: 10.7270/Q2GB25JD |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364669
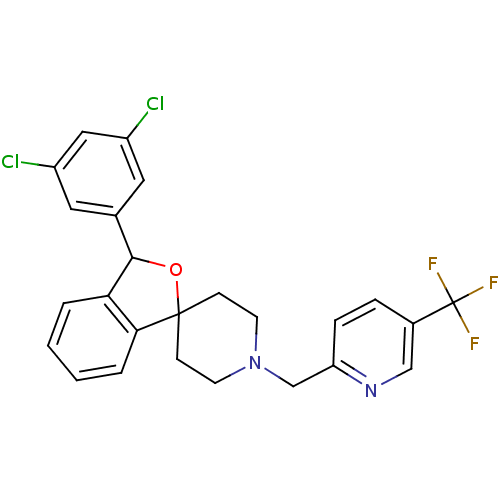
(CHEMBL1951458)Show SMILES FC(F)(F)c1ccc(CN2CCC3(CC2)OC(c2ccccc32)c2cc(Cl)cc(Cl)c2)nc1 Show InChI InChI=1S/C25H21Cl2F3N2O/c26-18-11-16(12-19(27)13-18)23-21-3-1-2-4-22(21)24(33-23)7-9-32(10-8-24)15-20-6-5-17(14-31-20)25(28,29)30/h1-6,11-14,23H,7-10,15H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50443351
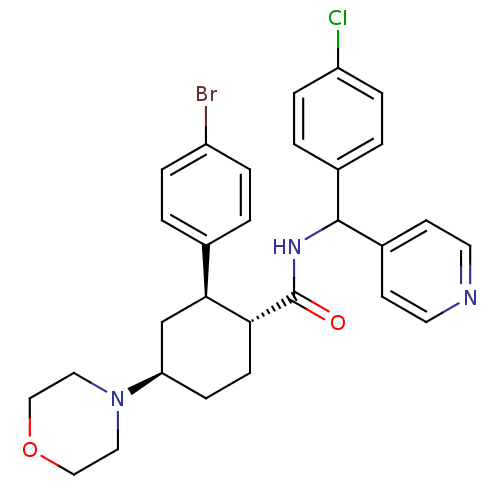
(CHEMBL3086037 | US8669252, 15)Show SMILES Clc1ccc(cc1)C(NC(=O)[C@@H]1CC[C@H](C[C@H]1c1ccc(Br)cc1)N1CCOCC1)c1ccncc1 |r| Show InChI InChI=1S/C29H31BrClN3O2/c30-23-5-1-20(2-6-23)27-19-25(34-15-17-36-18-16-34)9-10-26(27)29(35)33-28(22-11-13-32-14-12-22)21-3-7-24(31)8-4-21/h1-8,11-14,25-28H,9-10,15-19H2,(H,33,35)/t25-,26-,27+,28?/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.740 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 6228-33 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.094
BindingDB Entry DOI: 10.7270/Q2DZ09Q4 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50338007

((S)-1-(2-(5,6-dichloro-1H-benzo[d]imidazol-2-yl)py...)Show SMILES Cn1nc(cc1-c1ccncc1)C1CCN(CCC(=O)N2CCC[C@H]2c2nc3cc(Cl)c(Cl)cc3[nH]2)CC1 |r| Show InChI InChI=1S/C28H31Cl2N7O/c1-35-26(19-4-9-31-10-5-19)17-22(34-35)18-6-12-36(13-7-18)14-8-27(38)37-11-2-3-25(37)28-32-23-15-20(29)21(30)16-24(23)33-28/h4-5,9-10,15-18,25H,2-3,6-8,11-14H2,1H3,(H,32,33)/t25-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 21: 1299-305 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.090
BindingDB Entry DOI: 10.7270/Q2VX0GSS |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382912

(CHEMBL2023209 | US8569299, 4)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C30H34ClF3N4O/c1-18-13-28(38(35-18)21-6-8-26(33)25(31)15-21)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-5-20(32)14-27(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383426
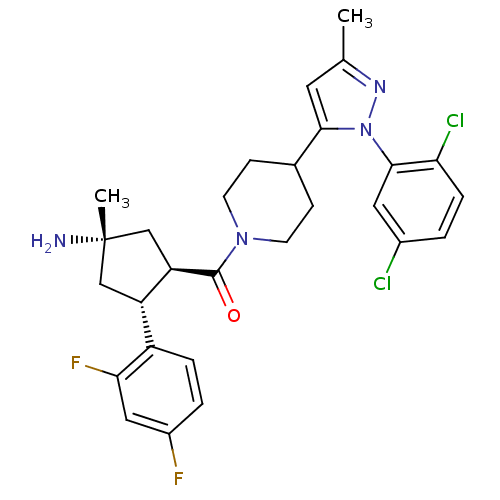
(CHEMBL2031589)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2C[C@@](C)(N)C[C@H]2c2ccc(F)cc2F)n(n1)-c1cc(Cl)ccc1Cl |r| Show InChI InChI=1S/C28H30Cl2F2N4O/c1-16-11-25(36(34-16)26-12-18(29)3-6-23(26)30)17-7-9-35(10-8-17)27(37)22-15-28(2,33)14-21(22)20-5-4-19(31)13-24(20)32/h3-6,11-13,17,21-22H,7-10,14-15,33H2,1-2H3/t21-,22+,28-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364681
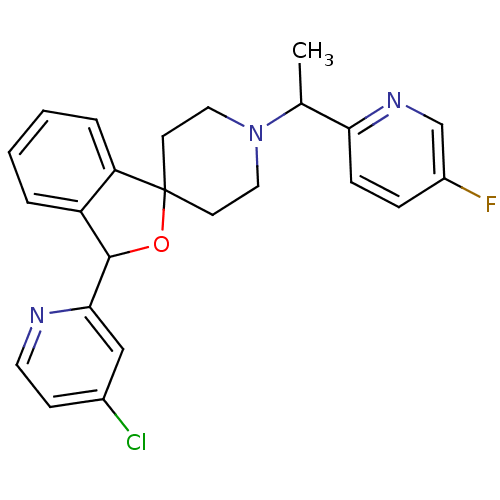
(CHEMBL1951470)Show SMILES CC(N1CCC2(CC1)OC(c1ccccc21)c1cc(Cl)ccn1)c1ccc(F)cn1 Show InChI InChI=1S/C24H23ClFN3O/c1-16(21-7-6-18(26)15-28-21)29-12-9-24(10-13-29)20-5-3-2-4-19(20)23(30-24)22-14-17(25)8-11-27-22/h2-8,11,14-16,23H,9-10,12-13H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364666
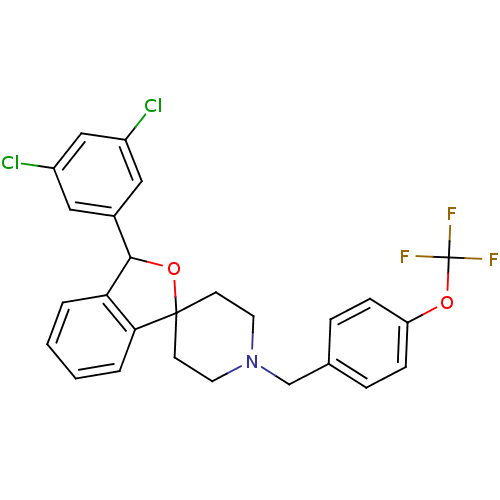
(CHEMBL1951456)Show SMILES FC(F)(F)Oc1ccc(CN2CCC3(CC2)OC(c2ccccc32)c2cc(Cl)cc(Cl)c2)cc1 Show InChI InChI=1S/C26H22Cl2F3NO2/c27-19-13-18(14-20(28)15-19)24-22-3-1-2-4-23(22)25(34-24)9-11-32(12-10-25)16-17-5-7-21(8-6-17)33-26(29,30)31/h1-8,13-15,24H,9-12,16H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382905

(CHEMBL2022794)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C30H34Cl2F2N4O/c1-18-13-28(38(35-18)27-8-5-20(31)14-25(27)32)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-6-21(33)15-26(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382904
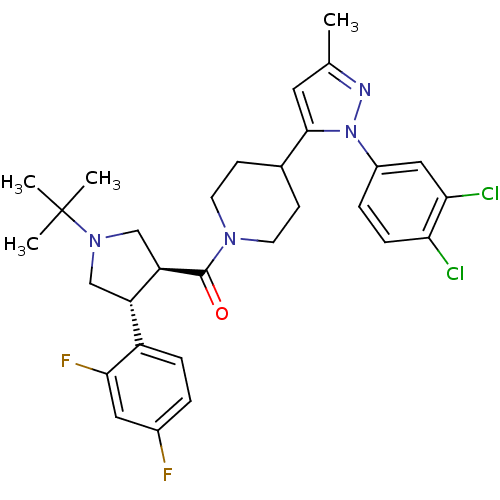
(CHEMBL2022793)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C30H34Cl2F2N4O/c1-18-13-28(38(35-18)21-6-8-25(31)26(32)15-21)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-5-20(33)14-27(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383419
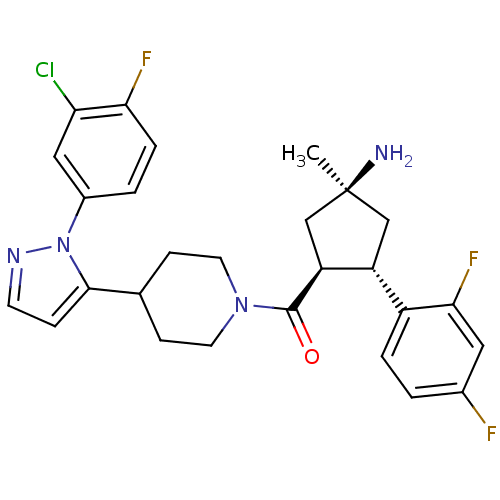
(CHEMBL2031597)Show SMILES C[C@@]1(N)C[C@@H]([C@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1ccnn1-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C27H28ClF3N4O/c1-27(32)14-20(19-4-2-17(29)12-24(19)31)21(15-27)26(36)34-10-7-16(8-11-34)25-6-9-33-35(25)18-3-5-23(30)22(28)13-18/h2-6,9,12-13,16,20-21H,7-8,10-11,14-15,32H2,1H3/t20-,21+,27+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364684

(CHEMBL1951473)Show SMILES CC(N1CCC2(CC1)OC(c1cccnc21)c1cc(Cl)cc(Cl)c1)c1ccc(F)cn1 Show InChI InChI=1S/C24H22Cl2FN3O/c1-15(21-5-4-19(27)14-29-21)30-9-6-24(7-10-30)23-20(3-2-8-28-23)22(31-24)16-11-17(25)13-18(26)12-16/h2-5,8,11-15,22H,6-7,9-10H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382899

(CHEMBL2022787)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1ccc(C)c(Cl)c1 |r| Show InChI InChI=1S/C31H37ClF2N4O/c1-19-6-8-23(16-27(19)32)38-29(14-20(2)35-38)21-10-12-36(13-11-21)30(39)26-18-37(31(3,4)5)17-25(26)24-9-7-22(33)15-28(24)34/h6-9,14-16,21,25-26H,10-13,17-18H2,1-5H3/t25-,26+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50443343
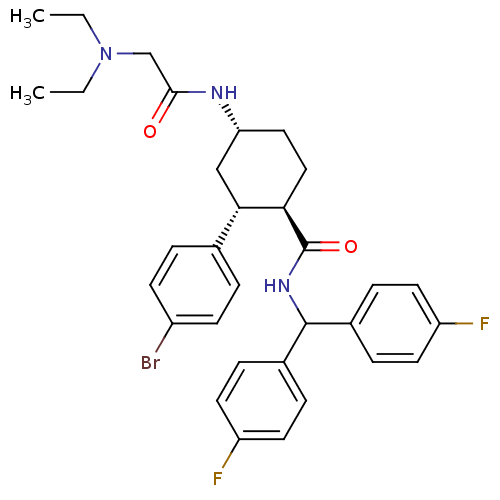
(CHEMBL3086028 | US8669252, 56)Show SMILES CCN(CC)CC(=O)N[C@@H]1CC[C@H]([C@@H](C1)c1ccc(Br)cc1)C(=O)NC(c1ccc(F)cc1)c1ccc(F)cc1 |r| Show InChI InChI=1S/C32H36BrF2N3O2/c1-3-38(4-2)20-30(39)36-27-17-18-28(29(19-27)21-5-11-24(33)12-6-21)32(40)37-31(22-7-13-25(34)14-8-22)23-9-15-26(35)16-10-23/h5-16,27-29,31H,3-4,17-20H2,1-2H3,(H,36,39)(H,37,40)/t27-,28-,29+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 6228-33 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.094
BindingDB Entry DOI: 10.7270/Q2DZ09Q4 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364673
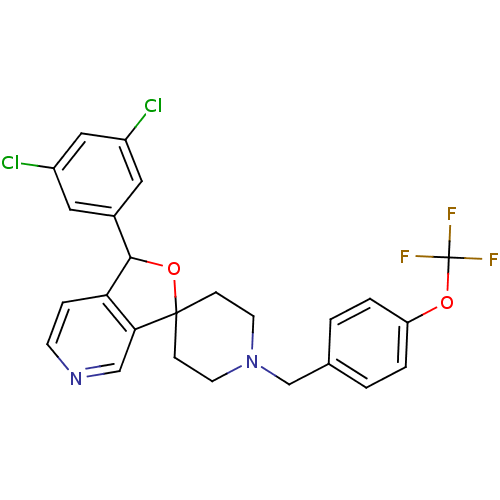
(CHEMBL1951462)Show SMILES FC(F)(F)Oc1ccc(CN2CCC3(CC2)OC(c2ccncc32)c2cc(Cl)cc(Cl)c2)cc1 Show InChI InChI=1S/C25H21Cl2F3N2O2/c26-18-11-17(12-19(27)13-18)23-21-5-8-31-14-22(21)24(34-23)6-9-32(10-7-24)15-16-1-3-20(4-2-16)33-25(28,29)30/h1-5,8,11-14,23H,6-7,9-10,15H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50443344

(CHEMBL3086045)Show SMILES Cc1ccccc1[C@H]1C[C@H](CC[C@@H]1C(=O)N1CC(C1)(c1ccccc1)c1ccccc1)NCc1ccc(Cl)cc1 |r| Show InChI InChI=1S/C36H37ClN2O/c1-26-10-8-9-15-32(26)34-22-31(38-23-27-16-18-30(37)19-17-27)20-21-33(34)35(40)39-24-36(25-39,28-11-4-2-5-12-28)29-13-6-3-7-14-29/h2-19,31,33-34,38H,20-25H2,1H3/t31-,33-,34+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 6228-33 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.094
BindingDB Entry DOI: 10.7270/Q2DZ09Q4 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50443353
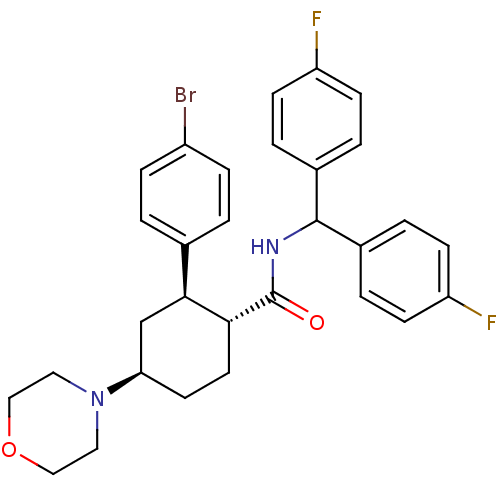
(CHEMBL3086036 | US8669252, 14)Show SMILES Fc1ccc(cc1)C(NC(=O)[C@@H]1CC[C@H](C[C@H]1c1ccc(Br)cc1)N1CCOCC1)c1ccc(F)cc1 |r| Show InChI InChI=1S/C30H31BrF2N2O2/c31-23-7-1-20(2-8-23)28-19-26(35-15-17-37-18-16-35)13-14-27(28)30(36)34-29(21-3-9-24(32)10-4-21)22-5-11-25(33)12-6-22/h1-12,26-29H,13-19H2,(H,34,36)/t26-,27-,28+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 6228-33 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.094
BindingDB Entry DOI: 10.7270/Q2DZ09Q4 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50361773

(CHEMBL1938509)Show SMILES C[C@H]([C@H](N)C(=O)NC(c1ccncc1)c1ccc(Cl)cc1)c1ccc(cc1)-c1ccccc1 |r| Show InChI InChI=1S/C28H26ClN3O/c1-19(20-7-9-22(10-8-20)21-5-3-2-4-6-21)26(30)28(33)32-27(24-15-17-31-18-16-24)23-11-13-25(29)14-12-23/h2-19,26-27H,30H2,1H3,(H,32,33)/t19-,26-,27?/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 22: 658-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.10.060
BindingDB Entry DOI: 10.7270/Q2B56K50 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364672
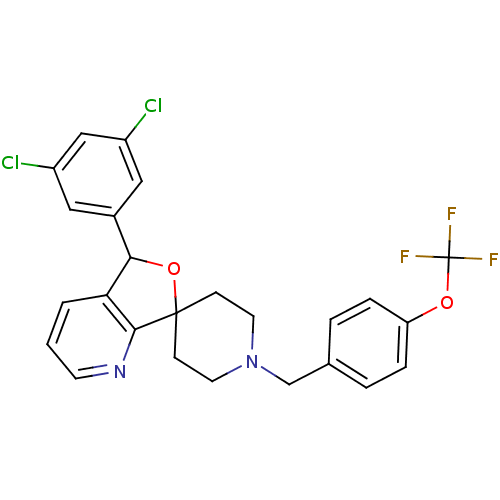
(CHEMBL1951461 | US8785634, 3)Show SMILES FC(F)(F)Oc1ccc(CN2CCC3(CC2)OC(c2cccnc32)c2cc(Cl)cc(Cl)c2)cc1 Show InChI InChI=1S/C25H21Cl2F3N2O2/c26-18-12-17(13-19(27)14-18)22-21-2-1-9-31-23(21)24(34-22)7-10-32(11-8-24)15-16-3-5-20(6-4-16)33-25(28,29)30/h1-6,9,12-14,22H,7-8,10-11,15H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383420
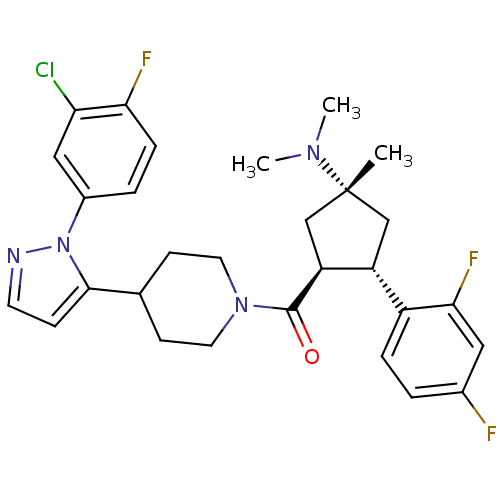
(CHEMBL2031596)Show SMILES CN(C)[C@@]1(C)C[C@@H]([C@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1ccnn1-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C29H32ClF3N4O/c1-29(35(2)3)16-22(21-6-4-19(31)14-26(21)33)23(17-29)28(38)36-12-9-18(10-13-36)27-8-11-34-37(27)20-5-7-25(32)24(30)15-20/h4-8,11,14-15,18,22-23H,9-10,12-13,16-17H2,1-3H3/t22-,23+,29-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382914

(CHEMBL2023211)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1cc(Cl)ccc1F |r| Show InChI InChI=1S/C30H34ClF3N4O/c1-18-13-27(38(35-18)28-14-20(31)5-8-25(28)33)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-6-21(32)15-26(22)34/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50443347
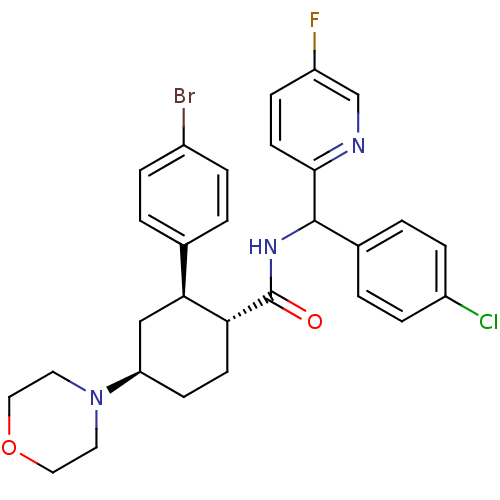
(CHEMBL3086041 | US8669252, 28)Show SMILES Fc1ccc(nc1)C(NC(=O)[C@@H]1CC[C@H](C[C@H]1c1ccc(Br)cc1)N1CCOCC1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C29H30BrClFN3O2/c30-21-5-1-19(2-6-21)26-17-24(35-13-15-37-16-14-35)10-11-25(26)29(36)34-28(20-3-7-22(31)8-4-20)27-12-9-23(32)18-33-27/h1-9,12,18,24-26,28H,10-11,13-17H2,(H,34,36)/t24-,25-,26+,28?/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 6228-33 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.094
BindingDB Entry DOI: 10.7270/Q2DZ09Q4 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50364682

(CHEMBL1951471 | US8785634, 82)Show SMILES FC(F)(F)Oc1ccc(cc1)S(=O)(=O)N1CCC2(CC1)OC(c1cccnc21)c1cc(Cl)cc(Cl)c1 Show InChI InChI=1S/C24H19Cl2F3N2O4S/c25-16-12-15(13-17(26)14-16)21-20-2-1-9-30-22(20)23(35-21)7-10-31(11-8-23)36(32,33)19-5-3-18(4-6-19)34-24(27,28)29/h1-6,9,12-14,21H,7-8,10-11H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 1550-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.002
BindingDB Entry DOI: 10.7270/Q2JS9QXQ |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383430
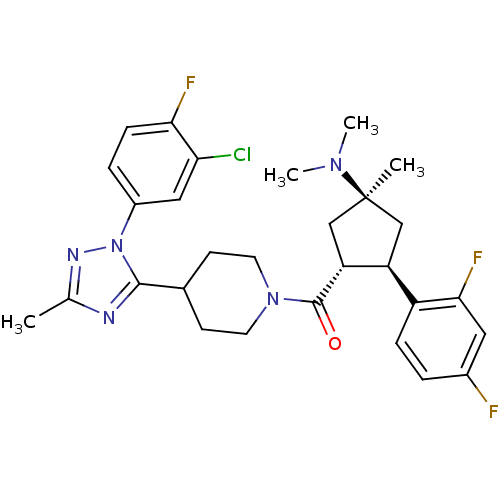
(CHEMBL2031584)Show SMILES CN(C)[C@]1(C)C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1nc(C)nn1-c1ccc(F)c(Cl)c1 |r| Show InChI InChI=1S/C29H33ClF3N5O/c1-17-34-27(38(35-17)20-6-8-25(32)24(30)14-20)18-9-11-37(12-10-18)28(39)23-16-29(2,36(3)4)15-22(23)21-7-5-19(31)13-26(21)33/h5-8,13-14,18,22-23H,9-12,15-16H2,1-4H3/t22-,23+,29-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50328523
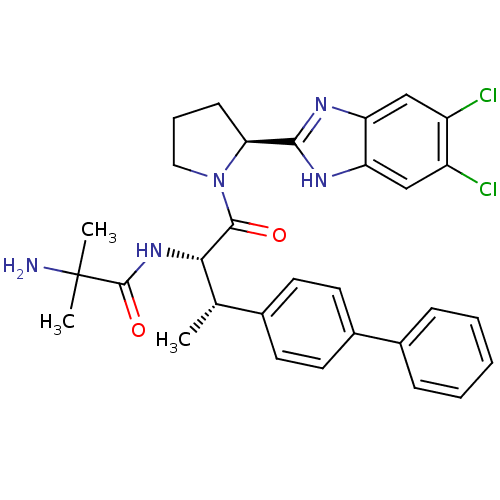
(2-Amino-N-{(2S,3S)-3-(biphenyl-4-yl)-1-[(2S)-2-(5,...)Show SMILES C[C@H]([C@H](NC(=O)C(C)(C)N)C(=O)N1CCC[C@H]1c1nc2cc(Cl)c(Cl)cc2[nH]1)c1ccc(cc1)-c1ccccc1 |r| Show InChI InChI=1S/C31H33Cl2N5O2/c1-18(19-11-13-21(14-12-19)20-8-5-4-6-9-20)27(37-30(40)31(2,3)34)29(39)38-15-7-10-26(38)28-35-24-16-22(32)23(33)17-25(24)36-28/h4-6,8-9,11-14,16-18,26-27H,7,10,15,34H2,1-3H3,(H,35,36)(H,37,40)/t18-,26-,27-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP by FRET |
J Med Chem 53: 7251-63 (2010)
Article DOI: 10.1021/jm101013m
BindingDB Entry DOI: 10.7270/Q2QV3MQR |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383433
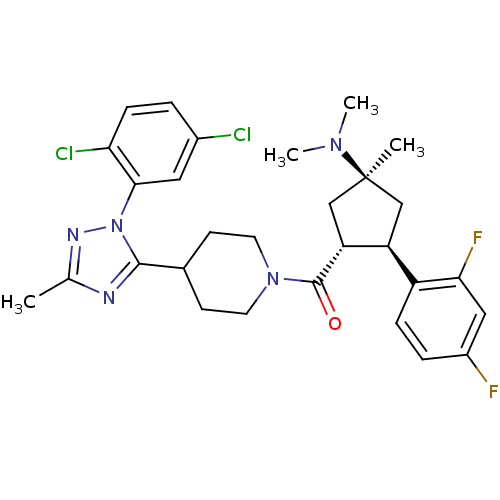
(CHEMBL2031582)Show SMILES CN(C)[C@]1(C)C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1nc(C)nn1-c1cc(Cl)ccc1Cl |r| Show InChI InChI=1S/C29H33Cl2F2N5O/c1-17-34-27(38(35-17)26-13-19(30)5-8-24(26)31)18-9-11-37(12-10-18)28(39)23-16-29(2,36(3)4)15-22(23)21-7-6-20(32)14-25(21)33/h5-8,13-14,18,22-23H,9-12,15-16H2,1-4H3/t22-,23+,29-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382910
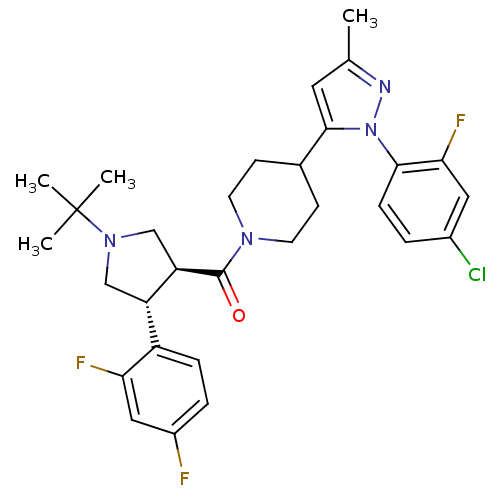
(CHEMBL2023207)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1ccc(Cl)cc1F |r| Show InChI InChI=1S/C30H34ClF3N4O/c1-18-13-28(38(35-18)27-8-5-20(31)14-26(27)34)19-9-11-36(12-10-19)29(39)24-17-37(30(2,3)4)16-23(24)22-7-6-21(32)15-25(22)33/h5-8,13-15,19,23-24H,9-12,16-17H2,1-4H3/t23-,24+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50328523

(2-Amino-N-{(2S,3S)-3-(biphenyl-4-yl)-1-[(2S)-2-(5,...)Show SMILES C[C@H]([C@H](NC(=O)C(C)(C)N)C(=O)N1CCC[C@H]1c1nc2cc(Cl)c(Cl)cc2[nH]1)c1ccc(cc1)-c1ccccc1 |r| Show InChI InChI=1S/C31H33Cl2N5O2/c1-18(19-11-13-21(14-12-19)20-8-5-4-6-9-20)27(37-30(40)31(2,3)34)29(39)38-15-7-10-26(38)28-35-24-16-22(32)23(33)17-25(24)36-28/h4-6,8-9,11-14,16-18,26-27H,7,10,15,34H2,1-3H3,(H,35,36)(H,37,40)/t18-,26-,27-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 22: 658-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.10.060
BindingDB Entry DOI: 10.7270/Q2B56K50 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50344263
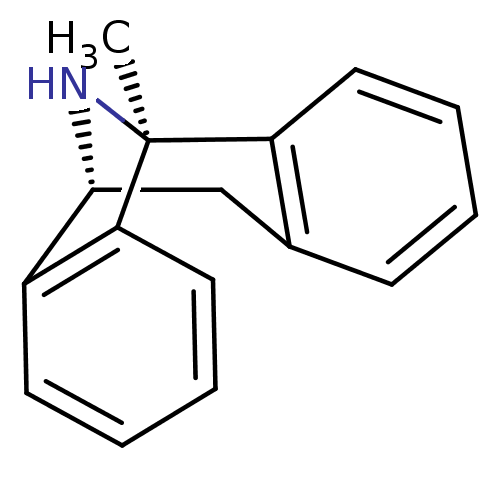
((+)-1-methyl-16-azatetracyclo[7.6.1.02,7.010,15]he...)Show InChI InChI=1S/C16H15N/c1-16-13-8-4-2-6-11(13)10-15(17-16)12-7-3-5-9-14(12)16/h2-9,15,17H,10H2,1H3/t15-,16+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Urbino Carlo Bo
Curated by ChEMBL
| Assay Description
Inhibitory concentration against human Adenosine A3 receptor expressed in HEK293 cells using 0.1 nM [3H]AB-MECA |
J Med Chem 48: 6887-96 (2005)
Article DOI: 10.1021/jm058018d
BindingDB Entry DOI: 10.7270/Q28C9X1X |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50361786
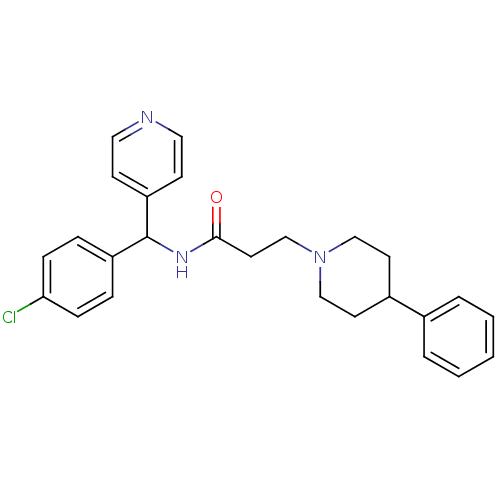
(CHEMBL1938522)Show SMILES Clc1ccc(cc1)C(NC(=O)CCN1CCC(CC1)c1ccccc1)c1ccncc1 Show InChI InChI=1S/C26H28ClN3O/c27-24-8-6-22(7-9-24)26(23-10-15-28-16-11-23)29-25(31)14-19-30-17-12-21(13-18-30)20-4-2-1-3-5-20/h1-11,15-16,21,26H,12-14,17-19H2,(H,29,31) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 22: 658-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.10.060
BindingDB Entry DOI: 10.7270/Q2B56K50 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50001318
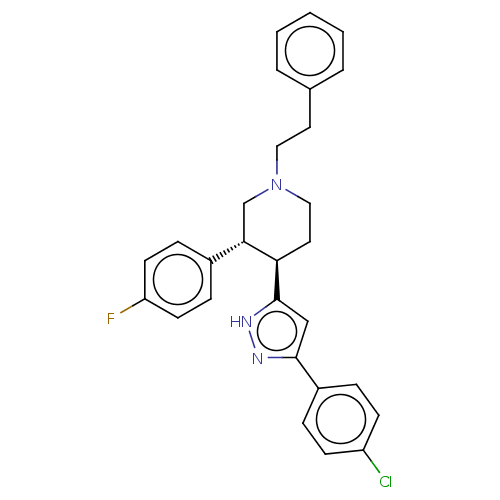
(CHEMBL3237377)Show SMILES OC(=O)C(F)(F)F.Fc1ccc(cc1)[C@@H]1CN(CCc2ccccc2)CC[C@H]1c1cc(n[nH]1)-c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C28H27ClFN3.C2HF3O2/c29-23-10-6-22(7-11-23)27-18-28(32-31-27)25-15-17-33(16-14-20-4-2-1-3-5-20)19-26(25)21-8-12-24(30)13-9-21;3-2(4,5)1(6)7/h1-13,18,25-26H,14-17,19H2,(H,31,32);(H,6,7)/t25-,26+;/m1./s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate after 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 24: 1657-60 (2014)
Article DOI: 10.1016/j.bmcl.2014.02.070
BindingDB Entry DOI: 10.7270/Q2GB25JD |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50361766

(CHEMBL1938502)Show SMILES C[C@H](NC(=O)[C@@H](NC(=O)C(C)(C)N)[C@@H](C)c1ccc(cc1)-c1ccccc1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C28H32ClN3O2/c1-18(20-10-12-23(13-11-20)22-8-6-5-7-9-22)25(32-27(34)28(3,4)30)26(33)31-19(2)21-14-16-24(29)17-15-21/h5-19,25H,30H2,1-4H3,(H,31,33)(H,32,34)/t18-,19-,25-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mouse PrCP using Mca-Ala-Pro-Lys(Dnp)-OH as substrate measured for 30 mins by continuous fluorometric assay |
Bioorg Med Chem Lett 22: 658-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.10.060
BindingDB Entry DOI: 10.7270/Q2B56K50 |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50382898
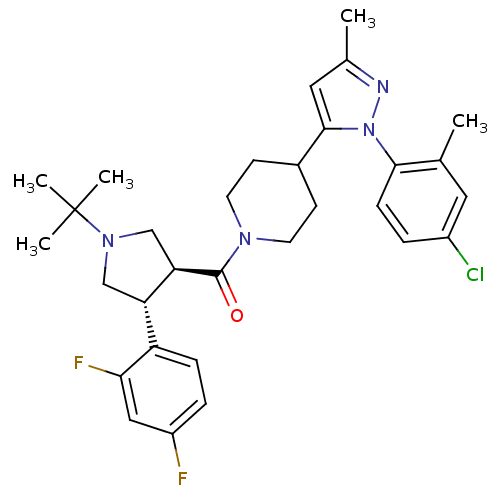
(CHEMBL2022786)Show SMILES Cc1cc(C2CCN(CC2)C(=O)[C@@H]2CN(C[C@H]2c2ccc(F)cc2F)C(C)(C)C)n(n1)-c1ccc(Cl)cc1C |r| Show InChI InChI=1S/C31H37ClF2N4O/c1-19-14-22(32)6-9-28(19)38-29(15-20(2)35-38)21-10-12-36(13-11-21)30(39)26-18-37(31(3,4)5)17-25(26)24-8-7-23(33)16-27(24)34/h6-9,14-16,21,25-26H,10-13,17-18H2,1-5H3/t25-,26+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2811-7 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.075
BindingDB Entry DOI: 10.7270/Q2T154PX |
More data for this
Ligand-Target Pair | |
Lysosomal Pro-X carboxypeptidase
(Mus musculus) | BDBM50383435
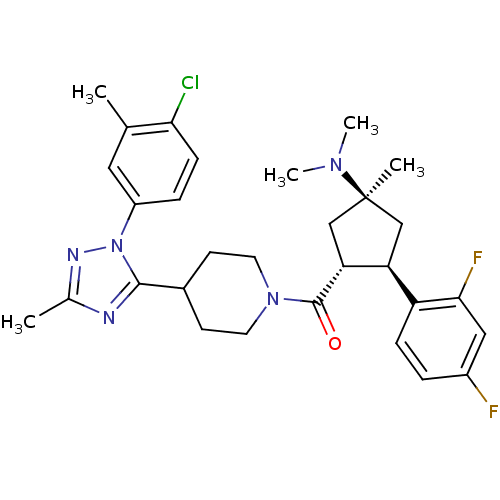
(CHEMBL2031580)Show SMILES CN(C)[C@]1(C)C[C@H]([C@@H](C1)c1ccc(F)cc1F)C(=O)N1CCC(CC1)c1nc(C)nn1-c1ccc(Cl)c(C)c1 |r| Show InChI InChI=1S/C30H36ClF2N5O/c1-18-14-22(7-9-26(18)31)38-28(34-19(2)35-38)20-10-12-37(13-11-20)29(39)25-17-30(3,36(4)5)16-24(25)23-8-6-21(32)15-27(23)33/h6-9,14-15,20,24-25H,10-13,16-17H2,1-5H3/t24-,25+,30-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of mouse PrCP |
Bioorg Med Chem Lett 22: 2818-22 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.077
BindingDB Entry DOI: 10.7270/Q2J967D7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data