

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosylhomocysteinase (Mus musculus) | BDBM50006222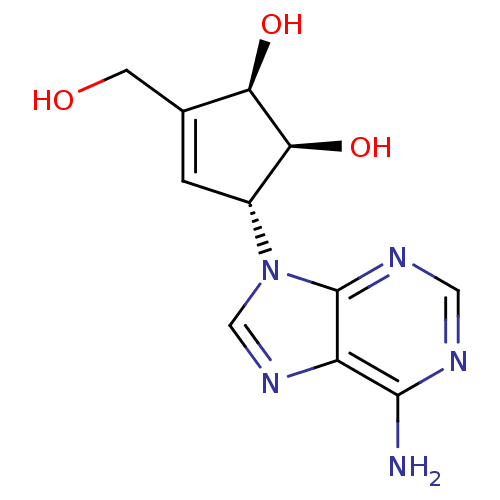 ((1S,2R,5R)-5-(6-Amino-purin-9-yl)-3-hydroxymethyl-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Florida Curated by ChEMBL | Assay Description Binding affinity of the compound was tested against L929 cells AdoHcy hydrolase activity | J Med Chem 37: 551-4 (1994) BindingDB Entry DOI: 10.7270/Q2DJ5DQ4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50034176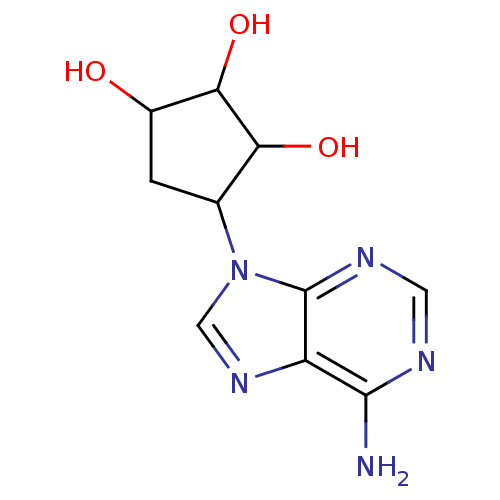 (4-(6-Amino-purin-9-yl)-cyclopentane-1,2,3-triol | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB PubMed | 11.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of South Florida Curated by ChEMBL | Assay Description Binding affinity of the compound was tested against L929 cells AdoHcy hydrolase activity | J Med Chem 37: 551-4 (1994) BindingDB Entry DOI: 10.7270/Q2DJ5DQ4 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50008288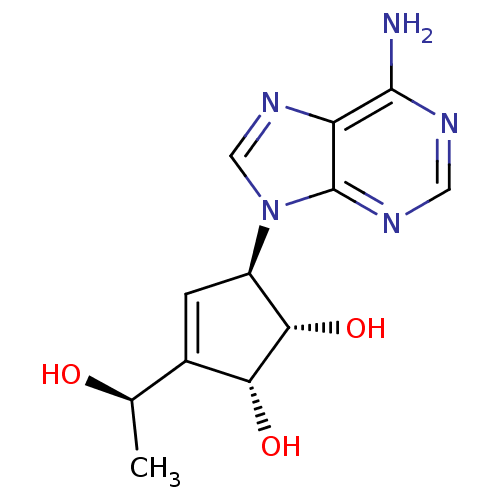 ((1S,2R,5R)-5-(6-Amino-purin-9-yl)-3-((R)-1-hydroxy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 86 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toyo Jozo Company, Ltd. Curated by ChEMBL | Assay Description Inhibitory effect of the compound against L929 Cell S-adenosyl-L-homocysteine hydrolase | J Med Chem 35: 324-31 (1992) BindingDB Entry DOI: 10.7270/Q2RX9B1R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50006223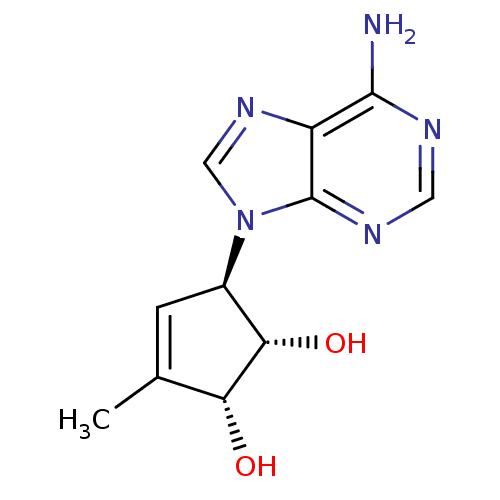 (5-(6-Amino-purin-9-yl)-3-methyl-cyclopent-3-ene-1,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toyo Jozo Company, Ltd. Curated by ChEMBL | Assay Description Inhibitory effect of the compound against L929 Cell S-adenosyl-L-homocysteine hydrolase | J Med Chem 35: 324-31 (1992) BindingDB Entry DOI: 10.7270/Q2RX9B1R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50280299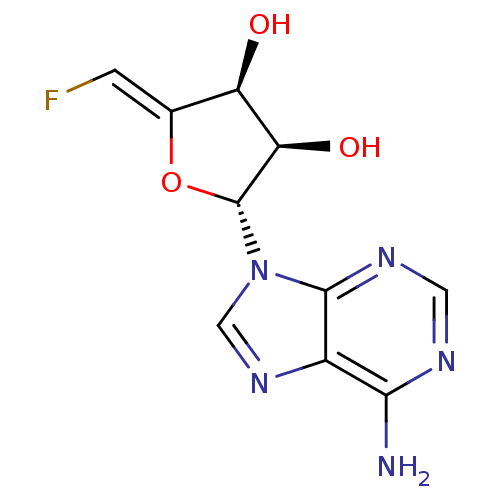 ((2R,3R,4S)-2-(6-Amino-purin-9-yl)-5-[1-fluoro-meth...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Activity determined in mouse liver S-adenosyl-L-homocysteine hydrolase and expressed as KI values. | J Med Chem 34: 647-56 (1991) BindingDB Entry DOI: 10.7270/Q25M6695 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50280301 ((2R,3R,4S)-2-(6-Amino-purin-9-yl)-5-[1-fluoro-meth...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Activity determined in mouse liver S-adenosyl-L-homocysteine hydrolase and expressed as KI values. | J Med Chem 34: 647-56 (1991) BindingDB Entry DOI: 10.7270/Q25M6695 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50368168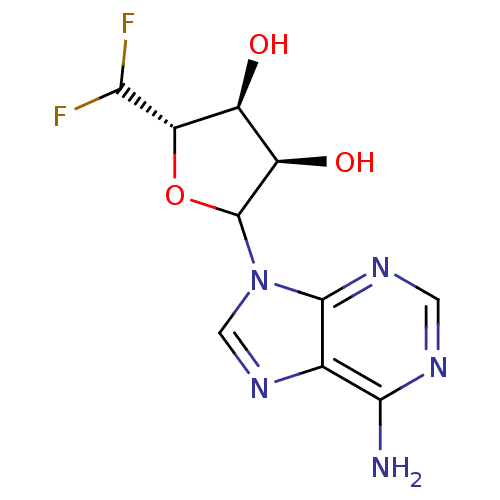 (CHEMBL609353) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Activity determined in mouse liver S-adenosyl-L-homocysteine hydrolase and expressed as KI values. | J Med Chem 34: 647-56 (1991) BindingDB Entry DOI: 10.7270/Q25M6695 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50144936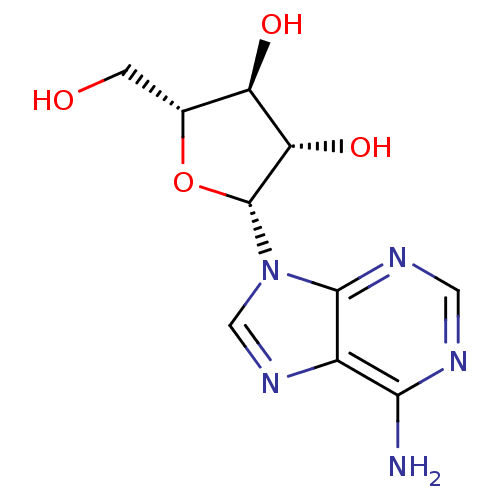 (CHEMBL1090 | VIDARABINE | adenine arabinoside) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merrell Dow Research Institute Curated by ChEMBL | Assay Description Activity determined in mouse liver S-adenosyl-L-homocysteine hydrolase and expressed as KI values. | J Med Chem 34: 647-56 (1991) BindingDB Entry DOI: 10.7270/Q25M6695 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosylhomocysteinase (Mus musculus) | BDBM50008289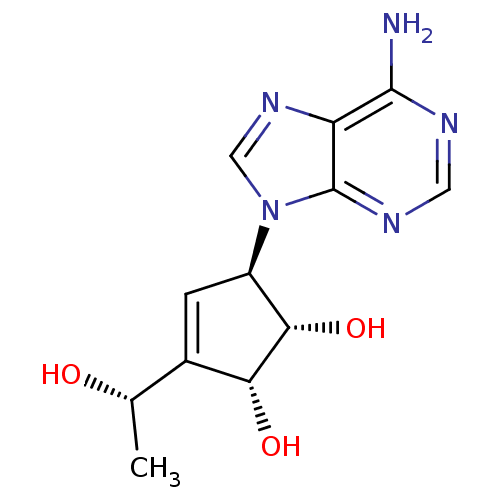 ((1S,2R,5R)-5-(6-Amino-purin-9-yl)-3-((S)-1-hydroxy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 5.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toyo Jozo Company, Ltd. Curated by ChEMBL | Assay Description Inhibitory effect of the compound against L929 Cell S-adenosyl-L-homocysteine hydrolase | J Med Chem 35: 324-31 (1992) BindingDB Entry DOI: 10.7270/Q2RX9B1R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||