

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide synthase, brain (409/422 = 97%)† (Homo sapiens (Human)) | BDBM50341682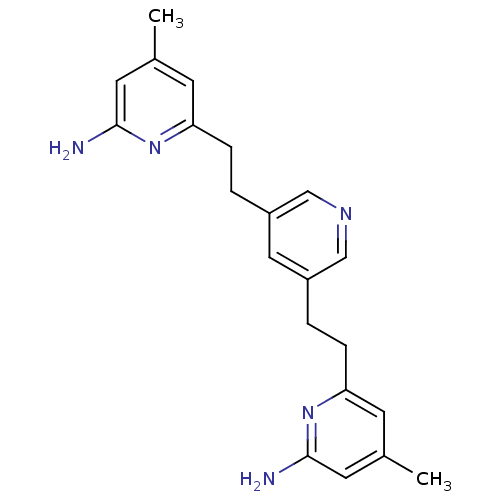 (6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California, Irvine | Assay Description Inhibition assay using nitric oxide synthases. | Biochemistry 49: 10803-10 (2010) Article DOI: 10.1021/bi1013479 BindingDB Entry DOI: 10.7270/Q2KS6Q51 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, brain (409/422 = 97%)† (Homo sapiens (Human)) | BDBM50341682 (6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, brain (422/422 = 100%)† (Rattus norvegicus (rat)) | BDBM50341682 (6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of rat recombinant nNOS expressed in Escherichia coli by UV-vis spectrometric analysis | J Med Chem 54: 2039-48 (2011) Article DOI: 10.1021/jm101071n BindingDB Entry DOI: 10.7270/Q2571CB0 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, brain (409/422 = 97%)† (Homo sapiens (Human)) | BDBM50341682 (6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of nNOS expressed in human 293T cells assessed as nitrite formation after 8 hrs by Griess reaction | J Med Chem 54: 2039-48 (2011) Article DOI: 10.1021/jm101071n BindingDB Entry DOI: 10.7270/Q2571CB0 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||