 Found 5 hits Enzyme Inhibition Constant Data
Found 5 hits Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Receptor-interacting serine/threonine-protein kinase 1
(324/324 = 100%)†
(Homo sapiens (Human)) | BDBM50446278
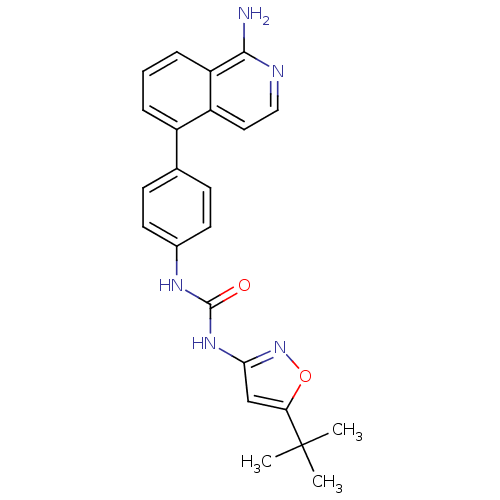
(CHEMBL3109202)Show SMILES CC(C)(C)c1cc(NC(=O)Nc2ccc(cc2)-c2cccc3c(N)nccc23)no1 Show InChI InChI=1S/C23H23N5O2/c1-23(2,3)19-13-20(28-30-19)27-22(29)26-15-9-7-14(8-10-15)16-5-4-6-18-17(16)11-12-25-21(18)24/h4-13H,1-3H3,(H2,24,25)(H2,26,27,28,29) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human N-terminal His-GST-TEV-fused RIP1 kinase domain (1 to 375) autophosphorylation expressed in baculovirus infected insect Sf9 cells... |
ACS Med Chem Lett 4: 1238-43 (2013)
Article DOI: 10.1021/ml400382p
BindingDB Entry DOI: 10.7270/Q2C24XW2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Receptor-interacting serine/threonine-protein kinase 1
(324/324 = 100%)†
(Homo sapiens (Human)) | BDBM50446278

(CHEMBL3109202)Show SMILES CC(C)(C)c1cc(NC(=O)Nc2ccc(cc2)-c2cccc3c(N)nccc23)no1 Show InChI InChI=1S/C23H23N5O2/c1-23(2,3)19-13-20(28-30-19)27-22(29)26-15-9-7-14(8-10-15)16-5-4-6-18-17(16)11-12-25-21(18)24/h4-13H,1-3H3,(H2,24,25)(H2,26,27,28,29) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Displacement of (14-(2-{[3-({2-{[4-(cyanomethyl)phenyl]amino}-6-[(5-cyclopropyl-1H-pyrazol-3-yl)amino]-4-pyrimidinyl}amino)propyl]amino}-2-oxoethyl)-... |
ACS Med Chem Lett 4: 1238-43 (2013)
Article DOI: 10.1021/ml400382p
BindingDB Entry DOI: 10.7270/Q2C24XW2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Receptor-interacting serine/threonine-protein kinase 1
(324/324 = 100%)†
(Homo sapiens (Human)) | BDBM50446278

(CHEMBL3109202)Show SMILES CC(C)(C)c1cc(NC(=O)Nc2ccc(cc2)-c2cccc3c(N)nccc23)no1 Show InChI InChI=1S/C23H23N5O2/c1-23(2,3)19-13-20(28-30-19)27-22(29)26-15-9-7-14(8-10-15)16-5-4-6-18-17(16)11-12-25-21(18)24/h4-13H,1-3H3,(H2,24,25)(H2,26,27,28,29) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Binding affinity to human RIP1 (1 to 375 residues) preincubated for 10 mins measured after 20 mins by fluorescence polarization assay |
J Med Chem 59: 2163-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01898
BindingDB Entry DOI: 10.7270/Q26H4K97 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Receptor-interacting serine/threonine-protein kinase 1
(324/324 = 100%)†
(Homo sapiens (Human)) | BDBM50446278

(CHEMBL3109202)Show SMILES CC(C)(C)c1cc(NC(=O)Nc2ccc(cc2)-c2cccc3c(N)nccc23)no1 Show InChI InChI=1S/C23H23N5O2/c1-23(2,3)19-13-20(28-30-19)27-22(29)26-15-9-7-14(8-10-15)16-5-4-6-18-17(16)11-12-25-21(18)24/h4-13H,1-3H3,(H2,24,25)(H2,26,27,28,29) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Binding affinity to human RIP1 (1 to 375 residues) preincubated for 10 mins measured after 20 mins by fluorescence polarization assay |
J Med Chem 59: 2163-78 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01898
BindingDB Entry DOI: 10.7270/Q26H4K97 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Receptor-interacting serine/threonine-protein kinase 1
(324/324 = 100%)†
(Homo sapiens (Human)) | BDBM50446278

(CHEMBL3109202)Show SMILES CC(C)(C)c1cc(NC(=O)Nc2ccc(cc2)-c2cccc3c(N)nccc23)no1 Show InChI InChI=1S/C23H23N5O2/c1-23(2,3)19-13-20(28-30-19)27-22(29)26-15-9-7-14(8-10-15)16-5-4-6-18-17(16)11-12-25-21(18)24/h4-13H,1-3H3,(H2,24,25)(H2,26,27,28,29) | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 630 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of RIP1 in human U937 cells assessed as prevention of TNFalpha/zVAD.fmk-induced necrotic cell death preincubated for 30 to 60 mins followe... |
ACS Med Chem Lett 4: 1238-43 (2013)
Article DOI: 10.1021/ml400382p
BindingDB Entry DOI: 10.7270/Q2C24XW2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data