

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Retinoic acid receptor alpha [200-419]/gamma [183-417] (Homo sapiens (Human)) | BDBM36810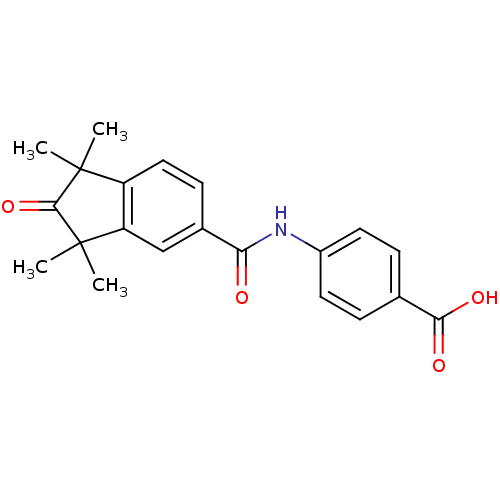 (BMS753 | US9963439, BMS753) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 1 | -47.8 | n/a | n/a | n/a | n/a | n/a | 8.0 | 4 |
Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC) | Assay Description Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)... | Chem Biol 6: 519-29 (1999) Article DOI: 10.1016/S1074-5521(99)80084-2 BindingDB Entry DOI: 10.7270/Q2CV4G39 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha [200-419]/gamma [183-417] (Homo sapiens (Human)) | BDBM36811 (BMS614) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid UniChem Patents | Article PubMed | 1 | -47.8 | n/a | n/a | n/a | n/a | n/a | 8.0 | 4 |
Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC) | Assay Description Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)... | Chem Biol 6: 519-29 (1999) Article DOI: 10.1016/S1074-5521(99)80084-2 BindingDB Entry DOI: 10.7270/Q2CV4G39 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor gamma [183-417] (Homo sapiens (Human)) | BDBM31889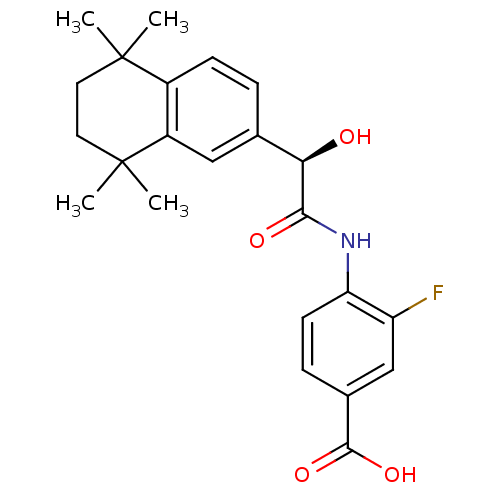 (BMS 961 | BMS270394 | BMS961) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | 1.5 | -46.8 | n/a | n/a | n/a | n/a | n/a | 8.0 | 4 |
Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC) | Assay Description Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)... | Chem Biol 6: 519-29 (1999) Article DOI: 10.1016/S1074-5521(99)80084-2 BindingDB Entry DOI: 10.7270/Q2CV4G39 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Retinoic acid receptor alpha [200-419] (Homo sapiens (Human)) | BDBM36810 (BMS753 | US9963439, BMS753) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | -46.2 | n/a | n/a | n/a | n/a | n/a | 8.0 | 4 |
Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC) | Assay Description Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)... | Chem Biol 6: 519-29 (1999) Article DOI: 10.1016/S1074-5521(99)80084-2 BindingDB Entry DOI: 10.7270/Q2CV4G39 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Retinoic acid receptor alpha [200-419] (Homo sapiens (Human)) | BDBM36811 (BMS614) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid UniChem Patents | MMDB Article PubMed | 2.5 | -45.6 | n/a | n/a | n/a | n/a | n/a | 8.0 | 4 |
Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC) | Assay Description Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)... | Chem Biol 6: 519-29 (1999) Article DOI: 10.1016/S1074-5521(99)80084-2 BindingDB Entry DOI: 10.7270/Q2CV4G39 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||