

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096675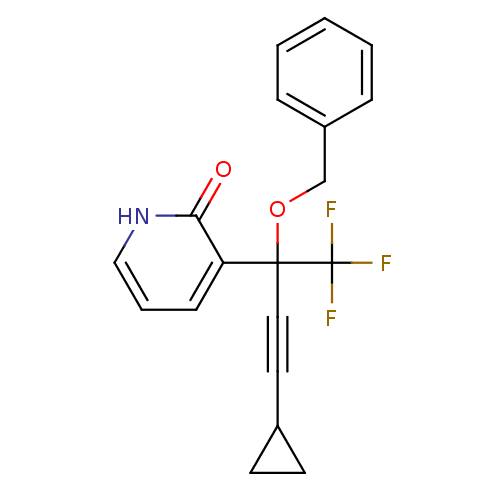 (3-(1-Benzyloxy-3-cyclopropyl-1-trifluoromethyl-pro...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096666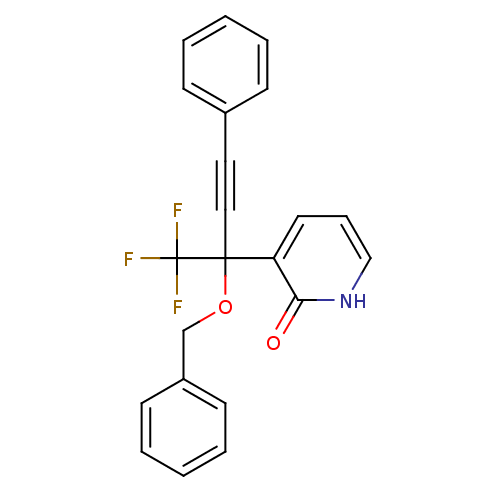 (3-(1-Benzyloxy-3-phenyl-1-trifluoromethyl-prop-2-y...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096660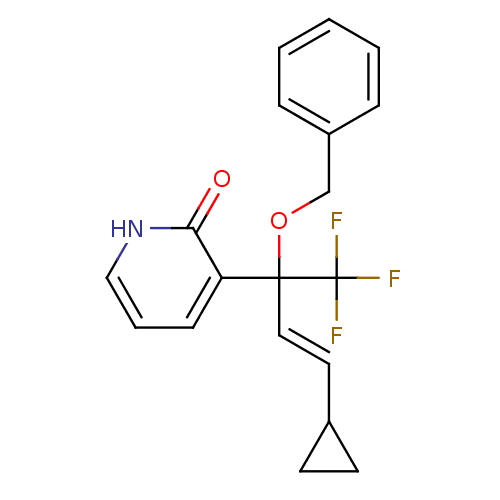 (3-((E)-1-Benzyloxy-3-cyclopropyl-1-trifluoromethyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM1317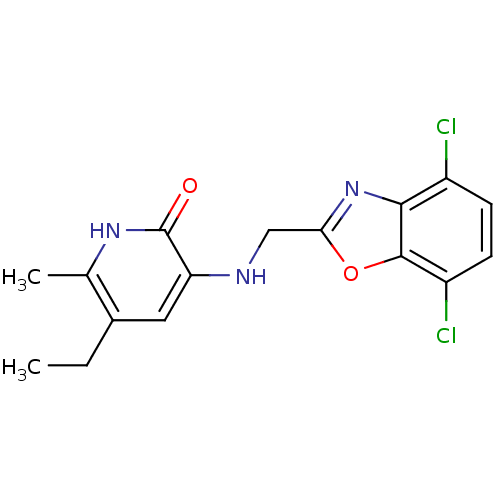 (3-[[(4,7-Dichlorobenzoxazol-2-yl)-methyl]amino]-5-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096668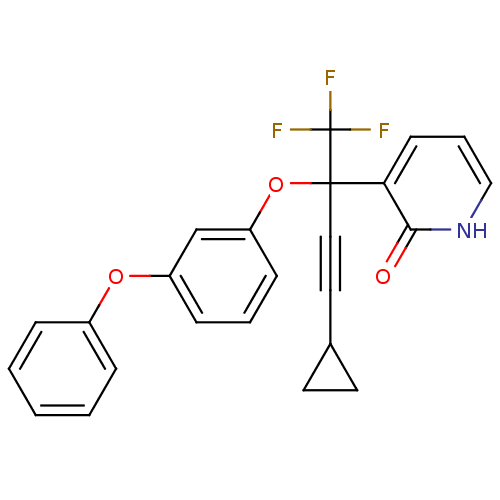 (3-[3-Cyclopropyl-1-(3-phenoxy-phenoxy)-1-trifluoro...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096654 (3-[3-Cyclopropyl-1-(pyridin-2-ylmethoxy)-1-trifluo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096664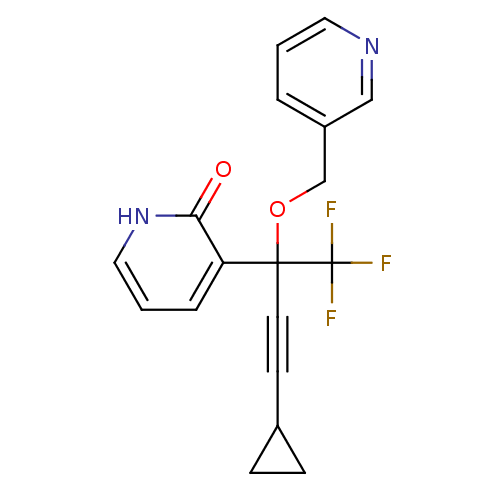 (3-[3-Cyclopropyl-1-(pyridin-3-ylmethoxy)-1-trifluo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM2898 ((4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluor...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Compound was evaluated for its ability to inhibit the NNRTI HIV-1 enzyme by 50% using enzyme assay | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096667 (3-(1-Benzyloxy-3-cyclopropyl-1-trifluoromethyl-pro...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096657 (3-[3-Cyclopropyl-1-(2,6-dichloro-benzyloxy)-1-trif...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096673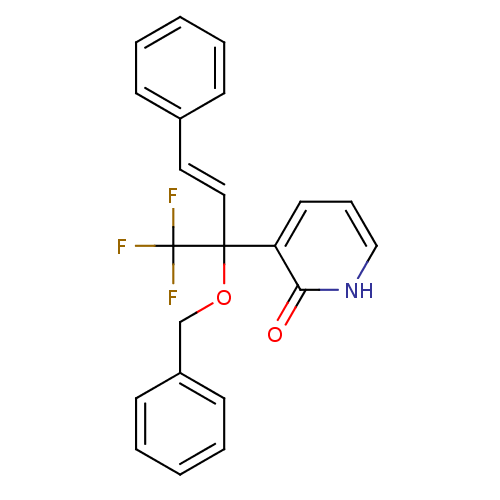 (3-((E)-1-Benzyloxy-3-phenyl-1-trifluoromethyl-ally...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM2483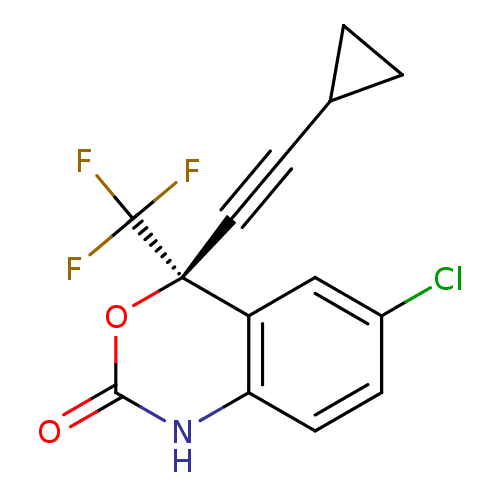 ((4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluor...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB PubMed | n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096661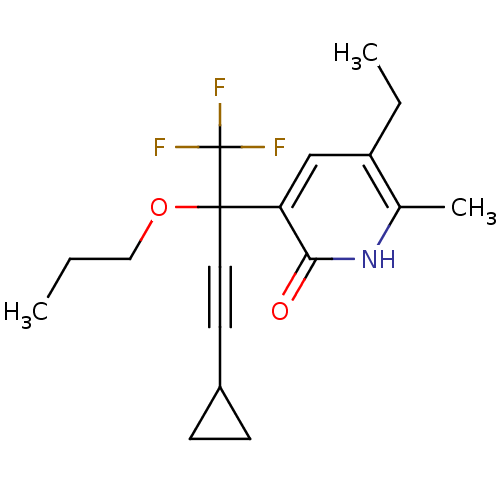 (3-(3-Cyclopropyl-1-propoxy-1-trifluoromethyl-prop-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 105 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096656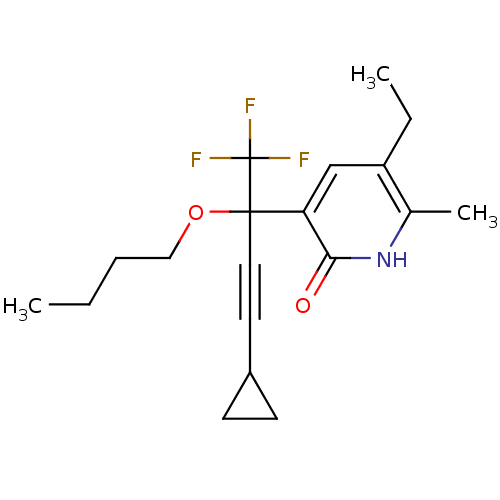 (3-(1-Butoxy-3-cyclopropyl-1-trifluoromethyl-prop-2...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 129 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096663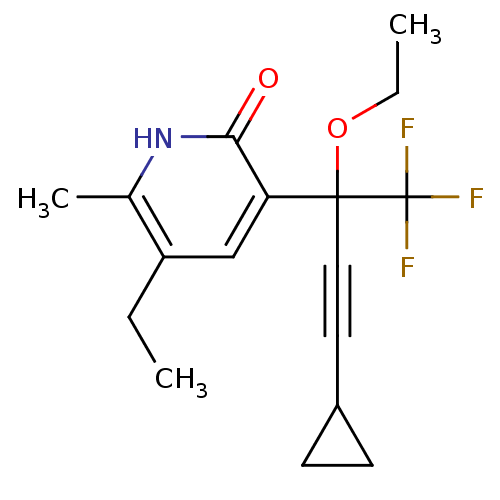 (3-(3-Cyclopropyl-1-ethoxy-1-trifluoromethyl-prop-2...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 183 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096672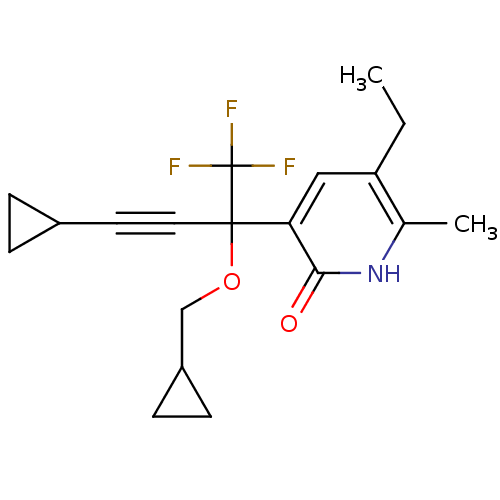 (3-(3-Cyclopropyl-1-cyclopropylmethoxy-1-trifluorom...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 188 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096670 (3-[3-Cyclopropyl-1-(2,6-difluoro-benzyloxy)-1-trif...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 613 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096674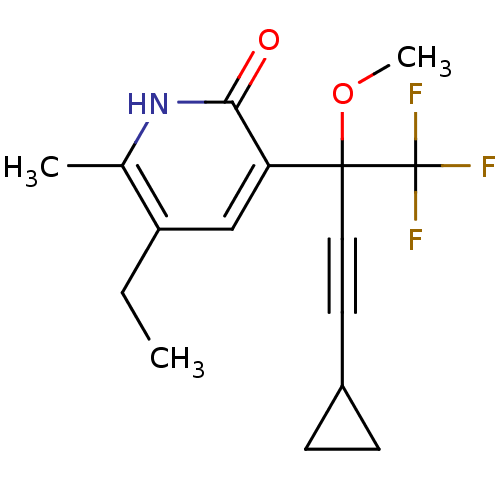 (3-(3-Cyclopropyl-1-methoxy-1-trifluoromethyl-prop-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 770 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096662 (3-(1-Benzyloxy-3-cyclopropyl-1-trifluoromethyl-pro...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.35E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096655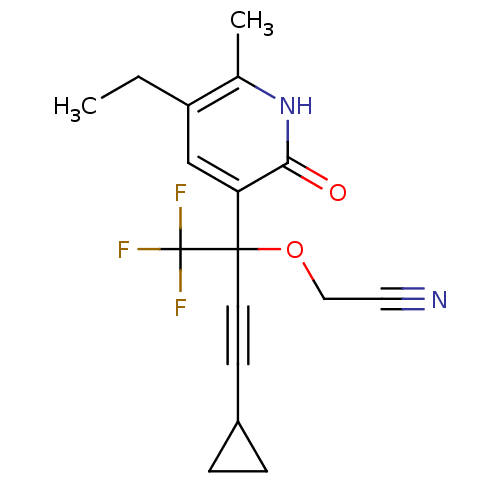 (CHEMBL106659 | [3-Cyclopropyl-1-(5-ethyl-6-methyl-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096669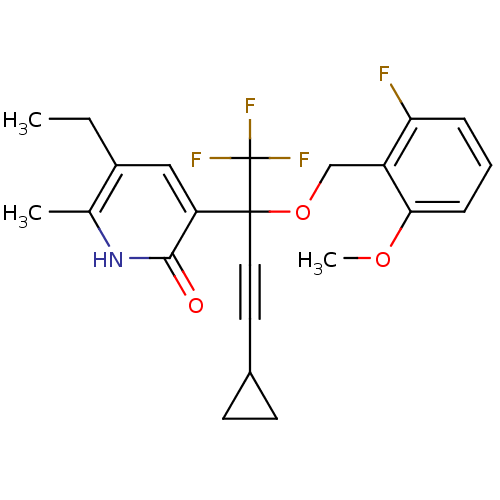 (3-[3-Cyclopropyl-1-(2-fluoro-6-methoxy-benzyloxy)-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.95E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096658 (3-[3-Cyclopropyl-1-(2,4-dichloro-benzyloxy)-1-trif...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096659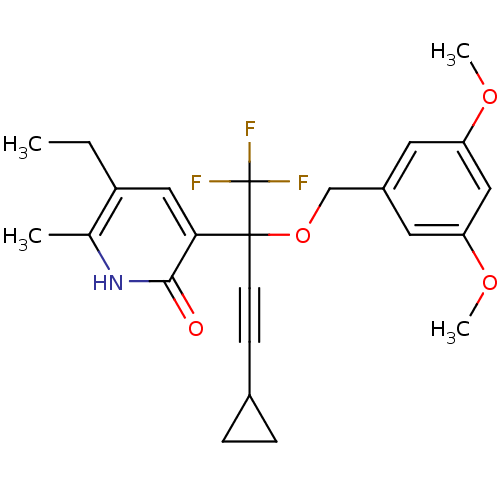 (3-[3-Cyclopropyl-1-(3,5-dimethoxy-benzyloxy)-1-tri...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096665 (3-[1-(2-Chloro-6-fluoro-benzyloxy)-3-cyclopropyl-1...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 6.09E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50096671 (3-[3-Cyclopropyl-1-(pyridin-4-ylmethoxy)-1-trifluo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company Curated by ChEMBL | Assay Description Inhibition of the NNRTI HIV-1 enzyme by 50% using enzyme assay. | Bioorg Med Chem Lett 11: 309-12 (2001) BindingDB Entry DOI: 10.7270/Q23T9HSR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||