

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096730 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096729 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Benzylaminooxaly...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 3 receptor | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096726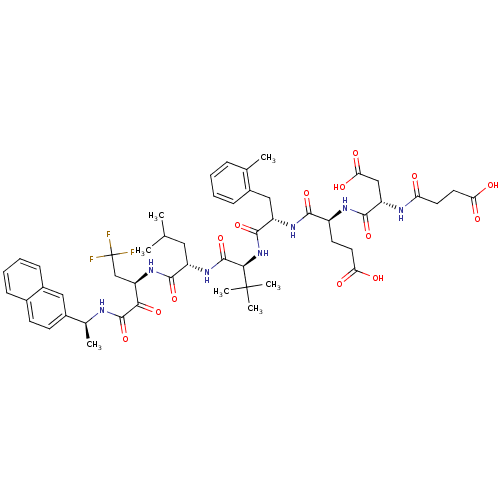 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 3 receptor | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096734 ((S)-4-[(S)-3-Carboxy-2-(3-carboxy-propionylamino)-...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 with a Y181C mutation in RT (reverse ... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096727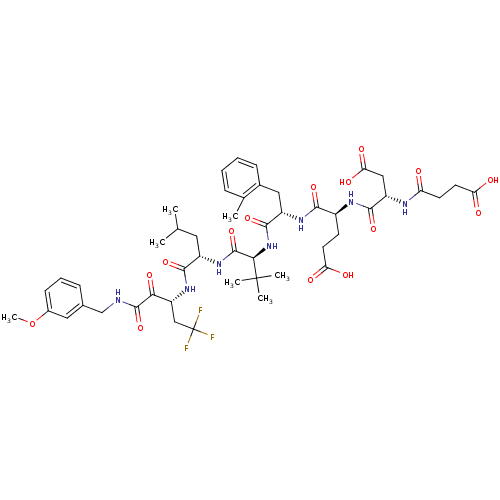 ((S)-4-[(S)-3-Carboxy-2-(3-carboxy-propionylamino)-...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096725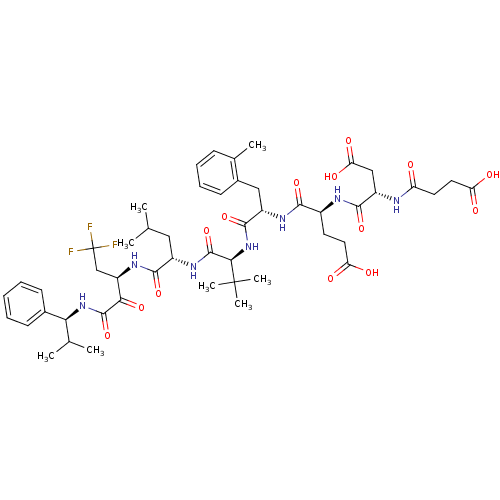 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against delta receptor of (endomorphin 2) in mouse vas deferens was determined | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096728 ((S)-4-((S)-1-{(S)-1-[(S)-1-((S)-1-Amino-2-carbamoy...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 with a Y181C mutation in RT (reverse ... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096733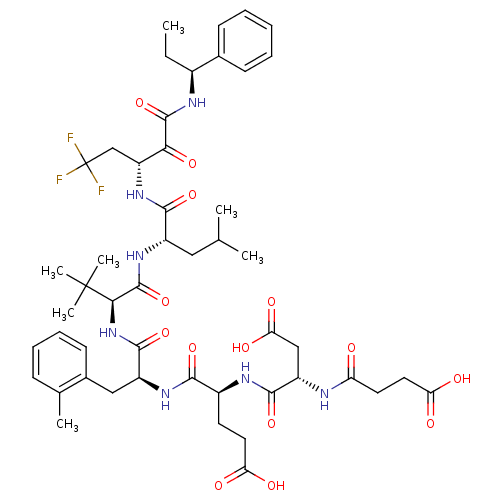 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 3 receptor | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096732 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 with a Y181C mutation in RT (reverse ... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096724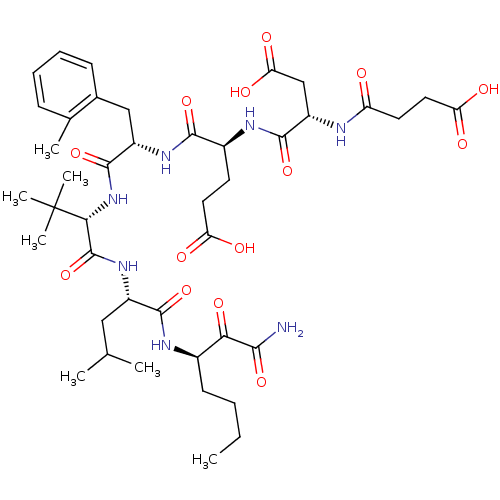 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Aminooxalyl-pent...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Chymotrypsin-C (Homo sapiens (Human)) | BDBM50096724 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Aminooxalyl-pent...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Compound was tested for inhibition of Serine protease chymotrypsin | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096731 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil elastase (Homo sapiens (Human)) | BDBM50096724 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Aminooxalyl-pent...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Compound was tested for inhibition of Elastase | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin (Homo sapiens (Human)) | BDBM50096724 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Aminooxalyl-pent...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against Opioid receptor mu 1 of (endomorphin 2) in guinea pig ileum was determined | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||