

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Integrase (Human immunodeficiency virus 1) | BDBM50056891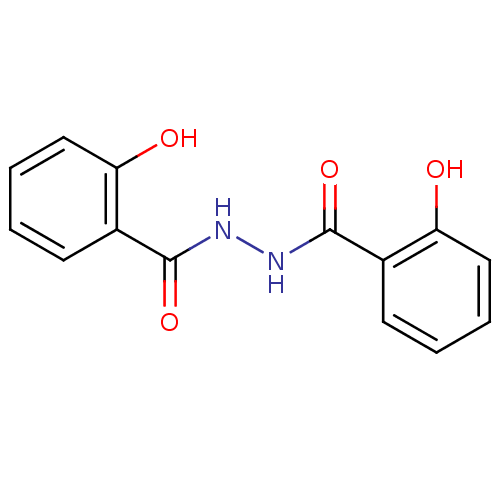 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970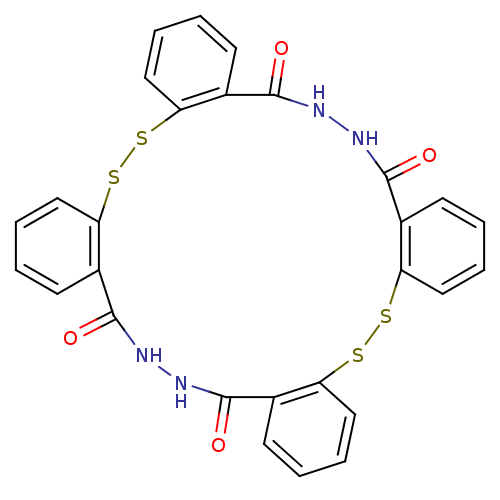 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972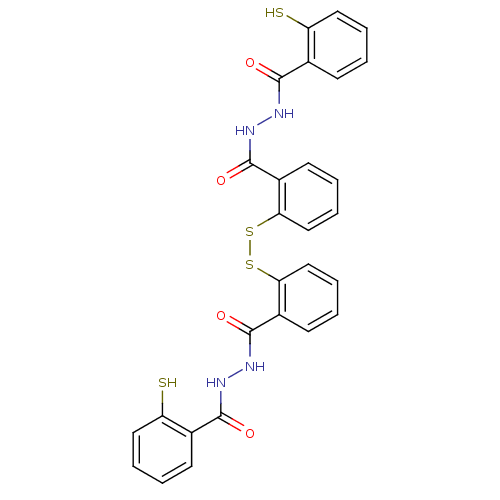 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971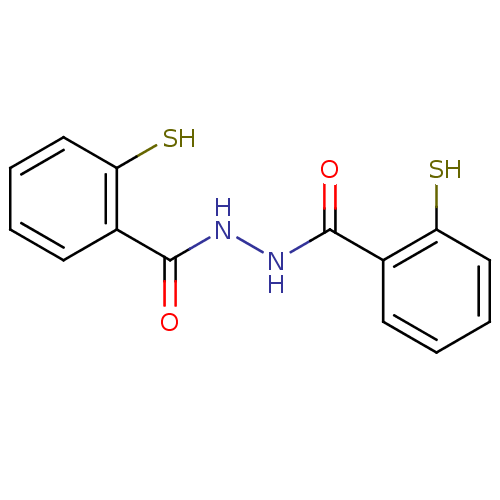 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 7.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | 3.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mn+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >9.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against viral protease | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | >9.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against viral protease | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >9.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against viral protease | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against rCdG of HIV-1 Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against rCdG of HIV-1 Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against RAdT of HIV-1 Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against viral attachment | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against viral protease | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against viral attachment | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against RAdT of HIV-1 Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity of the compound against RAdT of HIV-1 Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against rCdG of HIV-1 Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in 3''-processing of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in 3'' processing of postasse... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and DNA on ice for 15 min followed by drug for 1h) in strand transfer of postass... | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Integrase (Human immunodeficiency virus 1) | BDBM50056891 (2-Hydroxy-benzoic acid N'-(2-hydroxy-benzoyl)-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 integrase (preincubated with Mg+2, and drug for 30 min followed by DNA for 1h) in strand transfer of preassemble | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121971 (2-Mercapto-benzoic acid N'-(2-mercapto-benzoyl)-hy...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents | PubMed | n/a | n/a | n/a | n/a | 4.30E+3 | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Effective concentration of the compound against viral replication by Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121972 (CHEMBL157373 | Compound 3) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 1.78E+4 | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Effective concentration of the compound against viral replication by Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50121970 (11,12,13,14,25,26,27,28-octahydrotetrabenzo[c,i,m,...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a |
University of Southern California Curated by ChEMBL | Assay Description Effective concentration of the compound against viral replication by Reverse transcriptase | J Med Chem 45: 5661-70 (2002) BindingDB Entry DOI: 10.7270/Q2M61JMB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||