

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127511 (4-hydroxy-2-(methoxymethyl)-5-(6-oxo-1-prop-2-ynyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 22.3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127517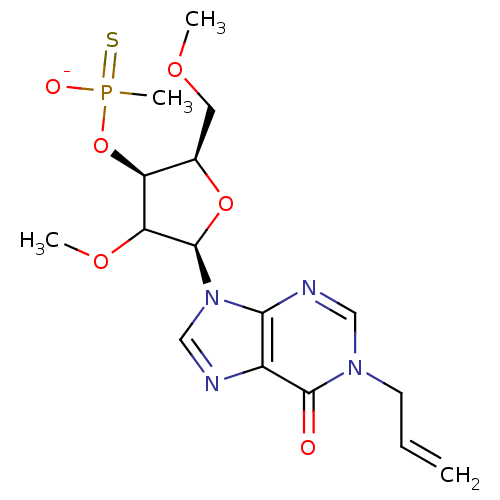 (O-[5-(1-allyl-6-oxo-1,6-dihydro-9H-purin-9-yl)-4-m...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127514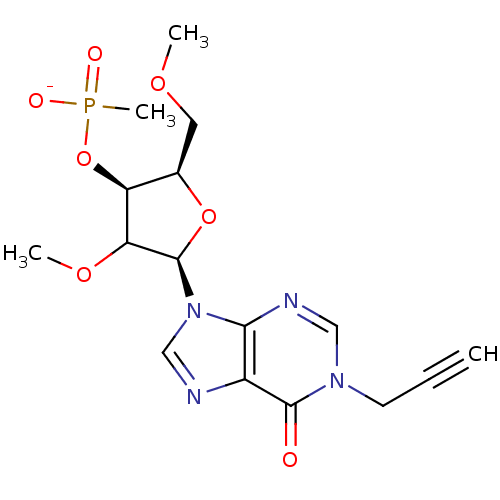 (4-methoxy-2-(methoxymethyl)-5-(6-oxo-1-prop-2-ynyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127512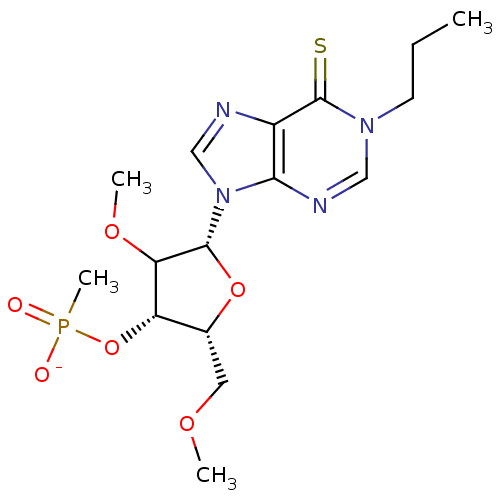 ((2R,3S,5R)-4-methoxy-2-(methoxymethyl)-5-(1-propyl...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 820 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127513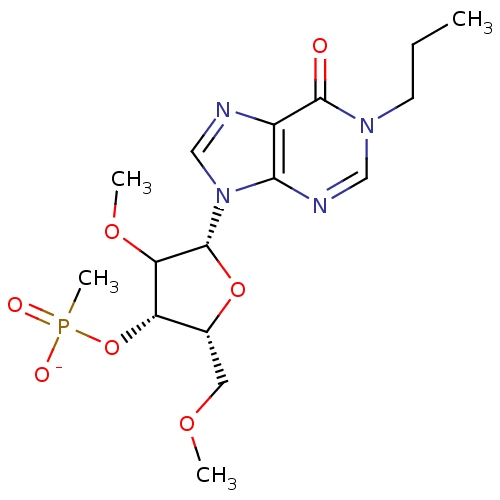 ((2R,3S,5R)-4-methoxy-2-(methoxymethyl)-5-(6-oxo-1-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127515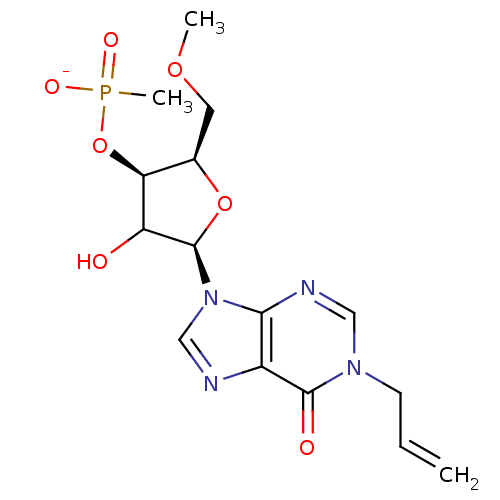 (5-(1-allyl-6-oxo-1,6-dihydro-9H-purin-9-yl)-4-hydr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 9.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50127516 (5-(1-allyl-6-oxo-1,6-dihydro-9H-purin-9-yl)-4-meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.05E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibitory activity of compound against reverse transcriptase (RT) in cell free RT. | J Med Chem 46: 1878-85 (2003) Article DOI: 10.1021/jm0203276 BindingDB Entry DOI: 10.7270/Q25X29NG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||