 Found 21 hits of Enzyme Inhibition Constant Data
Found 21 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132386
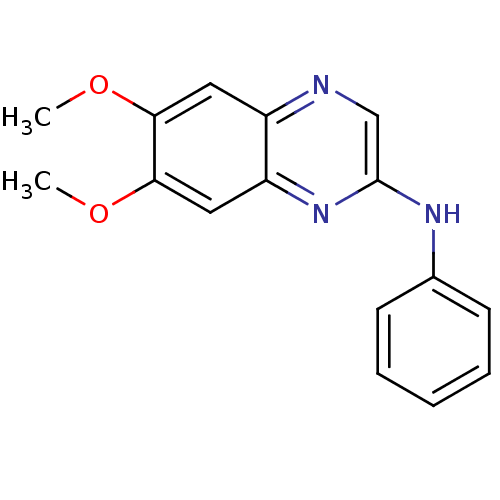
((6,7-Dimethoxy-quinoxalin-2-yl)-phenyl-amine | CHE...)Show InChI InChI=1S/C16H15N3O2/c1-20-14-8-12-13(9-15(14)21-2)19-16(10-17-12)18-11-6-4-3-5-7-11/h3-10H,1-2H3,(H,18,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132406
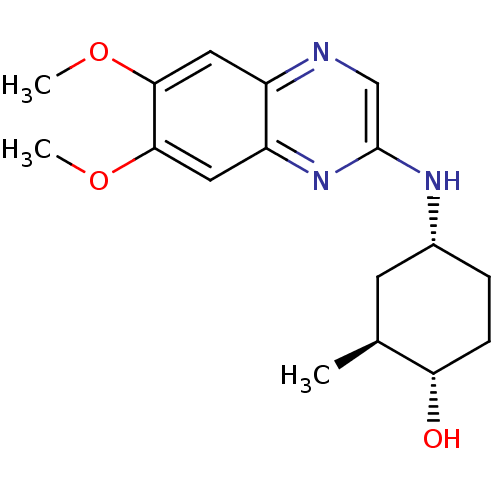
(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-2-methyl-cy...)Show SMILES COc1cc2ncc(N[C@@H]3CC[C@H](O)[C@@H](C)C3)nc2cc1OC Show InChI InChI=1S/C17H23N3O3/c1-10-6-11(4-5-14(10)21)19-17-9-18-12-7-15(22-2)16(23-3)8-13(12)20-17/h7-11,14,21H,4-6H2,1-3H3,(H,19,20)/t10-,11+,14-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132405
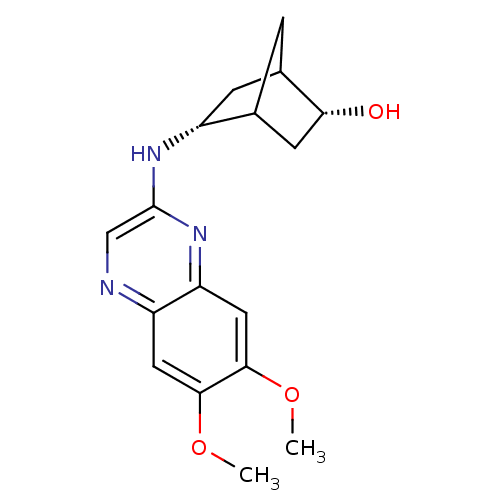
(5-(6,7-Dimethoxy-quinoxalin-2-ylamino)-bicyclo[2.2...)Show SMILES COc1cc2ncc(N[C@@H]3CC4CC3C[C@H]4O)nc2cc1OC |TLB:16:15:12:9.10,8:9:12:14.15| Show InChI InChI=1S/C17H21N3O3/c1-22-15-6-12-13(7-16(15)23-2)20-17(8-18-12)19-11-4-10-3-9(11)5-14(10)21/h6-11,14,21H,3-5H2,1-2H3,(H,19,20)/t9?,10?,11-,14-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132409
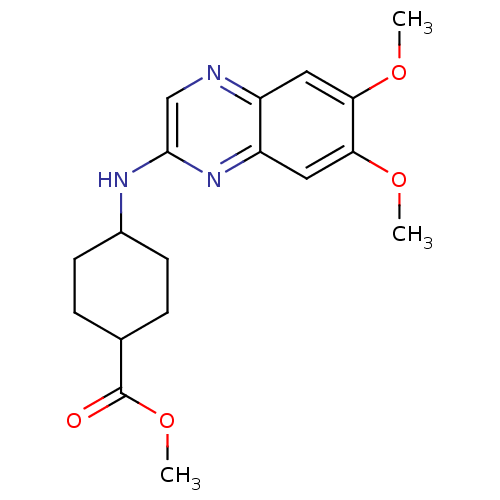
(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohexane...)Show SMILES COC(=O)C1CCC(CC1)Nc1cnc2cc(OC)c(OC)cc2n1 |(8.36,2.63,;7.03,3.39,;5.7,2.62,;5.71,1.09,;4.37,3.39,;4.36,4.92,;3.03,5.69,;1.71,4.92,;1.71,3.37,;3.03,2.62,;.38,5.68,;-.95,4.9,;-.95,3.35,;-2.28,2.57,;-3.62,3.34,;-4.95,2.57,;-6.27,3.34,;-7.62,2.57,;-7.62,1.04,;-6.27,4.89,;-7.62,5.67,;-8.95,4.89,;-4.95,5.67,;-3.62,4.89,;-2.3,5.67,)| Show InChI InChI=1S/C18H23N3O4/c1-23-15-8-13-14(9-16(15)24-2)21-17(10-19-13)20-12-6-4-11(5-7-12)18(22)25-3/h8-12H,4-7H2,1-3H3,(H,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132395
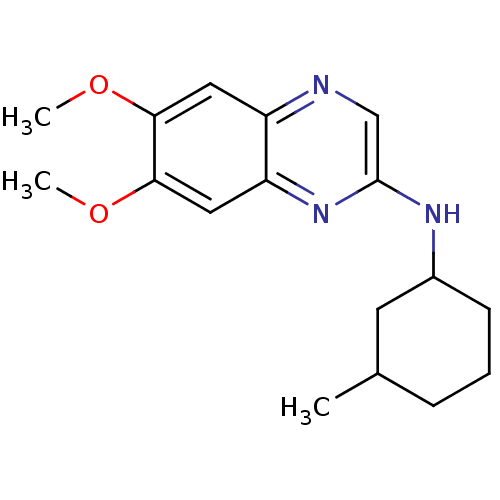
((6,7-Dimethoxy-quinoxalin-2-yl)-(3-methyl-cyclohex...)Show InChI InChI=1S/C17H23N3O2/c1-11-5-4-6-12(7-11)19-17-10-18-13-8-15(21-2)16(22-3)9-14(13)20-17/h8-12H,4-7H2,1-3H3,(H,19,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132409

(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohexane...)Show SMILES COC(=O)C1CCC(CC1)Nc1cnc2cc(OC)c(OC)cc2n1 |(8.36,2.63,;7.03,3.39,;5.7,2.62,;5.71,1.09,;4.37,3.39,;4.36,4.92,;3.03,5.69,;1.71,4.92,;1.71,3.37,;3.03,2.62,;.38,5.68,;-.95,4.9,;-.95,3.35,;-2.28,2.57,;-3.62,3.34,;-4.95,2.57,;-6.27,3.34,;-7.62,2.57,;-7.62,1.04,;-6.27,4.89,;-7.62,5.67,;-8.95,4.89,;-4.95,5.67,;-3.62,4.89,;-2.3,5.67,)| Show InChI InChI=1S/C18H23N3O4/c1-23-15-8-13-14(9-16(15)24-2)21-17(10-19-13)20-12-6-4-11(5-7-12)18(22)25-3/h8-12H,4-7H2,1-3H3,(H,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132407
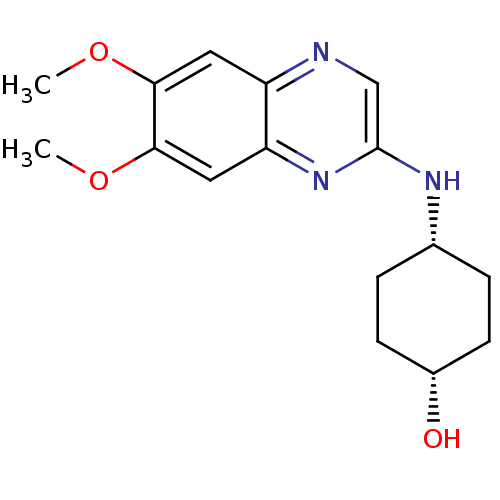
(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohexano...)Show SMILES COc1cc2ncc(N[C@@H]3CC[C@H](O)CC3)nc2cc1OC |wD:9.8,12.12,(3.37,-6.65,;3.37,-5.12,;4.71,-4.35,;6.03,-5.12,;7.37,-4.35,;8.7,-5.12,;10.05,-4.33,;10.03,-2.79,;11.36,-2.01,;12.7,-2.78,;14.01,-2,;15.35,-2.77,;15.35,-4.31,;16.68,-5.07,;14.03,-5.08,;12.7,-4.31,;8.7,-2.03,;7.37,-2.8,;6.03,-2.03,;4.71,-2.81,;3.37,-2.03,;2.05,-2.8,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132391
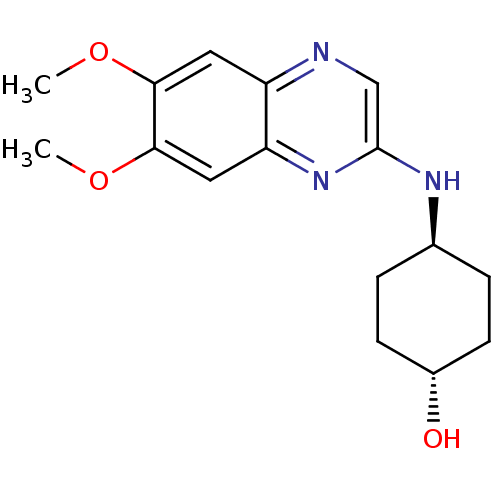
(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132399
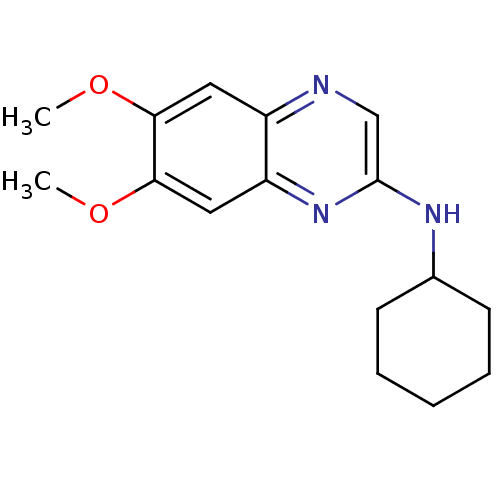
(CHEMBL107402 | Cyclohexyl-(6,7-dimethoxy-quinoxali...)Show InChI InChI=1S/C16H21N3O2/c1-20-14-8-12-13(9-15(14)21-2)19-16(10-17-12)18-11-6-4-3-5-7-11/h8-11H,3-7H2,1-2H3,(H,18,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 124 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 299 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Mitogenesis, PDGF-AA, assay. |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132409

(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohexane...)Show SMILES COC(=O)C1CCC(CC1)Nc1cnc2cc(OC)c(OC)cc2n1 |(8.36,2.63,;7.03,3.39,;5.7,2.62,;5.71,1.09,;4.37,3.39,;4.36,4.92,;3.03,5.69,;1.71,4.92,;1.71,3.37,;3.03,2.62,;.38,5.68,;-.95,4.9,;-.95,3.35,;-2.28,2.57,;-3.62,3.34,;-4.95,2.57,;-6.27,3.34,;-7.62,2.57,;-7.62,1.04,;-6.27,4.89,;-7.62,5.67,;-8.95,4.89,;-4.95,5.67,;-3.62,4.89,;-2.3,5.67,)| Show InChI InChI=1S/C18H23N3O4/c1-23-15-8-13-14(9-16(15)24-2)21-17(10-19-13)20-12-6-4-11(5-7-12)18(22)25-3/h8-12H,4-7H2,1-3H3,(H,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 345 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 353 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Mitogenesis, PDGF-BB, assay |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50039082
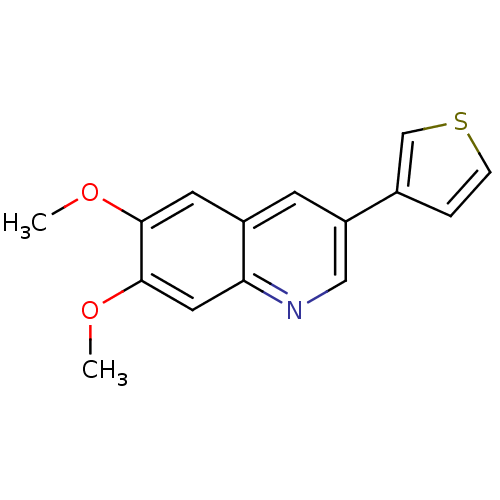
(6,7-Dimethoxy-3-thiophen-3-yl-quinoline | CHEMBL66...)Show InChI InChI=1S/C15H13NO2S/c1-17-14-6-11-5-12(10-3-4-19-9-10)8-16-13(11)7-15(14)18-2/h3-9H,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 461 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132404
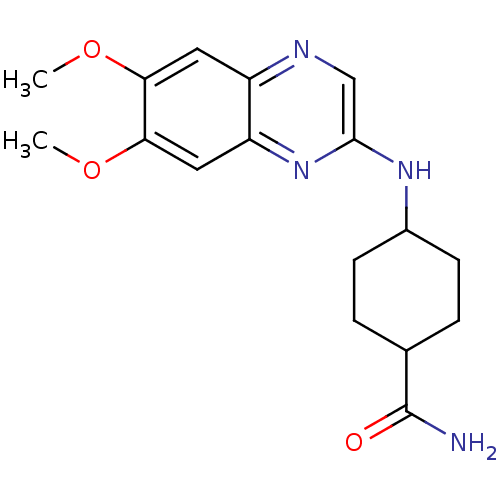
(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohexane...)Show SMILES COc1cc2ncc(NC3CCC(CC3)C(N)=O)nc2cc1OC |(-7.56,-1.47,;-7.56,.07,;-6.22,.84,;-4.89,.07,;-3.55,.84,;-2.23,.07,;-.88,.85,;-.88,2.4,;.44,3.18,;1.78,2.41,;3.1,3.19,;4.43,2.42,;4.43,.88,;3.1,.11,;1.78,.88,;5.76,.12,;5.77,-1.42,;7.1,.88,;-2.23,3.16,;-3.55,2.39,;-4.89,3.16,;-6.22,2.37,;-7.56,3.16,;-8.88,2.39,)| Show InChI InChI=1S/C17H22N4O3/c1-23-14-7-12-13(8-15(14)24-2)21-16(9-19-12)20-11-5-3-10(4-6-11)17(18)22/h7-11H,3-6H2,1-2H3,(H2,18,22)(H,20,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 930 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50132408
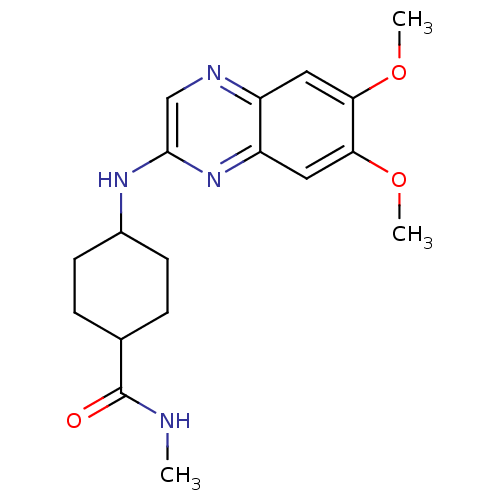
(4-(6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohexane...)Show SMILES CNC(=O)C1CCC(CC1)Nc1cnc2cc(OC)c(OC)cc2n1 |(7.11,-2.19,;5.77,-1.42,;5.76,.12,;7.1,.88,;4.43,.88,;4.43,2.42,;3.1,3.19,;1.78,2.41,;1.78,.88,;3.1,.11,;.44,3.18,;-.88,2.4,;-.88,.85,;-2.23,.07,;-3.55,.84,;-4.89,.07,;-6.22,.84,;-7.56,.07,;-7.56,-1.47,;-6.22,2.37,;-7.56,3.16,;-8.88,2.39,;-4.89,3.16,;-3.55,2.39,;-2.23,3.16,)| Show InChI InChI=1S/C18H24N4O3/c1-19-18(23)11-4-6-12(7-5-11)21-17-10-20-13-8-15(24-2)16(25-3)9-14(13)22-17/h8-12H,4-7H2,1-3H3,(H,19,23)(H,21,22) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Epidermal growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Insulin receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK2
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Janus kinase 2 |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 10/8/9
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Platelet-derived growth factor receptor |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK3
(Homo sapiens (Human)) | BDBM50132391

(4-((R)-6,7-Dimethoxy-quinoxalin-2-ylamino)-cyclohe...)Show SMILES COc1cc2ncc(N[C@H]3CC[C@H](O)CC3)nc2cc1OC |wU:12.12,wD:9.8,(-.13,-6.56,;-.13,-5.02,;1.2,-4.25,;2.53,-5.02,;3.88,-4.23,;5.21,-5,;6.54,-4.23,;6.54,-2.69,;7.85,-1.9,;9.2,-2.67,;9.2,-4.21,;10.53,-4.98,;11.86,-4.21,;13.19,-4.98,;11.86,-2.67,;10.53,-1.9,;5.19,-1.92,;3.88,-2.69,;2.53,-1.92,;1.2,-2.71,;-.13,-1.94,;-.13,-.4,)| Show InChI InChI=1S/C16H21N3O3/c1-21-14-7-12-13(8-15(14)22-2)19-16(9-17-12)18-10-3-5-11(20)6-4-10/h7-11,20H,3-6H2,1-2H3,(H,18,19)/t10-,11- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of Janus kinase 3 |
Bioorg Med Chem Lett 13: 3097-100 (2003)
BindingDB Entry DOI: 10.7270/Q2HH6JGM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data