

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50134782 (6-amino-9-[(Z)-(2-{[(1,2,3,3-tetrahydroxy-1,2,3-tr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 Reverse transcriptase wild-type (RT wt) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50164644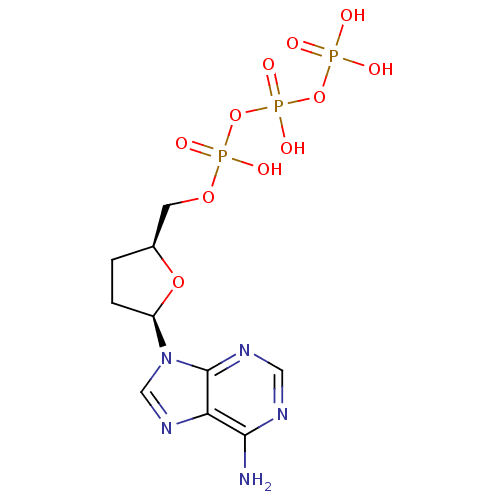 (2',3'-Dideoxyadenosine Triphosphate (Ddatp) | 2',3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 reverse transcriptase (RT M184V) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50164644 (2',3'-Dideoxyadenosine Triphosphate (Ddatp) | 2',3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 Reverse transcriptase wild-type (RT wt) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50164644 (2',3'-Dideoxyadenosine Triphosphate (Ddatp) | 2',3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 reverse transcriptase (RT M184I) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50134782 (6-amino-9-[(Z)-(2-{[(1,2,3,3-tetrahydroxy-1,2,3-tr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 reverse transcriptase (RT M184V) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50134782 (6-amino-9-[(Z)-(2-{[(1,2,3,3-tetrahydroxy-1,2,3-tr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 reverse transcriptase (RT M184I) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50134783 (6-amino-9-[(E)-(2-{[(1,2,3,3-tetrahydroxy-1,2,3-tr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 Reverse transcriptase wild-type (RT wt) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50134783 (6-amino-9-[(E)-(2-{[(1,2,3,3-tetrahydroxy-1,2,3-tr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 reverse transcriptase (RT M184V) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50134783 (6-amino-9-[(E)-(2-{[(1,2,3,3-tetrahydroxy-1,2,3-tr...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wayne State University School of Medicine Curated by ChEMBL | Assay Description Inhibitory activity against HIV-1 reverse transcriptase (RT M184I) | J Med Chem 46: 4799-802 (2003) Article DOI: 10.1021/jm030048y BindingDB Entry DOI: 10.7270/Q2D50NQB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||