 Found 63 hits of Enzyme Inhibition Constant Data
Found 63 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157952
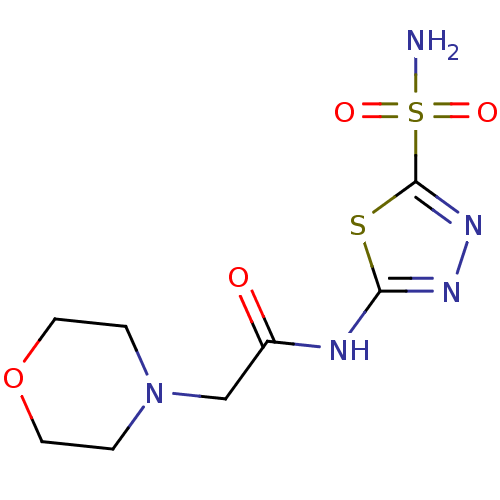
(2-Morpholin-4-yl-N-(5-sulfamoyl-[1,3,4]thiadiazol-...)Show InChI InChI=1S/C8H13N5O4S2/c9-19(15,16)8-12-11-7(18-8)10-6(14)5-13-1-3-17-4-2-13/h1-5H2,(H2,9,15,16)(H,10,11,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157949
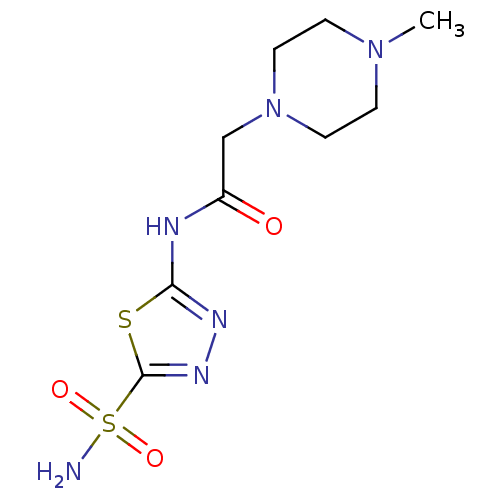
(2-(4-Methyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C9H16N6O3S2/c1-14-2-4-15(5-3-14)6-7(16)11-8-12-13-9(19-8)20(10,17)18/h2-6H2,1H3,(H2,10,17,18)(H,11,12,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157958
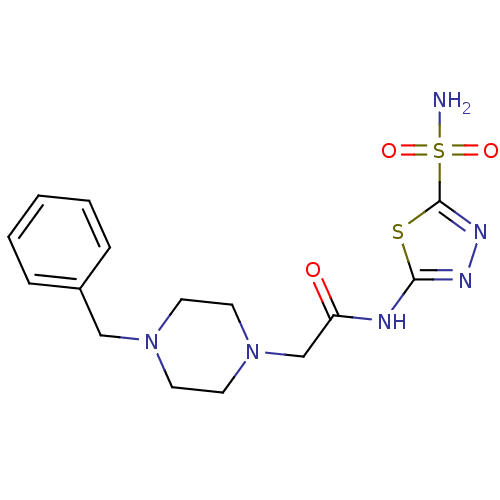
(2-(4-Benzyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show SMILES NS(=O)(=O)c1nnc(NC(=O)CN2CCN(Cc3ccccc3)CC2)s1 Show InChI InChI=1S/C15H20N6O3S2/c16-26(23,24)15-19-18-14(25-15)17-13(22)11-21-8-6-20(7-9-21)10-12-4-2-1-3-5-12/h1-5H,6-11H2,(H2,16,23,24)(H,17,18,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157950
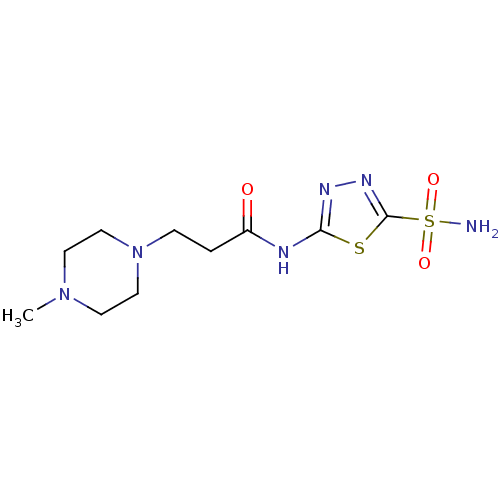
(3-(4-Methyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C10H18N6O3S2/c1-15-4-6-16(7-5-15)3-2-8(17)12-9-13-14-10(20-9)21(11,18)19/h2-7H2,1H3,(H2,11,18,19)(H,12,13,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157955
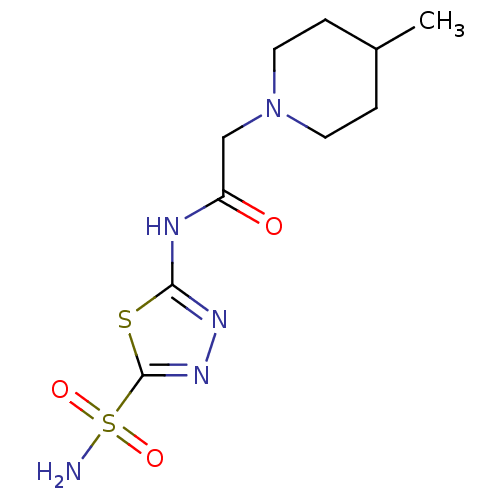
(2-(4-Methyl-piperidin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C10H17N5O3S2/c1-7-2-4-15(5-3-7)6-8(16)12-9-13-14-10(19-9)20(11,17)18/h7H,2-6H2,1H3,(H2,11,17,18)(H,12,13,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157950

(3-(4-Methyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C10H18N6O3S2/c1-15-4-6-16(7-5-15)3-2-8(17)12-9-13-14-10(20-9)21(11,18)19/h2-7H2,1H3,(H2,11,18,19)(H,12,13,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157958

(2-(4-Benzyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show SMILES NS(=O)(=O)c1nnc(NC(=O)CN2CCN(Cc3ccccc3)CC2)s1 Show InChI InChI=1S/C15H20N6O3S2/c16-26(23,24)15-19-18-14(25-15)17-13(22)11-21-8-6-20(7-9-21)10-12-4-2-1-3-5-12/h1-5H,6-11H2,(H2,16,23,24)(H,17,18,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157945
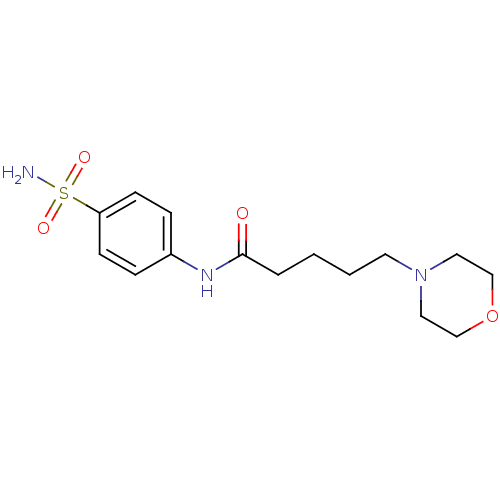
(5-Morpholin-4-yl-pentanoic acid (4-sulfamoyl-pheny...)Show InChI InChI=1S/C15H23N3O4S/c16-23(20,21)14-6-4-13(5-7-14)17-15(19)3-1-2-8-18-9-11-22-12-10-18/h4-7H,1-3,8-12H2,(H,17,19)(H2,16,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10883
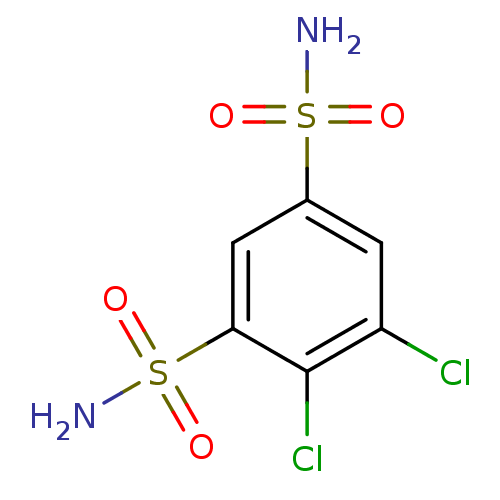
(4,5-dichlorobenzene-1,3-disulfonamide | CHEMBL17 |...)Show InChI InChI=1S/C6H6Cl2N2O4S2/c7-4-1-3(15(9,11)12)2-5(6(4)8)16(10,13)14/h1-2H,(H2,9,11,12)(H2,10,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| DrugBank
MMDB
PDB
Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157955

(2-(4-Methyl-piperidin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C10H17N5O3S2/c1-7-2-4-15(5-3-7)6-8(16)12-9-13-14-10(19-9)20(11,17)18/h7H,2-6H2,1H3,(H2,11,17,18)(H,12,13,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10884

((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...)Show SMILES CCN[C@H]1C[C@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C10H16N2O4S3/c1-3-12-8-4-6(2)18(13,14)10-7(8)5-9(17-10)19(11,15)16/h5-6,8,12H,3-4H2,1-2H3,(H2,11,15,16)/t6-,8-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157951

(3-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-propionami...)Show InChI InChI=1S/C13H19N3O4S/c14-21(18,19)12-3-1-11(2-4-12)15-13(17)5-6-16-7-9-20-10-8-16/h1-4H,5-10H2,(H,15,17)(H2,14,18,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 9.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157946
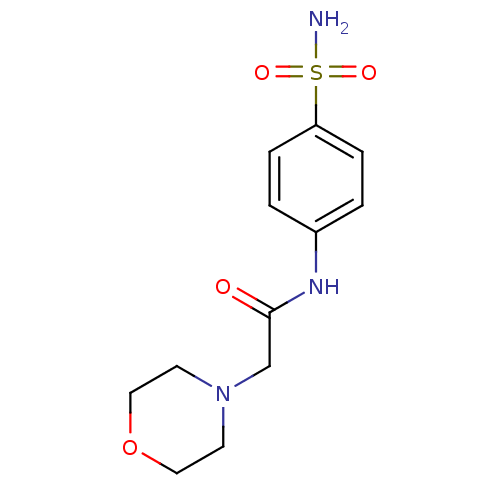
(2-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-acetamide ...)Show InChI InChI=1S/C12H17N3O4S/c13-20(17,18)11-3-1-10(2-4-11)14-12(16)9-15-5-7-19-8-6-15/h1-4H,5-9H2,(H,14,16)(H2,13,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157949

(2-(4-Methyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C9H16N6O3S2/c1-14-2-4-15(5-3-14)6-7(16)11-8-12-13-9(19-8)20(10,17)18/h2-6H2,1H3,(H2,10,17,18)(H,11,12,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10880

(AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...)Show InChI InChI=1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157952

(2-Morpholin-4-yl-N-(5-sulfamoyl-[1,3,4]thiadiazol-...)Show InChI InChI=1S/C8H13N5O4S2/c9-19(15,16)8-12-11-7(18-8)10-6(14)5-13-1-3-17-4-2-13/h1-5H2,(H2,9,15,16)(H,10,11,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10881
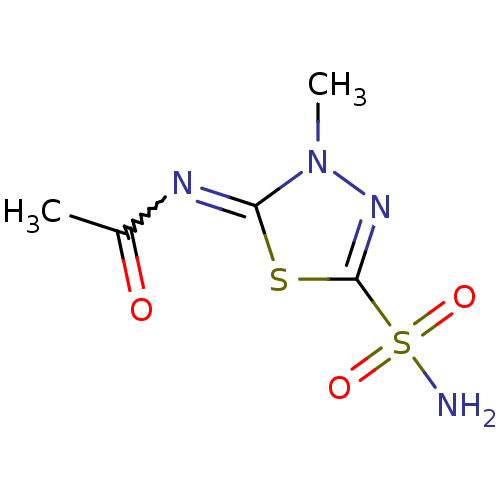
(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157952

(2-Morpholin-4-yl-N-(5-sulfamoyl-[1,3,4]thiadiazol-...)Show InChI InChI=1S/C8H13N5O4S2/c9-19(15,16)8-12-11-7(18-8)10-6(14)5-13-1-3-17-4-2-13/h1-5H2,(H2,9,15,16)(H,10,11,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10880

(AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...)Show InChI InChI=1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10883

(4,5-dichlorobenzene-1,3-disulfonamide | CHEMBL17 |...)Show InChI InChI=1S/C6H6Cl2N2O4S2/c7-4-1-3(15(9,11)12)2-5(6(4)8)16(10,13)14/h1-2H,(H2,9,11,12)(H2,10,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| DrugBank
Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10881

(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157949

(2-(4-Methyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C9H16N6O3S2/c1-14-2-4-15(5-3-14)6-7(16)11-8-12-13-9(19-8)20(10,17)18/h2-6H2,1H3,(H2,10,17,18)(H,11,12,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157950

(3-(4-Methyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C10H18N6O3S2/c1-15-4-6-16(7-5-15)3-2-8(17)12-9-13-14-10(20-9)21(11,18)19/h2-7H2,1H3,(H2,11,18,19)(H,12,13,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10883

(4,5-dichlorobenzene-1,3-disulfonamide | CHEMBL17 |...)Show InChI InChI=1S/C6H6Cl2N2O4S2/c7-4-1-3(15(9,11)12)2-5(6(4)8)16(10,13)14/h1-2H,(H2,9,11,12)(H2,10,13,14) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157955

(2-(4-Methyl-piperidin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show InChI InChI=1S/C10H17N5O3S2/c1-7-2-4-15(5-3-7)6-8(16)12-9-13-14-10(19-9)20(11,17)18/h7H,2-6H2,1H3,(H2,11,17,18)(H,12,13,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10882
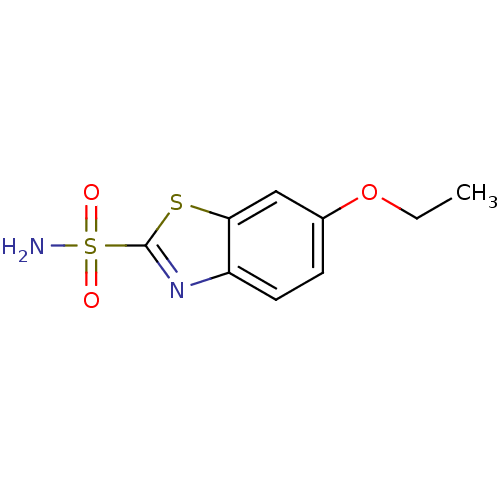
(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10868
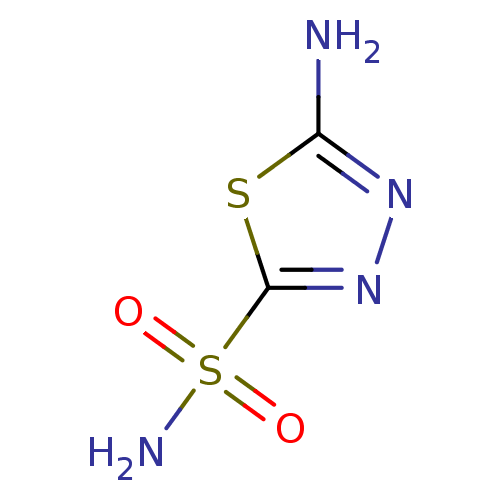
(1,3,4-Thiadiazole-2-sulfonamide, 6 | 1,3,4-thiadia...)Show InChI InChI=1S/C2H4N4O2S2/c3-1-5-6-2(9-1)10(4,7)8/h(H2,3,5)(H2,4,7,8) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10882

(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10884

((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...)Show SMILES CCN[C@H]1C[C@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C10H16N2O4S3/c1-3-12-8-4-6(2)18(13,14)10-7(8)5-9(17-10)19(11,15)16/h5-6,8,12H,3-4H2,1-2H3,(H2,11,15,16)/t6-,8-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10868

(1,3,4-Thiadiazole-2-sulfonamide, 6 | 1,3,4-thiadia...)Show InChI InChI=1S/C2H4N4O2S2/c3-1-5-6-2(9-1)10(4,7)8/h(H2,3,5)(H2,4,7,8) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| PDB
Article
PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157958

(2-(4-Benzyl-piperazin-1-yl)-N-(5-sulfamoyl-[1,3,4]...)Show SMILES NS(=O)(=O)c1nnc(NC(=O)CN2CCN(Cc3ccccc3)CC2)s1 Show InChI InChI=1S/C15H20N6O3S2/c16-26(23,24)15-19-18-14(25-15)17-13(22)11-21-8-6-20(7-9-21)10-12-4-2-1-3-5-12/h1-5H,6-11H2,(H2,16,23,24)(H,17,18,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157945

(5-Morpholin-4-yl-pentanoic acid (4-sulfamoyl-pheny...)Show InChI InChI=1S/C15H23N3O4S/c16-23(20,21)14-6-4-13(5-7-14)17-15(19)3-1-2-8-18-9-11-22-12-10-18/h4-7H,1-3,8-12H2,(H,17,19)(H2,16,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 77 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157951

(3-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-propionami...)Show InChI InChI=1S/C13H19N3O4S/c14-21(18,19)12-3-1-11(2-4-12)15-13(17)5-6-16-7-9-20-10-8-16/h1-4H,5-10H2,(H,15,17)(H2,14,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 82 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157946

(2-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-acetamide ...)Show InChI InChI=1S/C12H17N3O4S/c13-20(17,18)11-3-1-10(2-4-11)14-12(16)9-15-5-7-19-8-6-15/h1-4H,5-9H2,(H,14,16)(H2,13,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 87 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157954

(4-(4-Methyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C16H25N3O3S/c1-13-8-11-19(12-9-13)10-2-3-16(20)18-14-4-6-15(7-5-14)23(17,21)22/h4-7,13H,2-3,8-12H2,1H3,(H,18,20)(H2,17,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157956
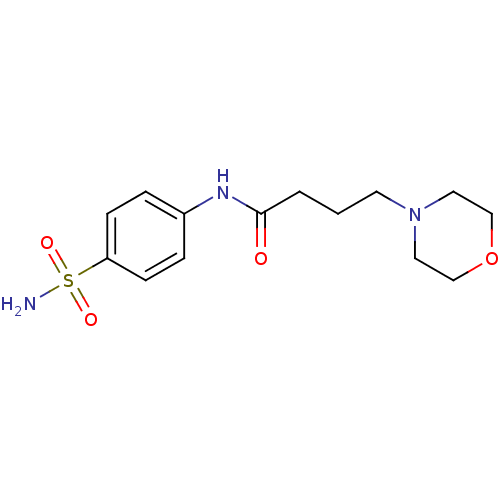
(4-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-butyramide...)Show InChI InChI=1S/C14H21N3O4S/c15-22(19,20)13-5-3-12(4-6-13)16-14(18)2-1-7-17-8-10-21-11-9-17/h3-6H,1-2,7-11H2,(H,16,18)(H2,15,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 104 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157945

(5-Morpholin-4-yl-pentanoic acid (4-sulfamoyl-pheny...)Show InChI InChI=1S/C15H23N3O4S/c16-23(20,21)14-6-4-13(5-7-14)17-15(19)3-1-2-8-18-9-11-22-12-10-18/h4-7H,1-3,8-12H2,(H,17,19)(H2,16,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157953
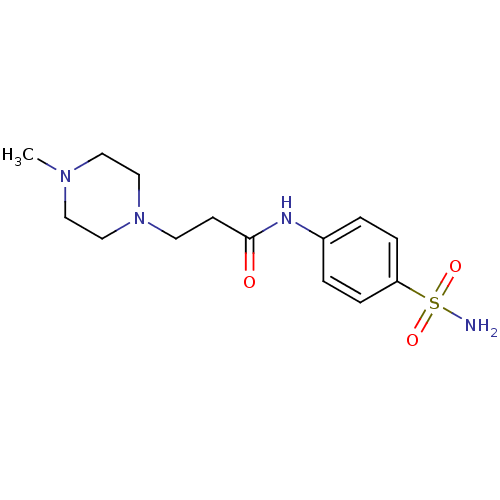
(3-(4-Methyl-piperazin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C14H22N4O3S/c1-17-8-10-18(11-9-17)7-6-14(19)16-12-2-4-13(5-3-12)22(15,20)21/h2-5H,6-11H2,1H3,(H,16,19)(H2,15,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 165 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157948

(3-(4-Methyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C15H23N3O3S/c1-12-6-9-18(10-7-12)11-8-15(19)17-13-2-4-14(5-3-13)22(16,20)21/h2-5,12H,6-11H2,1H3,(H,17,19)(H2,16,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 173 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157946

(2-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-acetamide ...)Show InChI InChI=1S/C12H17N3O4S/c13-20(17,18)11-3-1-10(2-4-11)14-12(16)9-15-5-7-19-8-6-15/h1-4H,5-9H2,(H,14,16)(H2,13,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 181 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157951

(3-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-propionami...)Show InChI InChI=1S/C13H19N3O4S/c14-21(18,19)12-3-1-11(2-4-12)15-13(17)5-6-16-7-9-20-10-8-16/h1-4H,5-10H2,(H,15,17)(H2,14,18,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 184 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157948

(3-(4-Methyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C15H23N3O3S/c1-12-6-9-18(10-7-12)11-8-15(19)17-13-2-4-14(5-3-13)22(16,20)21/h2-5,12H,6-11H2,1H3,(H,17,19)(H2,16,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 197 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157957
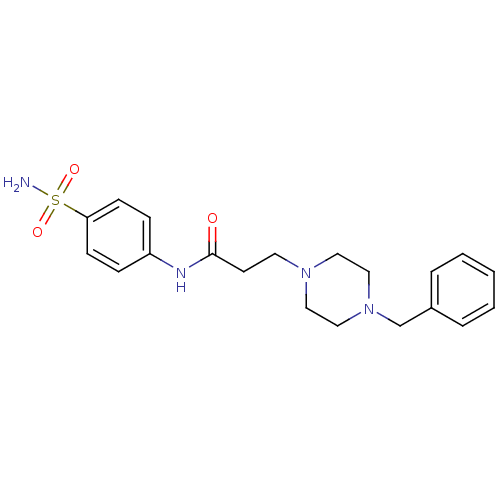
(3-(4-Benzyl-piperazin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CCN2CCN(Cc3ccccc3)CC2)cc1 Show InChI InChI=1S/C20H26N4O3S/c21-28(26,27)19-8-6-18(7-9-19)22-20(25)10-11-23-12-14-24(15-13-23)16-17-4-2-1-3-5-17/h1-9H,10-16H2,(H,22,25)(H2,21,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 234 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157953

(3-(4-Methyl-piperazin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C14H22N4O3S/c1-17-8-10-18(11-9-17)7-6-14(19)16-12-2-4-13(5-3-12)22(15,20)21/h2-5H,6-11H2,1H3,(H,16,19)(H2,15,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 235 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157957

(3-(4-Benzyl-piperazin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CCN2CCN(Cc3ccccc3)CC2)cc1 Show InChI InChI=1S/C20H26N4O3S/c21-28(26,27)19-8-6-18(7-9-19)22-20(25)10-11-23-12-14-24(15-13-23)16-17-4-2-1-3-5-17/h1-9H,10-16H2,(H,22,25)(H2,21,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157956

(4-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-butyramide...)Show InChI InChI=1S/C14H21N3O4S/c15-22(19,20)13-5-3-12(4-6-13)16-14(18)2-1-7-17-8-10-21-11-9-17/h3-6H,1-2,7-11H2,(H,16,18)(H2,15,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 246 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM50157947

(3-(4-Benzyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CCN2CCC(Cc3ccccc3)CC2)cc1 Show InChI InChI=1S/C21H27N3O3S/c22-28(26,27)20-8-6-19(7-9-20)23-21(25)12-15-24-13-10-18(11-14-24)16-17-4-2-1-3-5-17/h1-9,18H,10-16H2,(H,23,25)(H2,22,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 248 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157947

(3-(4-Benzyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CCN2CCC(Cc3ccccc3)CC2)cc1 Show InChI InChI=1S/C21H27N3O3S/c22-28(26,27)20-8-6-19(7-9-20)23-21(25)12-15-24-13-10-18(11-14-24)16-17-4-2-1-3-5-17/h1-9,18H,10-16H2,(H,23,25)(H2,22,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 258 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM50157954

(4-(4-Methyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C16H25N3O3S/c1-13-8-11-19(12-9-13)10-2-3-16(20)18-14-4-6-15(7-5-14)23(17,21)22/h4-7,13H,2-3,8-12H2,1H3,(H,18,20)(H2,17,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 265 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 9
(Homo sapiens (Human)) | BDBM10857
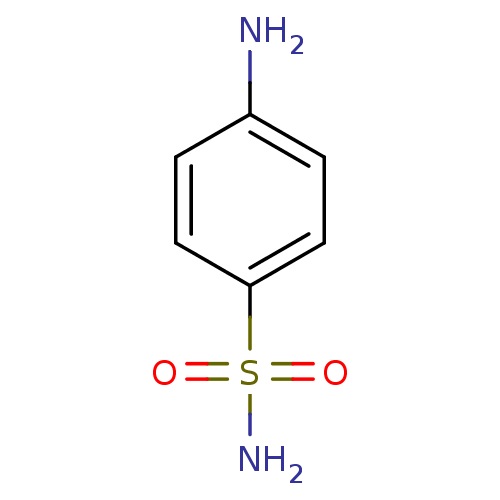
(4-aminobenzene-1-sulfonamide | CHEMBL21 | Sulfanil...)Show InChI InChI=1S/C6H8N2O2S/c7-5-1-3-6(4-2-5)11(8,9)10/h1-4H,7H2,(H2,8,9,10) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 294 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against catalytic domain of human Carbonic anhydrase IX expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 2
(Homo sapiens (Human)) | BDBM10857

(4-aminobenzene-1-sulfonamide | CHEMBL21 | Sulfanil...)Show InChI InChI=1S/C6H8N2O2S/c7-5-1-3-6(4-2-5)11(8,9)10/h1-4H,7H2,(H2,8,9,10) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase II expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157956

(4-Morpholin-4-yl-N-(4-sulfamoyl-phenyl)-butyramide...)Show InChI InChI=1S/C14H21N3O4S/c15-22(19,20)13-5-3-12(4-6-13)16-14(18)2-1-7-17-8-10-21-11-9-17/h3-6H,1-2,7-11H2,(H,16,18)(H2,15,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 371 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157948

(3-(4-Methyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C15H23N3O3S/c1-12-6-9-18(10-7-12)11-8-15(19)17-13-2-4-14(5-3-13)22(16,20)21/h2-5,12H,6-11H2,1H3,(H,17,19)(H2,16,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 381 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157957

(3-(4-Benzyl-piperazin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CCN2CCN(Cc3ccccc3)CC2)cc1 Show InChI InChI=1S/C20H26N4O3S/c21-28(26,27)19-8-6-18(7-9-19)22-20(25)10-11-23-12-14-24(15-13-23)16-17-4-2-1-3-5-17/h1-9H,10-16H2,(H,22,25)(H2,21,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 549 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157953

(3-(4-Methyl-piperazin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C14H22N4O3S/c1-17-8-10-18(11-9-17)7-6-14(19)16-12-2-4-13(5-3-12)22(15,20)21/h2-5H,6-11H2,1H3,(H,16,19)(H2,15,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 644 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157947

(3-(4-Benzyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CCN2CCC(Cc3ccccc3)CC2)cc1 Show InChI InChI=1S/C21H27N3O3S/c22-28(26,27)20-8-6-19(7-9-20)23-21(25)12-15-24-13-10-18(11-14-24)16-17-4-2-1-3-5-17/h1-9,18H,10-16H2,(H,23,25)(H2,22,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 752 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10881

(CHEMBL288100 | MZA3 | Methazolamide | Methazolamid...)Show InChI InChI=1S/C5H8N4O3S2/c1-3(10)7-4-9(2)8-5(13-4)14(6,11)12/h1-2H3,(H2,6,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM50157954

(4-(4-Methyl-piperidin-1-yl)-N-(4-sulfamoyl-phenyl)...)Show InChI InChI=1S/C16H25N3O3S/c1-13-8-11-19(12-9-13)10-2-3-16(20)18-14-4-6-15(7-5-14)23(17,21)22/h4-7,13H,2-3,8-12H2,1H3,(H,18,20)(H2,17,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10880

(AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...)Show InChI InChI=1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10882

(6-ethoxy-1,3-benzothiazole-2-sulfonamide | CHEMBL1...)Show InChI InChI=1S/C9H10N2O3S2/c1-2-14-6-3-4-7-8(5-6)15-9(11-7)16(10,12)13/h3-5H,2H2,1H3,(H2,10,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10868

(1,3,4-Thiadiazole-2-sulfonamide, 6 | 1,3,4-thiadia...)Show InChI InChI=1S/C2H4N4O2S2/c3-1-5-6-2(9-1)10(4,7)8/h(H2,3,5)(H2,4,7,8) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 8.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10857

(4-aminobenzene-1-sulfonamide | CHEMBL21 | Sulfanil...)Show InChI InChI=1S/C6H8N2O2S/c7-5-1-3-6(4-2-5)11(8,9)10/h1-4H,7H2,(H2,8,9,10) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |
Carbonic anhydrase 1
(Homo sapiens (Human)) | BDBM10884

((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...)Show SMILES CCN[C@H]1C[C@H](C)S(=O)(=O)c2sc(cc12)S(N)(=O)=O |r| Show InChI InChI=1S/C10H16N2O4S3/c1-3-12-8-4-6(2)18(13,14)10-7(8)5-9(17-10)19(11,15)16/h5-6,8,12H,3-4H2,1-2H3,(H2,11,15,16)/t6-,8-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harran University
Curated by ChEMBL
| Assay Description
Inhibitory potency against human cloned Carbonic anhydrase I expressed in Escherichia coli strain BL21 |
Bioorg Med Chem Lett 15: 367-72 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.070
BindingDB Entry DOI: 10.7270/Q2B858WB |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data