 Found 13 hits of Enzyme Inhibition Constant Data
Found 13 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50182212
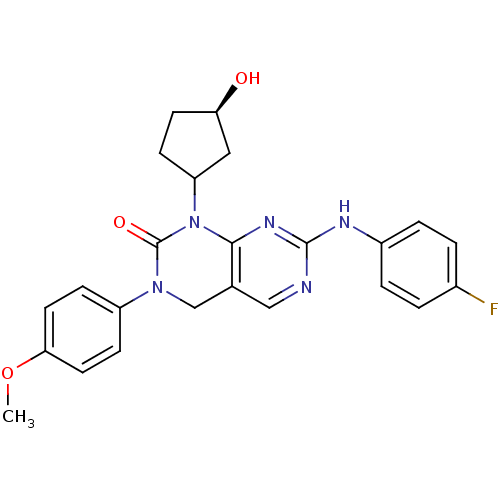
(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against recombinant human KDR |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against recombinant human PDGFR |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1/2/3/4
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 92 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against recombinant human FGFR |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Fyn
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 466 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Fyn |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 545 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against recombinant human EGFR |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Ephrin type-B receptor 3
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against EphB3 |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
cAMP-dependent protein kinase catalytic subunit alpha/beta/gamma
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.01E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against PKA |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 1
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Erk2 |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.25E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against AKT |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.25E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against PKC delta |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.25E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against PKC alpha |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 1/G2/mitotic-specific cyclin-B1/G2/mitotic-specific cyclin-B2/G2/mitotic-specific cyclin-B3
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.25E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CDK1/cyclinB |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Sgk1
(Homo sapiens (Human)) | BDBM50182212

(7-(4-fluorophenylamino)-1-((1S,3R)-3-hydroxycyclop...)Show SMILES COc1ccc(cc1)N1Cc2cnc(Nc3ccc(F)cc3)nc2N(C2CC[C@@H](O)C2)C1=O Show InChI InChI=1S/C24H24FN5O3/c1-33-21-10-7-18(8-11-21)29-14-15-13-26-23(27-17-4-2-16(25)3-5-17)28-22(15)30(24(29)32)19-6-9-20(31)12-19/h2-5,7-8,10-11,13,19-20,31H,6,9,12,14H2,1H3,(H,26,27,28)/t19?,20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.25E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffmann-La Roche Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against SGK |
Bioorg Med Chem Lett 16: 1950-3 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.092
BindingDB Entry DOI: 10.7270/Q2S46SRS |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data