 Found 29 hits of Enzyme Inhibition Constant Data
Found 29 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183217
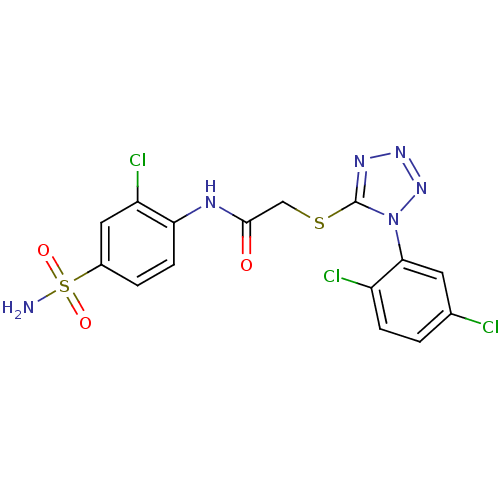
(CHEMBL206522 | N-(2-chloro-4-sulfamoyl-phenyl)-2-[...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CSc2nnnn2-c2cc(Cl)ccc2Cl)c(Cl)c1 Show InChI InChI=1S/C15H11Cl3N6O3S2/c16-8-1-3-10(17)13(5-8)24-15(21-22-23-24)28-7-14(25)20-12-4-2-9(6-11(12)18)29(19,26)27/h1-6H,7H2,(H,20,25)(H2,19,26,27) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183196
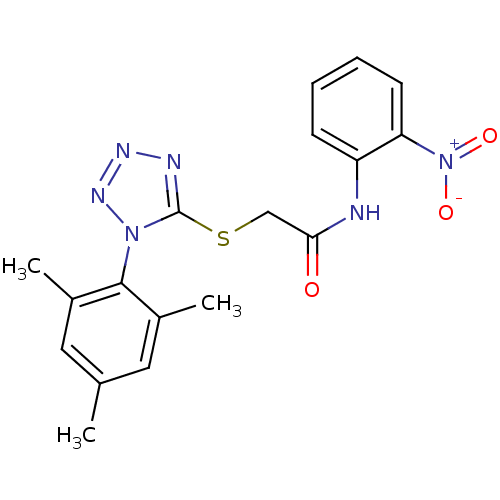
(2-(1-mesityl-1H-tetrazol-5-ylthio)-N-(2-nitropheny...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccccc1[N+]([O-])=O |(-7.83,-15.14,;-7.5,-13.63,;-8.64,-12.59,;-8.3,-11.09,;-9.44,-10.05,;-6.84,-10.62,;-5.7,-11.65,;-4.23,-11.18,;-6.03,-13.17,;-6.51,-9.11,;-7.54,-7.96,;-6.77,-6.63,;-5.26,-6.95,;-5.11,-8.49,;-3.77,-9.26,;-2.44,-8.49,;-1.11,-9.26,;-1.11,-10.8,;.23,-8.5,;1.56,-9.27,;1.55,-10.81,;2.88,-11.58,;4.22,-10.81,;4.22,-9.27,;2.89,-8.5,;2.88,-6.95,;4.21,-6.18,;1.55,-6.18,)| Show InChI InChI=1S/C18H18N6O3S/c1-11-8-12(2)17(13(3)9-11)23-18(20-21-22-23)28-10-16(25)19-14-6-4-5-7-15(14)24(26)27/h4-9H,10H2,1-3H3,(H,19,25) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183199
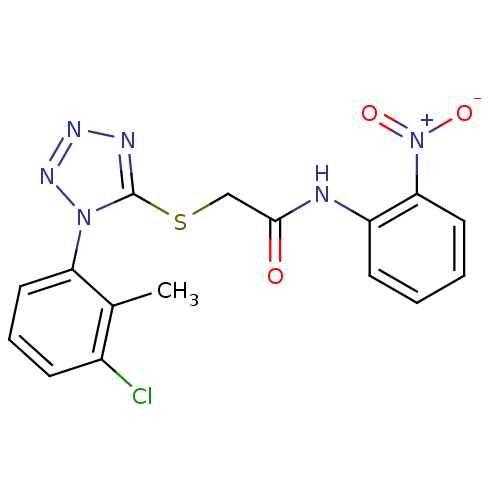
(2-(1-(3-chloro-2-methylphenyl)-1H-tetrazol-5-ylthi...)Show SMILES Cc1c(Cl)cccc1-n1nnnc1SCC(=O)Nc1ccccc1[N+]([O-])=O Show InChI InChI=1S/C16H13ClN6O3S/c1-10-11(17)5-4-8-13(10)22-16(19-20-21-22)27-9-15(24)18-12-6-2-3-7-14(12)23(25)26/h2-8H,9H2,1H3,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183200
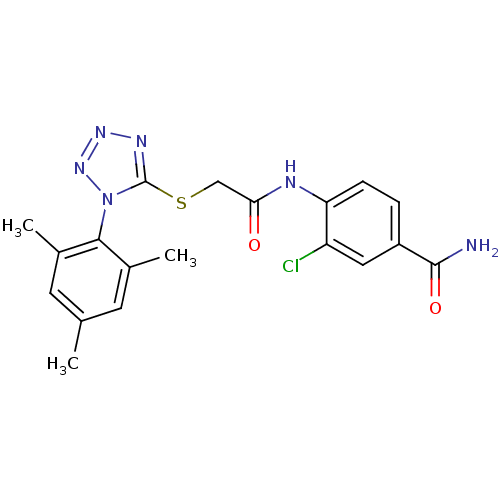
(3-chloro-4-(2-(1-mesityl-1H-tetrazol-5-ylthio)acet...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1Cl)C(N)=O |(14.82,-23.12,;15.16,-21.62,;14.02,-20.58,;14.35,-19.07,;13.22,-18.03,;15.82,-18.61,;16.96,-19.64,;18.42,-19.17,;16.63,-21.15,;16.14,-17.1,;15.12,-15.95,;15.89,-14.61,;17.4,-14.94,;17.55,-16.47,;18.88,-17.25,;20.22,-16.48,;21.55,-17.25,;21.55,-18.79,;22.88,-16.48,;24.22,-17.26,;24.21,-18.8,;25.54,-19.57,;26.88,-18.8,;26.87,-17.25,;25.54,-16.49,;25.54,-14.95,;28.21,-19.57,;29.54,-18.8,;28.21,-21.11,)| Show InChI InChI=1S/C19H19ClN6O2S/c1-10-6-11(2)17(12(3)7-10)26-19(23-24-25-26)29-9-16(27)22-15-5-4-13(18(21)28)8-14(15)20/h4-8H,9H2,1-3H3,(H2,21,28)(H,22,27) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183190
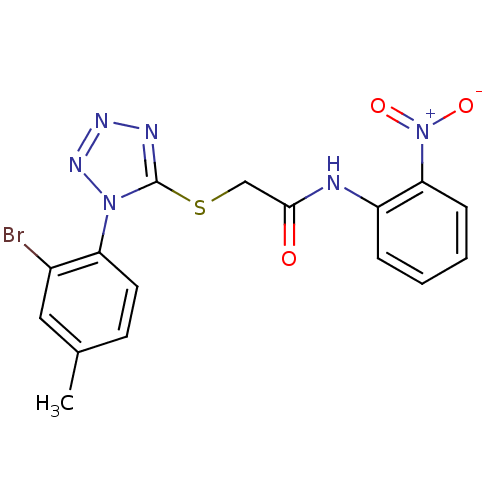
(2-(1-(2-bromo-4-methylphenyl)-1H-tetrazol-5-ylthio...)Show SMILES Cc1ccc(c(Br)c1)-n1nnnc1SCC(=O)Nc1ccccc1[N+]([O-])=O Show InChI InChI=1S/C16H13BrN6O3S/c1-10-6-7-13(11(17)8-10)22-16(19-20-21-22)27-9-15(24)18-12-4-2-3-5-14(12)23(25)26/h2-8H,9H2,1H3,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183210
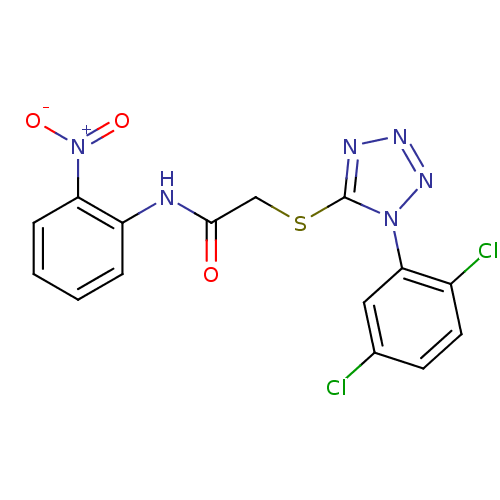
(2-(1-(2,5-dichlorophenyl)-1H-tetrazol-5-ylthio)-N-...)Show SMILES [O-][N+](=O)c1ccccc1NC(=O)CSc1nnnn1-c1cc(Cl)ccc1Cl Show InChI InChI=1S/C15H10Cl2N6O3S/c16-9-5-6-10(17)13(7-9)22-15(19-20-21-22)27-8-14(24)18-11-3-1-2-4-12(11)23(25)26/h1-7H,8H2,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183211
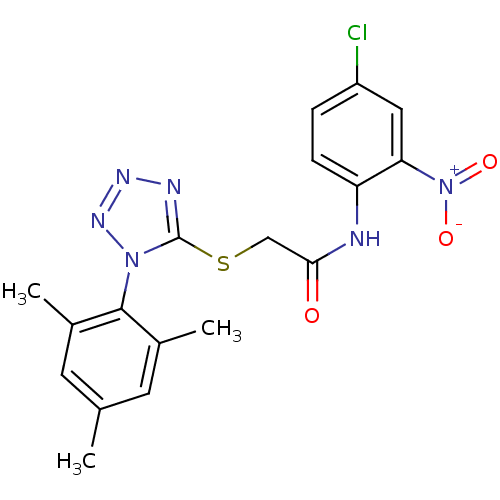
(CHEMBL205152 | N-(4-chloro-2-nitrophenyl)-2-(1-mes...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(Cl)cc1[N+]([O-])=O |(-7.25,-49.05,;-6.92,-47.54,;-8.05,-46.5,;-7.72,-45,;-8.86,-43.96,;-6.26,-44.53,;-5.12,-45.56,;-3.65,-45.09,;-5.45,-47.08,;-5.93,-43.02,;-6.96,-41.87,;-6.18,-40.54,;-4.68,-40.86,;-4.52,-42.4,;-3.19,-43.17,;-1.86,-42.4,;-.52,-43.17,;-.53,-44.71,;.81,-42.41,;2.14,-43.18,;2.14,-44.72,;3.47,-45.49,;4.8,-44.72,;6.14,-45.5,;4.8,-43.18,;3.47,-42.41,;3.47,-40.86,;4.8,-40.09,;2.13,-40.09,)| Show InChI InChI=1S/C18H17ClN6O3S/c1-10-6-11(2)17(12(3)7-10)24-18(21-22-23-24)29-9-16(26)20-14-5-4-13(19)8-15(14)25(27)28/h4-8H,9H2,1-3H3,(H,20,26) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183193
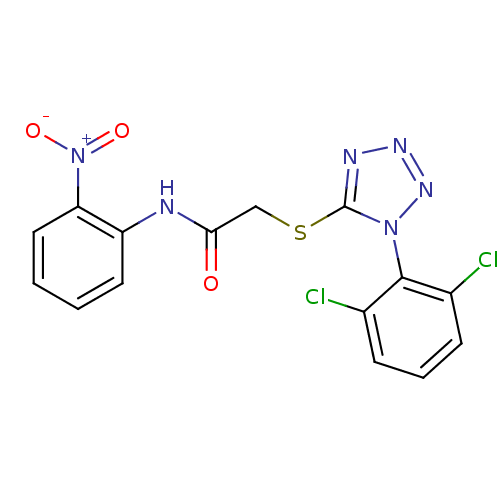
(2-(1-(2,6-dichlorophenyl)-1H-tetrazol-5-ylthio)-N-...)Show SMILES [O-][N+](=O)c1ccccc1NC(=O)CSc1nnnn1-c1c(Cl)cccc1Cl |(6.73,-27,;5.4,-27.77,;4.07,-27,;5.41,-29.32,;6.74,-30.09,;6.74,-31.63,;5.4,-32.4,;4.07,-31.63,;4.08,-30.09,;2.75,-29.32,;1.41,-30.08,;1.41,-31.62,;.08,-29.31,;-1.25,-30.08,;-2.59,-29.31,;-2.74,-27.77,;-4.25,-27.45,;-5.02,-28.78,;-3.99,-29.93,;-4.32,-31.44,;-3.18,-32.47,;-1.71,-31.99,;-3.49,-33.97,;-4.97,-34.45,;-6.11,-33.41,;-5.78,-31.91,;-6.92,-30.87,)| Show InChI InChI=1S/C15H10Cl2N6O3S/c16-9-4-3-5-10(17)14(9)22-15(19-20-21-22)27-8-13(24)18-11-6-1-2-7-12(11)23(25)26/h1-7H,8H2,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183201
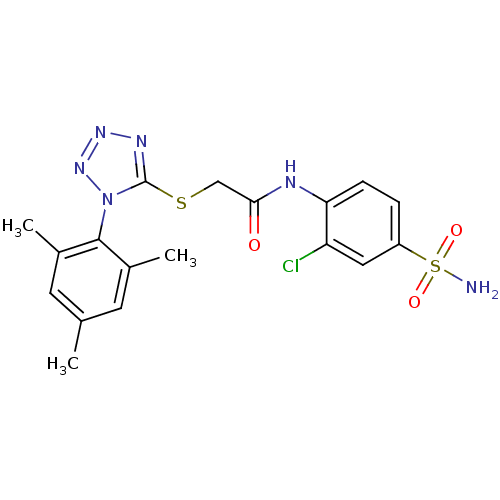
(CHEMBL379722 | N-(2-chloro-4-sulfamoyl-phenyl)-2-[...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1Cl)S(N)(=O)=O |(15.06,-12.69,;15.39,-11.18,;14.25,-10.14,;14.59,-8.64,;13.45,-7.6,;16.05,-8.17,;17.19,-9.2,;18.66,-8.73,;16.86,-10.72,;16.38,-6.66,;15.35,-5.51,;16.12,-4.18,;17.63,-4.5,;17.78,-6.04,;19.12,-6.81,;20.45,-6.04,;21.78,-6.81,;21.78,-8.35,;23.12,-6.05,;24.45,-6.82,;24.44,-8.36,;25.77,-9.13,;27.11,-8.36,;27.11,-6.82,;25.78,-6.05,;25.77,-4.51,;28.44,-9.13,;29.77,-9.9,;27.67,-10.47,;29.22,-7.8,)| Show InChI InChI=1S/C18H19ClN6O3S2/c1-10-6-11(2)17(12(3)7-10)25-18(22-23-24-25)29-9-16(26)21-15-5-4-13(8-14(15)19)30(20,27)28/h4-8H,9H2,1-3H3,(H,21,26)(H2,20,27,28) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183198
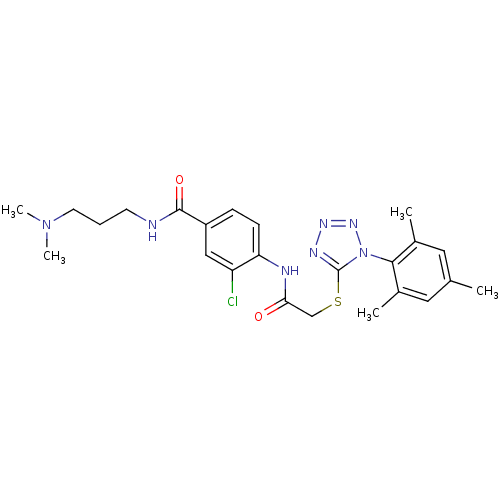
(3-chloro-N-(3-(dimethylamino)propyl)-4-(2-(1-mesit...)Show SMILES CN(C)CCCNC(=O)c1ccc(NC(=O)CSc2nnnn2-c2c(C)cc(C)cc2C)c(Cl)c1 |(11.95,-30.47,;10.62,-29.7,;10.62,-28.16,;9.29,-30.47,;7.95,-29.7,;6.62,-30.47,;5.29,-29.7,;3.95,-30.47,;3.95,-32.01,;2.62,-29.7,;1.28,-30.47,;-.05,-29.69,;-.04,-28.15,;-1.37,-27.38,;-2.71,-28.15,;-2.71,-29.69,;-4.04,-27.38,;-5.38,-28.14,;-6.71,-27.37,;-6.86,-25.84,;-8.37,-25.51,;-9.14,-26.84,;-8.12,-28,;-8.44,-29.5,;-9.91,-29.97,;-11.04,-28.93,;-10.24,-31.48,;-9.1,-32.52,;-9.43,-34.02,;-7.63,-32.05,;-7.3,-30.54,;-5.84,-30.07,;1.28,-27.38,;1.28,-25.84,;2.62,-28.15,)| Show InChI InChI=1S/C24H30ClN7O2S/c1-15-11-16(2)22(17(3)12-15)32-24(28-29-30-32)35-14-21(33)27-20-8-7-18(13-19(20)25)23(34)26-9-6-10-31(4)5/h7-8,11-13H,6,9-10,14H2,1-5H3,(H,26,34)(H,27,33) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM27606
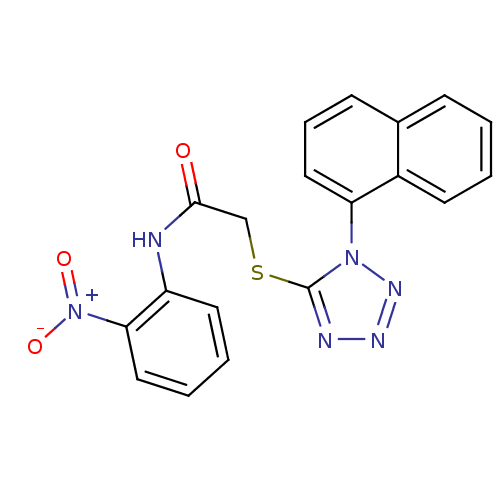
(2-{[1-(naphthalen-1-yl)-1H-1,2,3,4-tetrazol-5-yl]s...)Show SMILES [O-][N+](=O)c1ccccc1NC(=O)CSc1nnnn1-c1cccc2ccccc12 Show InChI InChI=1S/C19H14N6O3S/c26-18(20-15-9-3-4-10-17(15)25(27)28)12-29-19-21-22-23-24(19)16-11-5-7-13-6-1-2-8-14(13)16/h1-11H,12H2,(H,20,26) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183207
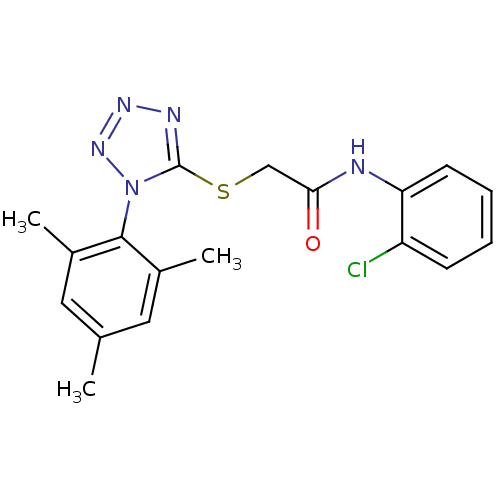
(CHEMBL377010 | N-(2-chlorophenyl)-2-(1-mesityl-1H-...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccccc1Cl |(7.91,-30.7,;8.21,-29.19,;7.04,-28.18,;7.34,-26.66,;6.17,-25.65,;8.79,-26.15,;9.95,-27.16,;11.41,-26.65,;9.67,-28.68,;9.07,-24.63,;8.02,-23.51,;8.76,-22.16,;10.27,-22.45,;10.46,-23.98,;11.81,-24.72,;13.13,-23.92,;14.48,-24.66,;14.52,-26.2,;15.8,-23.86,;17.15,-24.6,;17.18,-26.14,;18.52,-26.88,;19.84,-26.08,;19.8,-24.53,;18.45,-23.8,;18.41,-22.26,)| Show InChI InChI=1S/C18H18ClN5OS/c1-11-8-12(2)17(13(3)9-11)24-18(21-22-23-24)26-10-16(25)20-15-7-5-4-6-14(15)19/h4-9H,10H2,1-3H3,(H,20,25) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183206
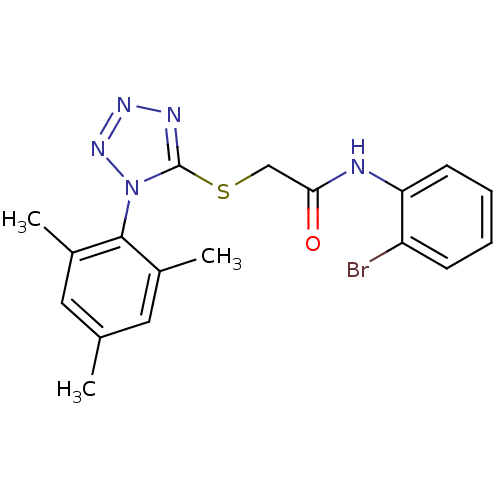
(CHEMBL381513 | N-(2-bromophenyl)-2-(1-mesityl-1H-t...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccccc1Br |(15.19,-14.32,;15.52,-12.82,;14.38,-11.78,;14.72,-10.27,;13.58,-9.23,;16.18,-9.8,;17.32,-10.84,;18.79,-10.36,;16.99,-12.35,;16.51,-8.3,;15.48,-7.14,;16.26,-5.81,;17.76,-6.14,;17.92,-7.67,;19.25,-8.44,;20.58,-7.67,;21.92,-8.45,;21.91,-9.99,;23.25,-7.68,;24.58,-8.45,;24.58,-9.99,;25.91,-10.76,;27.24,-10,;27.24,-8.45,;25.91,-7.68,;25.91,-6.14,)| Show InChI InChI=1S/C18H18BrN5OS/c1-11-8-12(2)17(13(3)9-11)24-18(21-22-23-24)26-10-16(25)20-15-7-5-4-6-14(15)19/h4-9H,10H2,1-3H3,(H,20,25) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183212
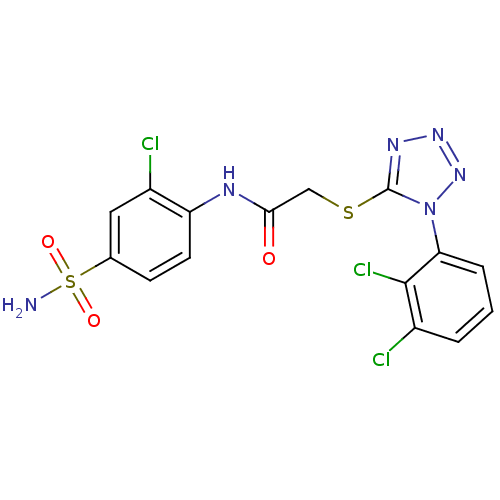
(CHEMBL383088 | N-(2-chloro-4-sulfamoyl-phenyl)-2-[...)Show SMILES NS(=O)(=O)c1ccc(NC(=O)CSc2nnnn2-c2cccc(Cl)c2Cl)c(Cl)c1 Show InChI InChI=1S/C15H11Cl3N6O3S2/c16-9-2-1-3-12(14(9)18)24-15(21-22-23-24)28-7-13(25)20-11-5-4-8(6-10(11)17)29(19,26)27/h1-6H,7H2,(H,20,25)(H2,19,26,27) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183213
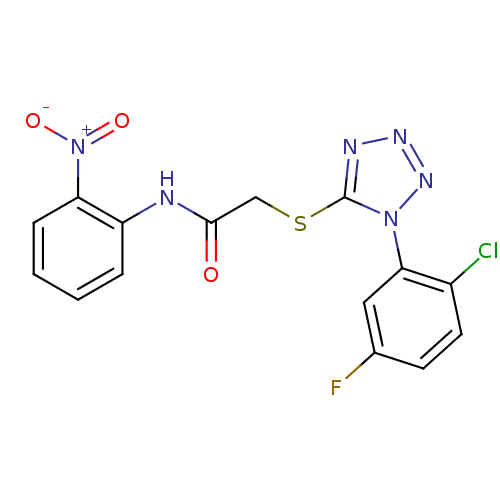
(2-(1-(2-chloro-5-fluorophenyl)-1H-tetrazol-5-ylthi...)Show SMILES [O-][N+](=O)c1ccccc1NC(=O)CSc1nnnn1-c1cc(F)ccc1Cl Show InChI InChI=1S/C15H10ClFN6O3S/c16-10-6-5-9(17)7-13(10)22-15(19-20-21-22)27-8-14(24)18-11-3-1-2-4-12(11)23(25)26/h1-7H,8H2,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183205
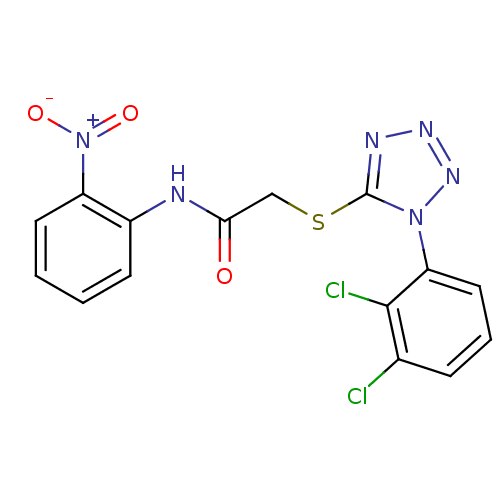
(2-(1-(2,3-dichlorophenyl)-1H-tetrazol-5-ylthio)-N-...)Show SMILES [O-][N+](=O)c1ccccc1NC(=O)CSc1nnnn1-c1cccc(Cl)c1Cl Show InChI InChI=1S/C15H10Cl2N6O3S/c16-9-4-3-7-12(14(9)17)22-15(19-20-21-22)27-8-13(24)18-10-5-1-2-6-11(10)23(25)26/h1-7H,8H2,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183192
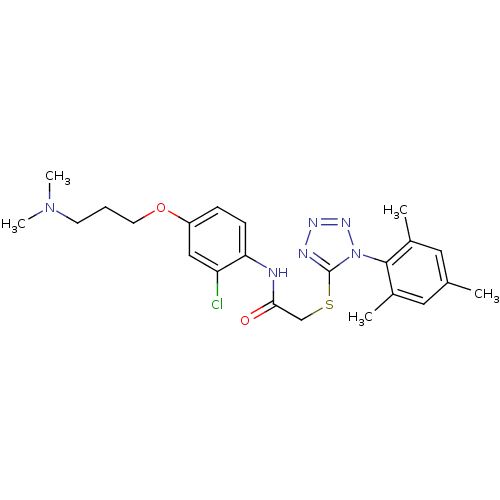
(CHEMBL377820 | N-(2-chloro-4-(3-(dimethylamino)pro...)Show SMILES CN(C)CCCOc1ccc(NC(=O)CSc2nnnn2-c2c(C)cc(C)cc2C)c(Cl)c1 |(29.31,-36.4,;29.31,-34.86,;30.65,-34.09,;27.98,-34.09,;27.98,-32.55,;29.31,-31.78,;29.32,-30.24,;27.98,-29.47,;26.64,-30.24,;25.31,-29.47,;25.32,-27.93,;23.99,-27.15,;22.65,-27.92,;22.65,-29.46,;21.32,-27.15,;19.99,-27.92,;18.65,-27.14,;18.5,-25.61,;17,-25.29,;16.22,-26.62,;17.25,-27.77,;16.92,-29.28,;15.46,-29.74,;14.32,-28.71,;15.12,-31.25,;16.26,-32.29,;15.93,-33.8,;17.73,-31.83,;18.06,-30.31,;19.53,-29.84,;26.65,-27.16,;26.65,-25.62,;27.98,-27.92,)| Show InChI InChI=1S/C23H29ClN6O2S/c1-15-11-16(2)22(17(3)12-15)30-23(26-27-28-30)33-14-21(31)25-20-8-7-18(13-19(20)24)32-10-6-9-29(4)5/h7-8,11-13H,6,9-10,14H2,1-5H3,(H,25,31) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183191

(2-(1-(2,3-dimethylphenyl)-1H-tetrazol-5-ylthio)-N-...)Show SMILES Cc1cccc(c1C)-n1nnnc1SCC(=O)Nc1ccccc1[N+]([O-])=O Show InChI InChI=1S/C17H16N6O3S/c1-11-6-5-9-14(12(11)2)22-17(19-20-21-22)27-10-16(24)18-13-7-3-4-8-15(13)23(25)26/h3-9H,10H2,1-2H3,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183209
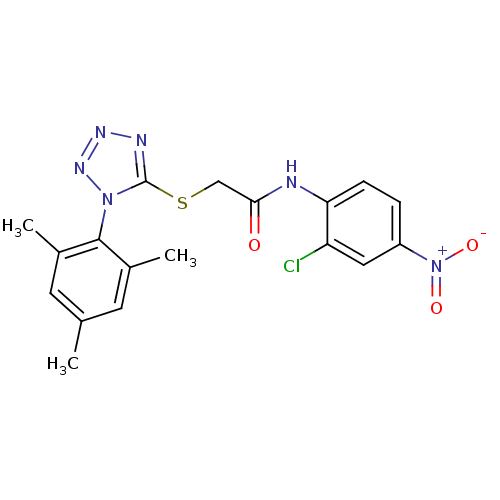
(CHEMBL204993 | N-(2-chloro-4-nitrophenyl)-2-(1-mes...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1Cl)[N+]([O-])=O |(14.87,-48.33,;15.2,-46.83,;14.07,-45.79,;14.4,-44.28,;13.26,-43.24,;15.86,-43.81,;17,-44.85,;18.47,-44.38,;16.67,-46.36,;16.19,-42.31,;15.16,-41.15,;15.94,-39.82,;17.44,-40.15,;17.6,-41.68,;18.93,-42.45,;20.26,-41.69,;21.6,-42.46,;21.59,-44,;22.93,-41.69,;24.26,-42.46,;24.26,-44,;25.59,-44.78,;26.92,-44.01,;26.92,-42.46,;25.59,-41.7,;25.59,-40.16,;28.26,-44.78,;28.26,-46.32,;29.59,-44.01,)| Show InChI InChI=1S/C18H17ClN6O3S/c1-10-6-11(2)17(12(3)7-10)24-18(21-22-23-24)29-9-16(26)20-15-5-4-13(25(27)28)8-14(15)19/h4-8H,9H2,1-3H3,(H,20,26) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183215
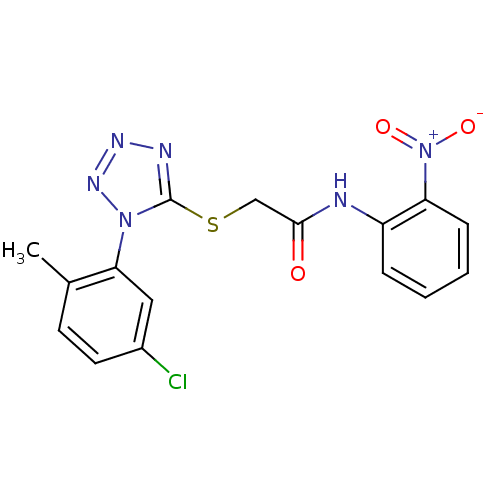
(2-(1-(5-chloro-2-methylphenyl)-1H-tetrazol-5-ylthi...)Show SMILES Cc1ccc(Cl)cc1-n1nnnc1SCC(=O)Nc1ccccc1[N+]([O-])=O Show InChI InChI=1S/C16H13ClN6O3S/c1-10-6-7-11(17)8-14(10)22-16(19-20-21-22)27-9-15(24)18-12-4-2-3-5-13(12)23(25)26/h2-8H,9H2,1H3,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183197

(CHEMBL205999 | N-(2-iodophenyl)-2-(1-mesityl-1H-te...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccccc1I |(-7.14,-25.92,;-6.81,-24.42,;-7.95,-23.38,;-7.61,-21.87,;-8.75,-20.83,;-6.15,-21.41,;-5.01,-22.44,;-3.54,-21.97,;-5.34,-23.95,;-5.82,-19.9,;-6.85,-18.75,;-6.07,-17.41,;-4.57,-17.74,;-4.41,-19.27,;-3.08,-20.05,;-1.75,-19.28,;-.41,-20.05,;-.42,-21.59,;.92,-19.28,;2.25,-20.06,;2.25,-21.6,;3.58,-22.37,;4.91,-21.6,;4.91,-20.05,;3.58,-19.29,;3.58,-17.75,)| Show InChI InChI=1S/C18H18IN5OS/c1-11-8-12(2)17(13(3)9-11)24-18(21-22-23-24)26-10-16(25)20-15-7-5-4-6-14(15)19/h4-9H,10H2,1-3H3,(H,20,25) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183204
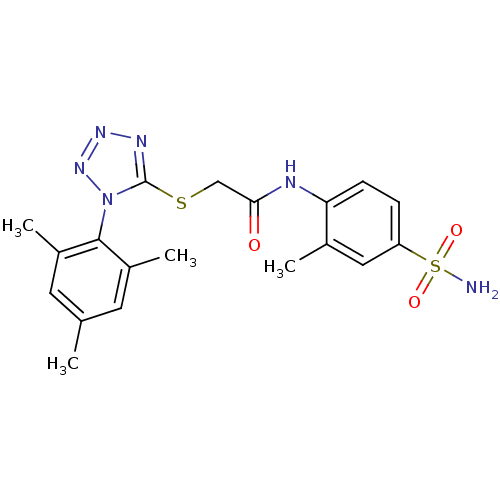
(CHEMBL208057 | N-(2-methyl-4-sulfamoyl-phenyl)-2-[...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1C)S(N)(=O)=O |(-6.98,-13.11,;-6.65,-11.6,;-7.79,-10.56,;-7.46,-9.06,;-8.59,-8.02,;-5.99,-8.59,;-4.85,-9.62,;-3.39,-9.15,;-5.18,-11.14,;-5.67,-7.08,;-6.69,-5.93,;-5.92,-4.6,;-4.41,-4.92,;-4.26,-6.46,;-2.93,-7.23,;-1.59,-6.46,;-.26,-7.23,;-.26,-8.77,;1.08,-6.47,;2.41,-7.24,;2.4,-8.78,;3.73,-9.55,;5.07,-8.78,;5.07,-7.24,;3.73,-6.47,;3.73,-4.93,;6.4,-9.55,;7.73,-10.32,;5.63,-10.89,;7.18,-8.22,)| Show InChI InChI=1S/C19H22N6O3S2/c1-11-7-13(3)18(14(4)8-11)25-19(22-23-24-25)29-10-17(26)21-16-6-5-15(9-12(16)2)30(20,27)28/h5-9H,10H2,1-4H3,(H,21,26)(H2,20,27,28) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183202
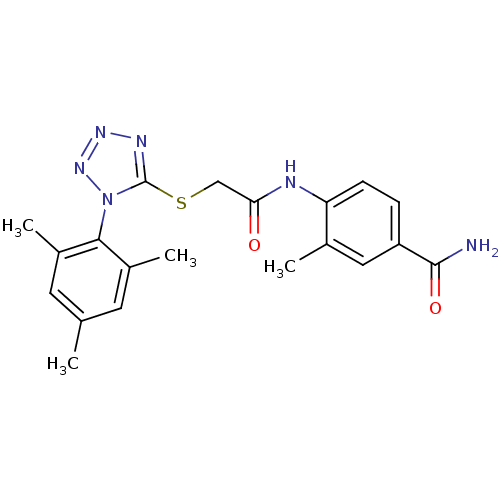
(4-(2-(1-mesityl-1H-tetrazol-5-ylthio)acetamido)-3-...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1C)C(N)=O |(-6.75,-23.61,;-6.42,-22.1,;-7.56,-21.06,;-7.22,-19.56,;-8.36,-18.52,;-5.76,-19.09,;-4.62,-20.12,;-3.15,-19.65,;-4.95,-21.64,;-5.43,-17.58,;-6.46,-16.43,;-5.68,-15.1,;-4.18,-15.42,;-4.03,-16.96,;-2.69,-17.73,;-1.36,-16.96,;-.03,-17.73,;-.03,-19.27,;1.31,-16.97,;2.64,-17.74,;2.63,-19.28,;3.96,-20.05,;5.3,-19.28,;5.3,-17.74,;3.97,-16.97,;3.97,-15.43,;6.64,-20.05,;7.97,-19.28,;6.63,-21.59,)| Show InChI InChI=1S/C20H22N6O2S/c1-11-7-13(3)18(14(4)8-11)26-20(23-24-25-26)29-10-17(27)22-16-6-5-15(19(21)28)9-12(16)2/h5-9H,10H2,1-4H3,(H2,21,28)(H,22,27) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183194

(2-(1-mesityl-1H-tetrazol-5-ylthio)-N-(2-methyl-4-n...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1C)[N+]([O-])=O |(-7.4,-3.06,;-7.06,-1.55,;-8.2,-.51,;-7.87,.99,;-9.01,2.03,;-6.4,1.46,;-5.26,.43,;-3.8,.9,;-5.59,-1.09,;-6.08,2.97,;-7.11,4.12,;-6.33,5.45,;-4.83,5.13,;-4.67,3.59,;-3.34,2.82,;-2,3.59,;-.67,2.82,;-.67,1.28,;.66,3.58,;2,2.81,;1.99,1.27,;3.32,.5,;4.66,1.27,;4.65,2.81,;3.32,3.58,;3.32,5.12,;5.99,.49,;5.99,-1.05,;7.33,1.26,)| Show InChI InChI=1S/C19H20N6O3S/c1-11-7-13(3)18(14(4)8-11)24-19(21-22-23-24)29-10-17(26)20-16-6-5-15(25(27)28)9-12(16)2/h5-9H,10H2,1-4H3,(H,20,26) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 165 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183216
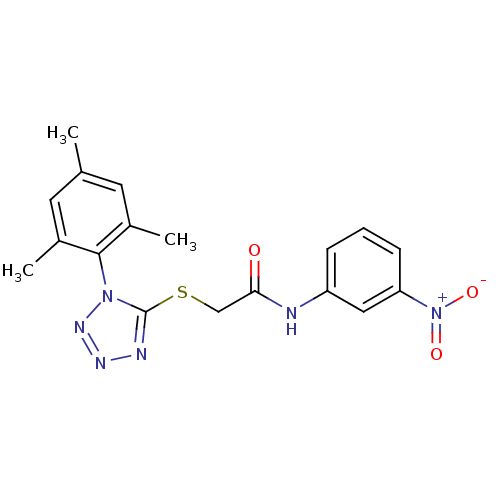
(2-(1-mesityl-1H-tetrazol-5-ylthio)-N-(3-nitropheny...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1cccc(c1)[N+]([O-])=O |(14.54,-25.45,;14.88,-23.95,;13.74,-22.91,;14.07,-21.4,;12.94,-20.36,;15.54,-20.93,;16.68,-21.97,;18.14,-21.49,;16.35,-23.48,;15.86,-19.43,;14.84,-18.27,;15.61,-16.94,;17.12,-17.27,;17.27,-18.8,;18.6,-19.57,;19.94,-18.8,;21.27,-19.58,;21.27,-21.12,;22.6,-18.81,;23.94,-19.58,;23.93,-21.12,;25.26,-21.89,;26.6,-21.13,;26.59,-19.58,;25.26,-18.81,;27.94,-18.8,;29.27,-19.57,;27.93,-17.26,)| Show InChI InChI=1S/C18H18N6O3S/c1-11-7-12(2)17(13(3)8-11)23-18(20-21-22-23)28-10-16(25)19-14-5-4-6-15(9-14)24(26)27/h4-9H,10H2,1-3H3,(H,19,25) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183208
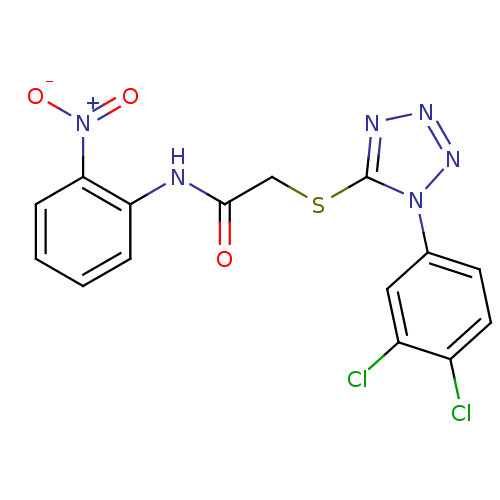
(2-(1-(3,4-dichlorophenyl)-1H-tetrazol-5-ylthio)-N-...)Show SMILES [O-][N+](=O)c1ccccc1NC(=O)CSc1nnnn1-c1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C15H10Cl2N6O3S/c16-10-6-5-9(7-11(10)17)22-15(19-20-21-22)27-8-14(24)18-12-3-1-2-4-13(12)23(25)26/h1-7H,8H2,(H,18,24) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183203
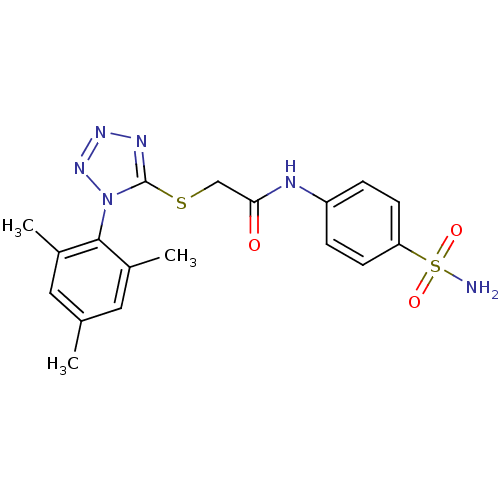
(CHEMBL208008 | N-(4-sulfamoyl-phenyl)-2-[1-(2,4,6-...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1)S(N)(=O)=O |(13.95,-2.13,;14.28,-.63,;13.14,.41,;13.47,1.92,;12.34,2.96,;14.94,2.39,;16.08,1.35,;17.54,1.82,;15.75,-.16,;15.26,3.89,;14.24,5.05,;15.01,6.38,;16.52,6.05,;16.67,4.52,;18,3.75,;19.34,4.51,;20.67,3.74,;20.67,2.2,;22.01,4.51,;23.34,3.74,;23.33,2.2,;24.66,1.42,;26,2.19,;26,3.74,;24.66,4.5,;27.33,1.42,;28.66,.65,;26.56,.09,;28.11,2.75,)| Show InChI InChI=1S/C18H20N6O3S2/c1-11-8-12(2)17(13(3)9-11)24-18(21-22-23-24)28-10-16(25)20-14-4-6-15(7-5-14)29(19,26)27/h4-9H,10H2,1-3H3,(H,20,25)(H2,19,26,27) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183214
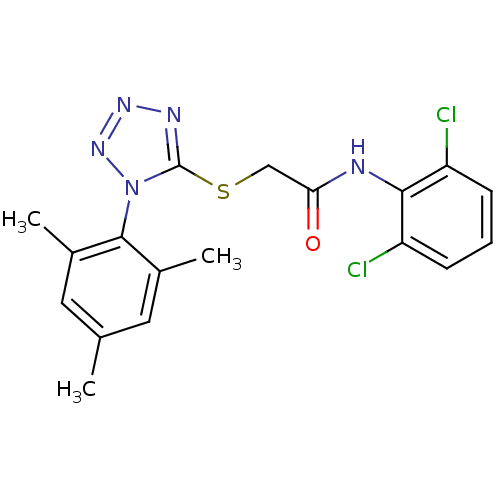
(CHEMBL205094 | N-(2,6-dichlorophenyl)-2-(1-mesityl...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1c(Cl)cccc1Cl |(15.42,-36.3,;15.76,-34.8,;14.62,-33.76,;14.95,-32.25,;13.81,-31.21,;16.42,-31.78,;17.56,-32.82,;19.02,-32.34,;17.23,-34.33,;16.74,-30.28,;15.71,-29.12,;16.49,-27.79,;17.99,-28.12,;18.15,-29.65,;19.48,-30.42,;20.82,-29.65,;22.15,-30.43,;22.15,-31.97,;23.48,-29.66,;24.82,-30.43,;26.14,-29.66,;26.14,-28.12,;27.47,-30.43,;27.48,-31.98,;26.14,-32.74,;24.81,-31.97,;23.47,-32.74,)| Show InChI InChI=1S/C18H17Cl2N5OS/c1-10-7-11(2)17(12(3)8-10)25-18(22-23-24-25)27-9-15(26)21-16-13(19)5-4-6-14(16)20/h4-8H,9H2,1-3H3,(H,21,26) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |
Reverse transcriptase/RNaseH
(Human immunodeficiency virus 1) | BDBM50183195

(2-(1-mesityl-1H-tetrazol-5-ylthio)-N-(4-nitropheny...)Show SMILES Cc1cc(C)c(c(C)c1)-n1nnnc1SCC(=O)Nc1ccc(cc1)[N+]([O-])=O |(-7.23,-36.04,;-6.89,-34.53,;-8.03,-33.49,;-7.7,-31.98,;-8.83,-30.95,;-6.23,-31.52,;-5.09,-32.55,;-3.63,-32.08,;-5.42,-34.07,;-5.91,-30.01,;-6.93,-28.86,;-6.16,-27.53,;-4.65,-27.85,;-4.5,-29.38,;-3.17,-30.16,;-1.83,-29.39,;-.5,-30.16,;-.5,-31.7,;.83,-29.39,;2.17,-30.17,;2.16,-31.71,;3.49,-32.48,;4.83,-31.71,;4.82,-30.16,;3.49,-29.4,;6.16,-32.49,;6.16,-34.03,;7.5,-31.72,)| Show InChI InChI=1S/C18H18N6O3S/c1-11-8-12(2)17(13(3)9-11)23-18(20-21-22-23)28-10-16(25)19-14-4-6-15(7-5-14)24(26)27/h4-9H,10H2,1-3H3,(H,19,25) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
MRL Rome
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 wild type Reverse Transcriptase |
Bioorg Med Chem Lett 16: 2748-52 (2006)
Article DOI: 10.1016/j.bmcl.2006.02.024
BindingDB Entry DOI: 10.7270/Q2D79B1R |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data