 Found 15 hits of Enzyme Inhibition Constant Data
Found 15 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50148911
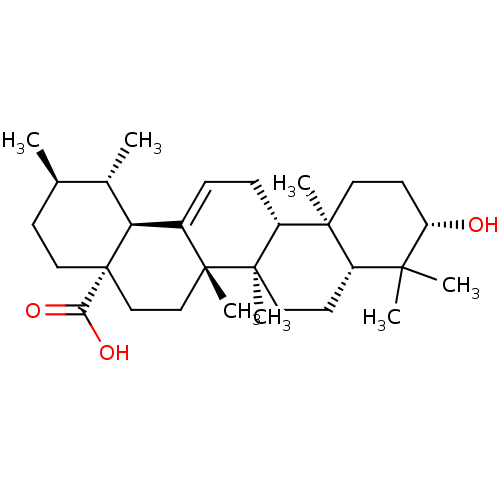
((3beta)-3-hydroxyurs-12-en-28-oic acid | 3beta-hyd...)Show SMILES C[C@@H]1CC[C@@]2(CC[C@]3(C)C(=CC[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5CC[C@@]34C)[C@@H]2[C@H]1C)C(O)=O |r,c:9| Show InChI InChI=1S/C30H48O3/c1-18-10-15-30(25(32)33)17-16-28(6)20(24(30)19(18)2)8-9-22-27(5)13-12-23(31)26(3,4)21(27)11-14-29(22,28)7/h8,18-19,21-24,31H,9-17H2,1-7H3,(H,32,33)/t18-,19+,21+,22-,23+,24+,27+,28-,29-,30+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50104694
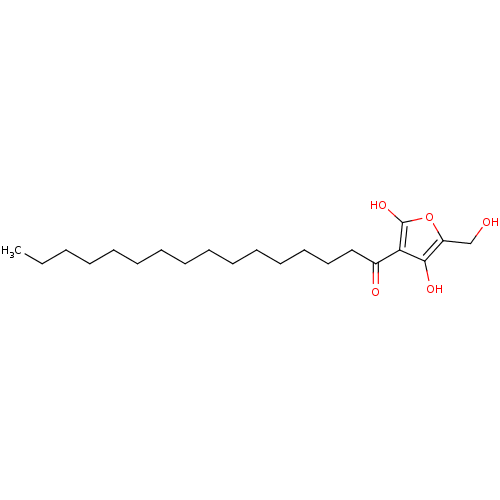
((R)-4-hydroxy-5-(hydroxymethyl)-3-palmitoylfuran-2...)Show InChI InChI=1S/C21H36O5/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-17(23)19-20(24)18(16-22)26-21(19)25/h22,24-25H,2-16H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241807

(CHEMBL470865 | abyssinone-VI-4-O-methyl ether)Show SMILES [#6]-[#8]-c1c(-[#6]\[#6]=[#6](\[#6])-[#6])cc(\[#6]=[#6]\[#6](=O)-c2ccc(-[#8])cc2-[#8])cc1-[#6]\[#6]=[#6](\[#6])-[#6] Show InChI InChI=1S/C26H30O4/c1-17(2)6-9-20-14-19(15-21(26(20)30-5)10-7-18(3)4)8-13-24(28)23-12-11-22(27)16-25(23)29/h6-8,11-16,27,29H,9-10H2,1-5H3/b13-8+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.48E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241806
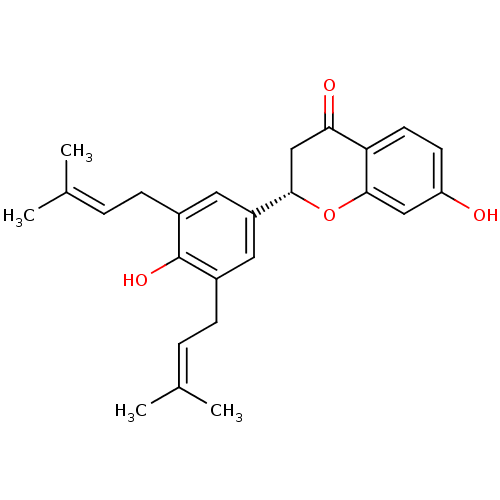
(CHEMBL470454 | abyssinone IV | abyssinone-IV)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(cc(-[#6]\[#6]=[#6](\[#6])-[#6])c1-[#8])-[#6@@H]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 |r| Show InChI InChI=1S/C25H28O4/c1-15(2)5-7-17-11-19(12-18(25(17)28)8-6-16(3)4)23-14-22(27)21-10-9-20(26)13-24(21)29-23/h5-6,9-13,23,26,28H,7-8,14H2,1-4H3/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241797
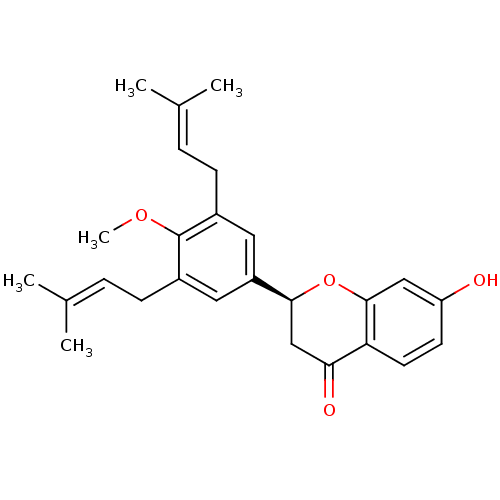
(CHEMBL470452 | abyssinone-IV-4'-O-methyl ether)Show SMILES [#6]-[#8]-c1c(-[#6]\[#6]=[#6](\[#6])-[#6])cc(cc1-[#6]\[#6]=[#6](\[#6])-[#6])-[#6@@H]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 |r| Show InChI InChI=1S/C26H30O4/c1-16(2)6-8-18-12-20(13-19(26(18)29-5)9-7-17(3)4)24-15-23(28)22-11-10-21(27)14-25(22)30-24/h6-7,10-14,24,27H,8-9,15H2,1-5H3/t24-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241796
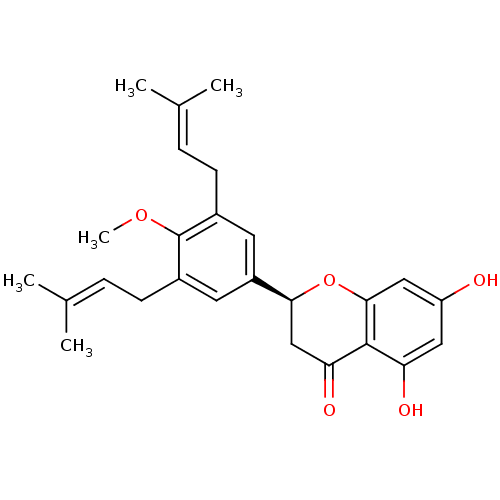
(4-methylabyssinone V | CHEMBL470249 | abyssinone-V...)Show SMILES [#6]-[#8]-c1c(-[#6]\[#6]=[#6](\[#6])-[#6])cc(cc1-[#6]\[#6]=[#6](\[#6])-[#6])-[#6@@H]-1-[#6]-[#6](=O)-c2c(-[#8])cc(-[#8])cc2-[#8]-1 |r| Show InChI InChI=1S/C26H30O5/c1-15(2)6-8-17-10-19(11-18(26(17)30-5)9-7-16(3)4)23-14-22(29)25-21(28)12-20(27)13-24(25)31-23/h6-7,10-13,23,27-28H,8-9,14H2,1-5H3/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.63E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241819
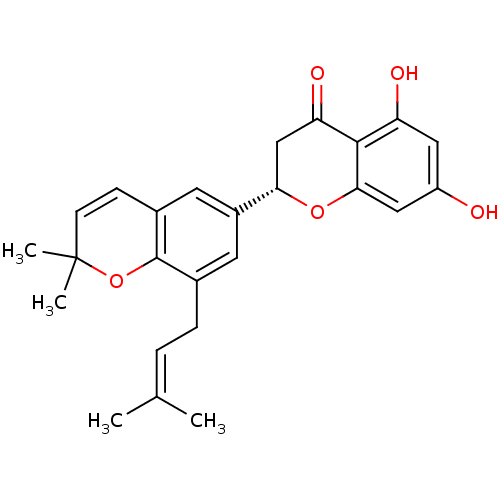
(CHEMBL470655 | sigmoidin E)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(cc2-[#6]=[#6]C([#6])([#6])[#8]-c12)-[#6@@H]-1-[#6]-[#6](=O)-c2c(-[#8])cc(-[#8])cc2-[#8]-1 |r,c:10| Show InChI InChI=1S/C25H26O5/c1-14(2)5-6-15-9-17(10-16-7-8-25(3,4)30-24(15)16)21-13-20(28)23-19(27)11-18(26)12-22(23)29-21/h5,7-12,21,26-27H,6,13H2,1-4H3/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.92E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241416
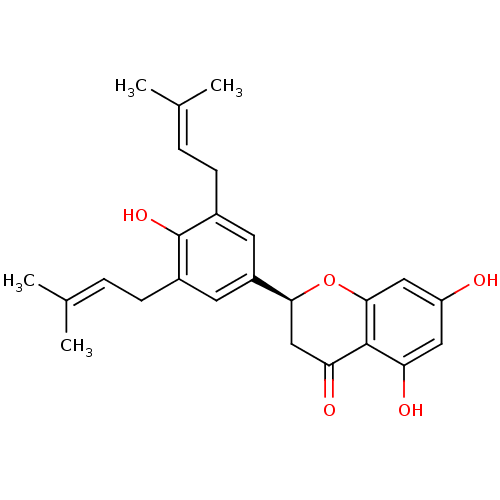
((2S)-5,7-dihydroxy-2-[4-hydroxy-3,5-bis(3-methylbu...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(cc(-[#6]\[#6]=[#6](\[#6])-[#6])c1-[#8])-[#6@@H]-1-[#6]-[#6](=O)-c2c(-[#8])cc(-[#8])cc2-[#8]-1 |r| Show InChI InChI=1S/C25H28O5/c1-14(2)5-7-16-9-18(10-17(25(16)29)8-6-15(3)4)22-13-21(28)24-20(27)11-19(26)12-23(24)30-22/h5-6,9-12,22,26-27,29H,7-8,13H2,1-4H3/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.97E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50200542
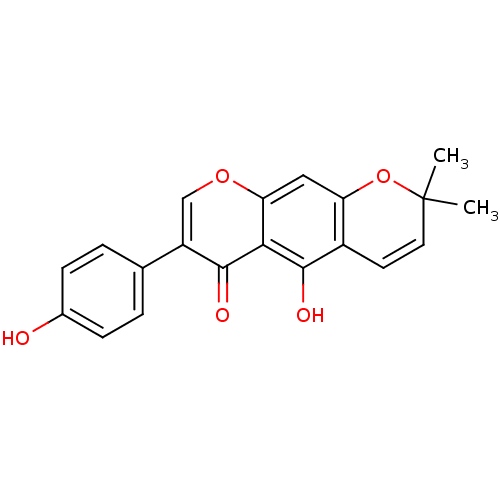
(5-hydroxy-3-(4-hydroxy-phenyl)-8,8-dimethyl-8H-pyr...)Show SMILES CC1(C)Oc2cc3occ(-c4ccc(O)cc4)c(=O)c3c(O)c2C=C1 |c:26| Show InChI InChI=1S/C20H16O5/c1-20(2)8-7-13-15(25-20)9-16-17(18(13)22)19(23)14(10-24-16)11-3-5-12(21)6-4-11/h3-10,21-22H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.15E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241820
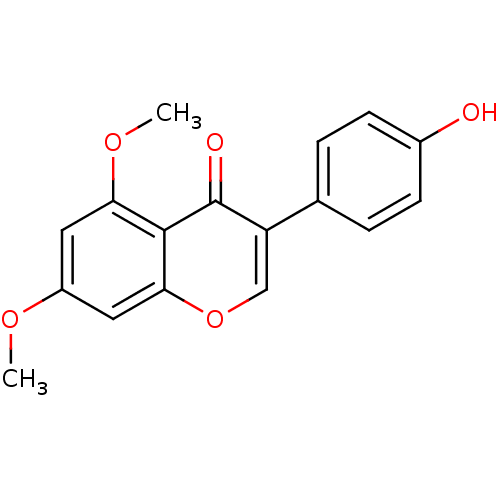
(4'-hydroxy-5,7-dimethoxyisoflavone | CHEMBL445815)Show InChI InChI=1S/C17H14O5/c1-20-12-7-14(21-2)16-15(8-12)22-9-13(17(16)19)10-3-5-11(18)6-4-10/h3-9,18H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50042944

((E)-1-(2,4-Dihydroxy-phenyl)-3-(4-hydroxy-phenyl)-...)Show InChI InChI=1S/C15H12O4/c16-11-4-1-10(2-5-11)3-8-14(18)13-7-6-12(17)9-15(13)19/h1-9,16-17,19H/b8-3+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241582
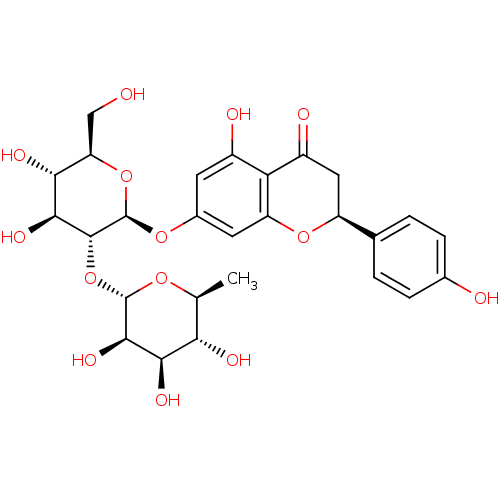
((S)-7-((2S,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxyme...)Show SMILES C[C@@H]1O[C@@H](O[C@@H]2[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]2Oc2cc(O)c3C(=O)C[C@H](Oc3c2)c2ccc(O)cc2)[C@H](O)[C@H](O)[C@H]1O |r| Show InChI InChI=1S/C27H32O14/c1-10-20(32)22(34)24(36)26(37-10)41-25-23(35)21(33)18(9-28)40-27(25)38-13-6-14(30)19-15(31)8-16(39-17(19)7-13)11-2-4-12(29)5-3-11/h2-7,10,16,18,20-30,32-36H,8-9H2,1H3/t10-,16-,18+,20-,21+,22+,23-,24+,25+,26-,27+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241805
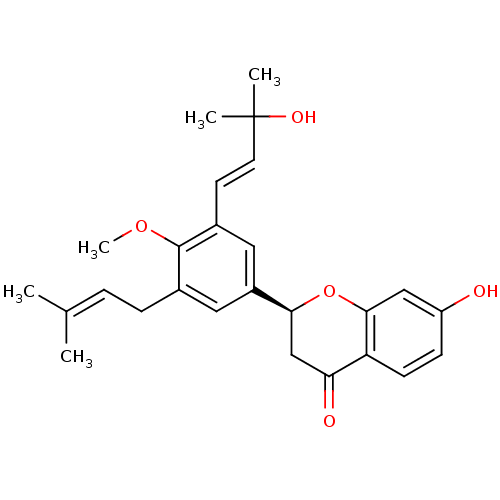
(7-hydroxy-4'-methoxy-3'-(3-hydroxy-3-methyl-trans-...)Show SMILES [#6]-[#8]-c1c(-[#6]\[#6]=[#6](\[#6])-[#6])cc(cc1\[#6]=[#6]\C([#6])([#6])[#8])-[#6@@H]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 |r| Show InChI InChI=1S/C26H30O5/c1-16(2)6-7-17-12-19(13-18(25(17)30-5)10-11-26(3,4)29)23-15-22(28)21-9-8-20(27)14-24(21)31-23/h6,8-14,23,27,29H,7,15H2,1-5H3/b11-10+/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50241408
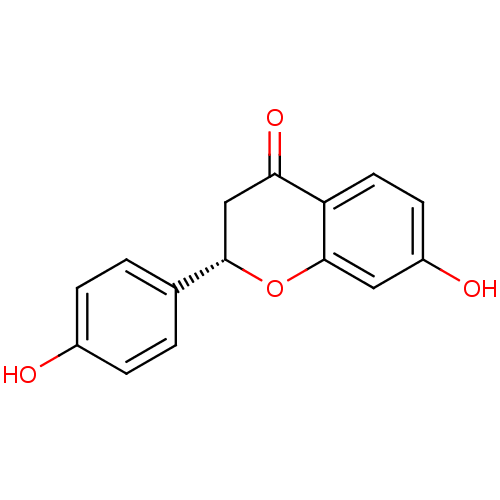
((2S)-liquiritigenin | 7-HYDROXY-2-(4-HYDROXY-PHENY...)Show InChI InChI=1S/C15H12O4/c16-10-3-1-9(2-4-10)14-8-13(18)12-6-5-11(17)7-15(12)19-14/h1-7,14,16-17H,8H2/t14-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50213034
(1-(2,4-Dihydroxy-phenyl)-3-(4-methoxy-phenyl)-prop...)Show InChI InChI=1S/C16H14O4/c1-20-13-6-2-11(3-7-13)4-9-15(18)14-8-5-12(17)10-16(14)19/h2-10,17,19H,1H3/b9-4+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PTP1B |
J Nat Prod 69: 1572-6 (2006)
Article DOI: 10.1021/np0601861
BindingDB Entry DOI: 10.7270/Q2B8591J |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data