 Found 7 hits of Enzyme Inhibition Constant Data
Found 7 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Aromatase
(Homo sapiens (Human)) | BDBM50213251

((rac)-7-hydroxy-2-(4-hydroxy-3-(3-methylbut-2-enyl...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(ccc1-[#8])-[#6]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 Show InChI InChI=1S/C20H20O4/c1-12(2)3-4-13-9-14(5-8-17(13)22)19-11-18(23)16-7-6-15(21)10-20(16)24-19/h3,5-10,19,21-22H,4,11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of aromatase by radiometric method |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50241582
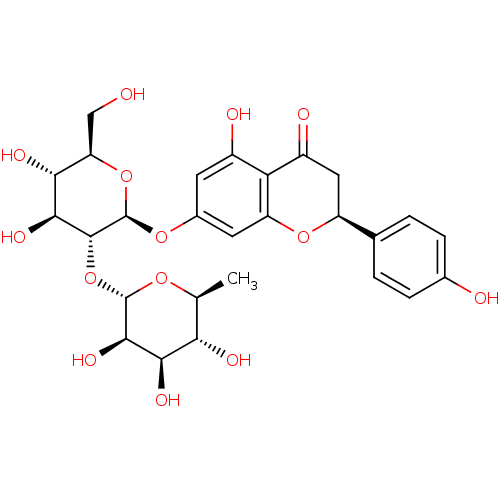
((S)-7-((2S,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxyme...)Show SMILES C[C@@H]1O[C@@H](O[C@@H]2[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]2Oc2cc(O)c3C(=O)C[C@H](Oc3c2)c2ccc(O)cc2)[C@H](O)[C@H](O)[C@H]1O |r| Show InChI InChI=1S/C27H32O14/c1-10-20(32)22(34)24(36)26(37-10)41-25-23(35)21(33)18(9-28)40-27(25)38-13-6-14(30)19-15(31)8-16(39-17(19)7-13)11-2-4-12(29)5-3-11/h2-7,10,16,18,20-30,32-36H,8-9H2,1H3/t10-,16-,18+,20-,21+,22+,23-,24+,25+,26-,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of aromatase |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A1
(Homo sapiens (Human)) | BDBM50271143

((3R,4R)-(-)-6-methoxy-1-oxo-3-pentyl-3,4-dihydro-1...)Show SMILES CCCCC[C@H]1OC(=O)c2ccc(OC)cc2[C@H]1OC(C)=O |r| Show InChI InChI=1S/C17H22O5/c1-4-5-6-7-15-16(21-11(2)18)14-10-12(20-3)8-9-13(14)17(19)22-15/h8-10,15-16H,4-7H2,1-3H3/t15-,16-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A1 |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213251

((rac)-7-hydroxy-2-(4-hydroxy-3-(3-methylbut-2-enyl...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(ccc1-[#8])-[#6]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 Show InChI InChI=1S/C20H20O4/c1-12(2)3-4-13-9-14(5-8-17(13)22)19-11-18(23)16-7-6-15(21)10-20(16)24-19/h3,5-10,19,21-22H,4,11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of aromatase by fluorimetric method |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213251

((rac)-7-hydroxy-2-(4-hydroxy-3-(3-methylbut-2-enyl...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(ccc1-[#8])-[#6]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 Show InChI InChI=1S/C20H20O4/c1-12(2)3-4-13-9-14(5-8-17(13)22)19-11-18(23)16-7-6-15(21)10-20(16)24-19/h3,5-10,19,21-22H,4,11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of aromatase by fluorimetric high throughput method |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50271143

((3R,4R)-(-)-6-methoxy-1-oxo-3-pentyl-3,4-dihydro-1...)Show SMILES CCCCC[C@H]1OC(=O)c2ccc(OC)cc2[C@H]1OC(C)=O |r| Show InChI InChI=1S/C17H22O5/c1-4-5-6-7-15-16(21-11(2)18)14-10-12(20-3)8-9-13(14)17(19)22-15/h8-10,15-16H,4-7H2,1-3H3/t15-,16-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >6.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C8 |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50271143

((3R,4R)-(-)-6-methoxy-1-oxo-3-pentyl-3,4-dihydro-1...)Show SMILES CCCCC[C@H]1OC(=O)c2ccc(OC)cc2[C@H]1OC(C)=O |r| Show InChI InChI=1S/C17H22O5/c1-4-5-6-7-15-16(21-11(2)18)14-10-12(20-3)8-9-13(14)17(19)22-15/h8-10,15-16H,4-7H2,1-3H3/t15-,16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >6.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidade Federal de Minas Gerais
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 |
J Nat Prod 71: 1082-4 (2008)
Article DOI: 10.1021/np800098f
BindingDB Entry DOI: 10.7270/Q2ZS2XF0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data