 Found 70 hits of Enzyme Inhibition Constant Data
Found 70 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Insulin receptor
(Homo sapiens (Human)) | BDBM50256480
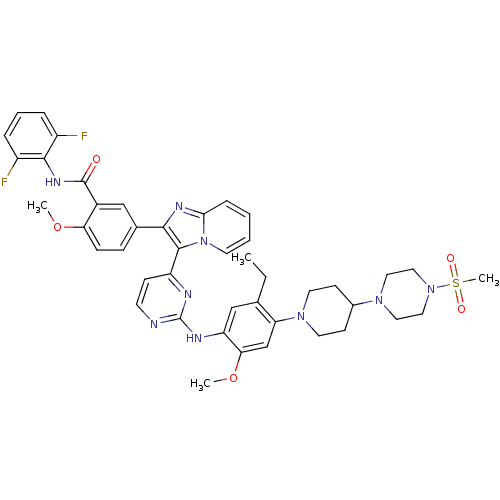
(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Binding affinity to insulin receptor by liquid scintillation counting |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Binding affinity to IGF1R by liquid scintillation counting |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50256478
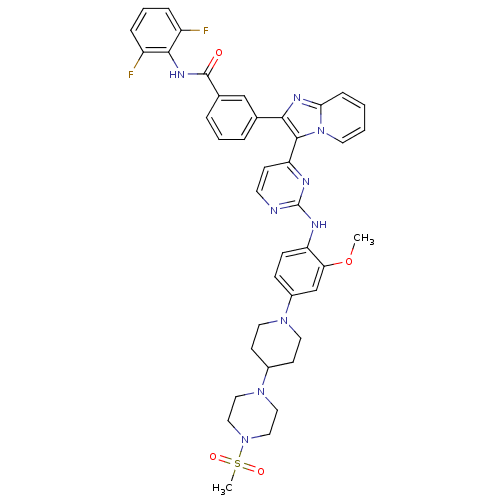
(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Binding affinity to insulin receptor by liquid scintillation counting |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Binding affinity to IGF1R by liquid scintillation counting |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256467
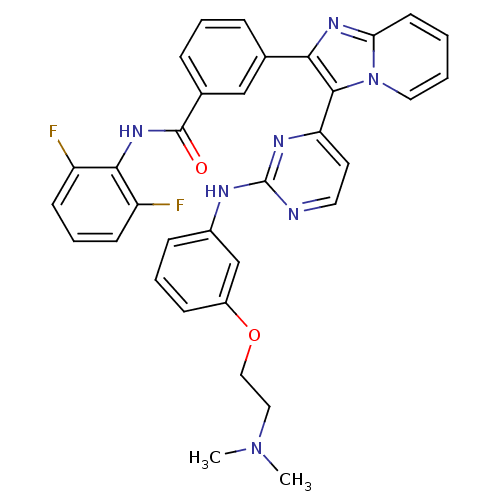
(CHEMBL448668 | N-(2,6-difluorophenyl)-3-(3-(2-(3-(...)Show SMILES CN(C)CCOc1cccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c1 Show InChI InChI=1S/C34H29F2N7O2/c1-42(2)18-19-45-25-11-6-10-24(21-25)38-34-37-16-15-28(39-34)32-30(40-29-14-3-4-17-43(29)32)22-8-5-9-23(20-22)33(44)41-31-26(35)12-7-13-27(31)36/h3-17,20-21H,18-19H2,1-2H3,(H,41,44)(H,37,38,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256469
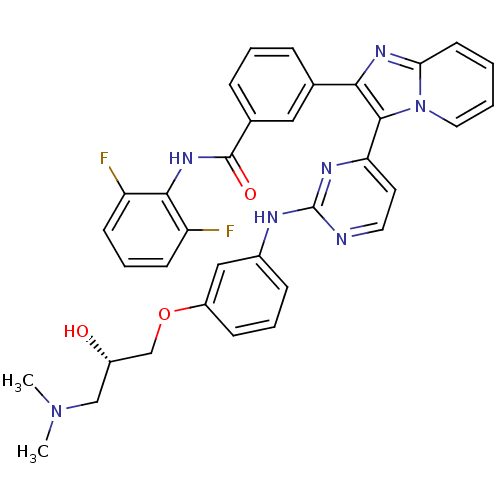
((S)-N-(2,6-difluorophenyl)-3-(3-(2-(3-(3-(dimethyl...)Show SMILES CN(C)C[C@H](O)COc1cccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c1 |r| Show InChI InChI=1S/C35H31F2N7O3/c1-43(2)20-25(45)21-47-26-11-6-10-24(19-26)39-35-38-16-15-29(40-35)33-31(41-30-14-3-4-17-44(30)33)22-8-5-9-23(18-22)34(46)42-32-27(36)12-7-13-28(32)37/h3-19,25,45H,20-21H2,1-2H3,(H,42,46)(H,38,39,40)/t25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256473
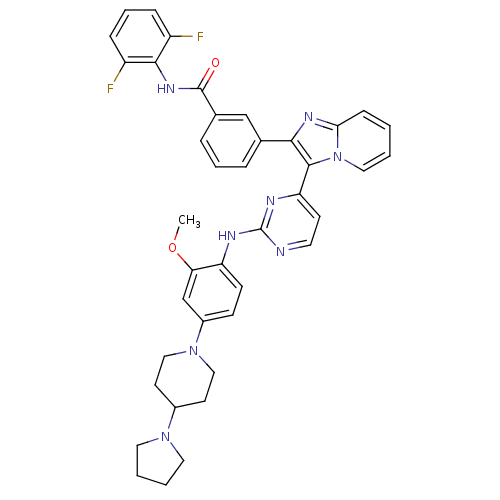
(CHEMBL502198 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCCC1 Show InChI InChI=1S/C40H38F2N8O2/c1-52-34-25-29(49-22-16-28(17-23-49)48-19-4-5-20-48)13-14-32(34)44-40-43-18-15-33(45-40)38-36(46-35-12-2-3-21-50(35)38)26-8-6-9-27(24-26)39(51)47-37-30(41)10-7-11-31(37)42/h2-3,6-15,18,21,24-25,28H,4-5,16-17,19-20,22-23H2,1H3,(H,47,51)(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256475
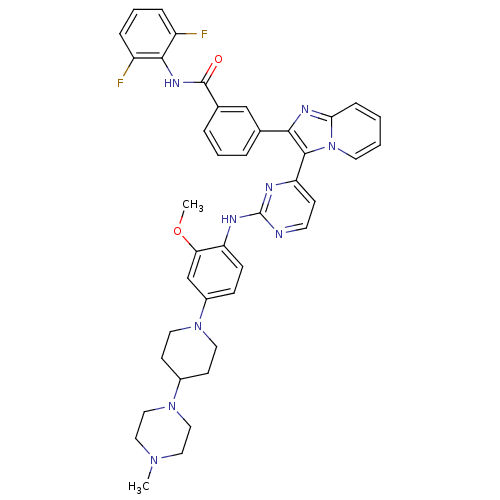
(CHEMBL449110 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(C)CC1 Show InChI InChI=1S/C41H41F2N9O2/c1-49-21-23-51(24-22-49)29-15-19-50(20-16-29)30-12-13-33(35(26-30)54-2)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256471
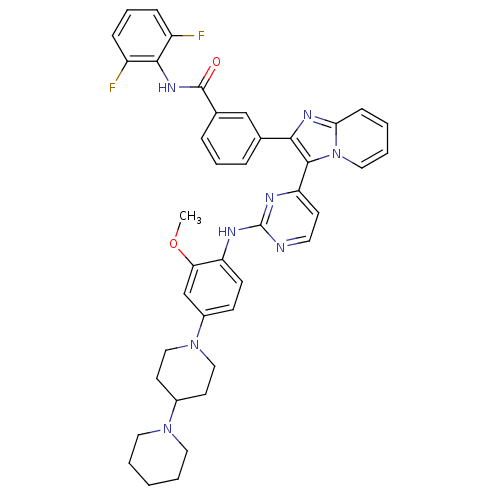
(3-(3-(2-(4-(1,4'-bipiperidin-1'-yl)-2-methoxypheny...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCCCC1 Show InChI InChI=1S/C41H40F2N8O2/c1-53-35-26-30(50-23-17-29(18-24-50)49-20-4-2-5-21-49)14-15-33(35)45-41-44-19-16-34(46-41)39-37(47-36-13-3-6-22-51(36)39)27-9-7-10-28(25-27)40(52)48-38-31(42)11-8-12-32(38)43/h3,6-16,19,22,25-26,29H,2,4-5,17-18,20-21,23-24H2,1H3,(H,48,52)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256474
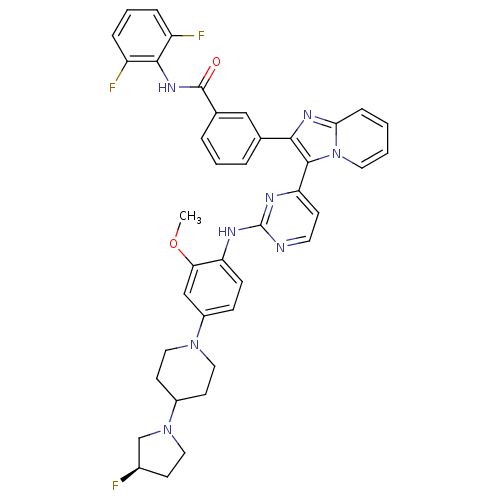
((R)-N-(2,6-difluorophenyl)-3-(3-(2-(4-(4-(3-fluoro...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CC[C@@H](F)C1 |r| Show InChI InChI=1S/C40H37F3N8O2/c1-53-34-23-29(49-20-15-28(16-21-49)50-19-14-27(41)24-50)11-12-32(34)45-40-44-17-13-33(46-40)38-36(47-35-10-2-3-18-51(35)38)25-6-4-7-26(22-25)39(52)48-37-30(42)8-5-9-31(37)43/h2-13,17-18,22-23,27-28H,14-16,19-21,24H2,1H3,(H,48,52)(H,44,45,46)/t27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256472
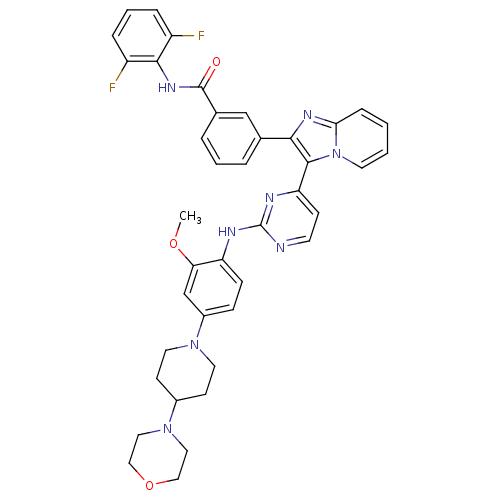
(CHEMBL502652 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCOCC1 Show InChI InChI=1S/C40H38F2N8O3/c1-52-34-25-29(48-18-14-28(15-19-48)49-20-22-53-23-21-49)11-12-32(34)44-40-43-16-13-33(45-40)38-36(46-35-10-2-3-17-50(35)38)26-6-4-7-27(24-26)39(51)47-37-30(41)8-5-9-31(37)42/h2-13,16-17,24-25,28H,14-15,18-23H2,1H3,(H,47,51)(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256410
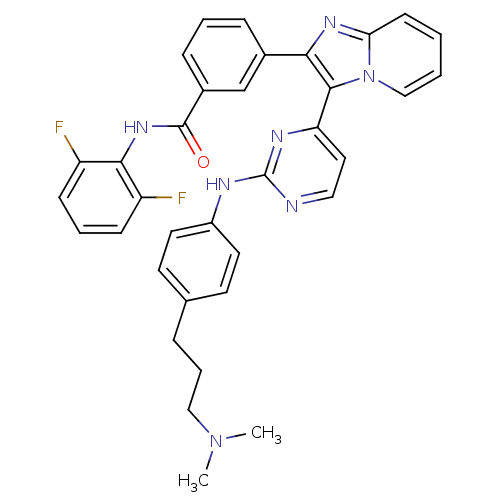
(CHEMBL447668 | N-(2,6-difluorophenyl)-3-(3-(2-(3-(...)Show SMILES CN(C)CCCc1ccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)cc1 Show InChI InChI=1S/C35H31F2N7O/c1-43(2)20-7-8-23-14-16-26(17-15-23)39-35-38-19-18-29(40-35)33-31(41-30-13-3-4-21-44(30)33)24-9-5-10-25(22-24)34(45)42-32-27(36)11-6-12-28(32)37/h3-6,9-19,21-22H,7-8,20H2,1-2H3,(H,42,45)(H,38,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256470
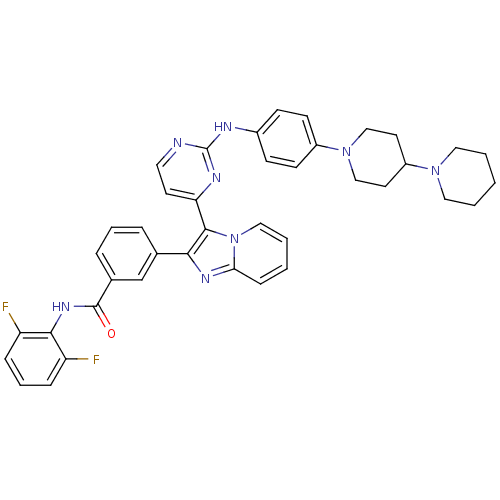
(3-(3-(2-(4-(1,4'-bipiperidin-1'-yl)phenylamino)pyr...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc(cc2)N2CCC(CC2)N2CCCCC2)n1 Show InChI InChI=1S/C40H38F2N8O/c41-32-10-7-11-33(42)37(32)47-39(51)28-9-6-8-27(26-28)36-38(50-23-5-2-12-35(50)46-36)34-17-20-43-40(45-34)44-29-13-15-30(16-14-29)49-24-18-31(19-25-49)48-21-3-1-4-22-48/h2,5-17,20,23,26,31H,1,3-4,18-19,21-22,24-25H2,(H,47,51)(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256477
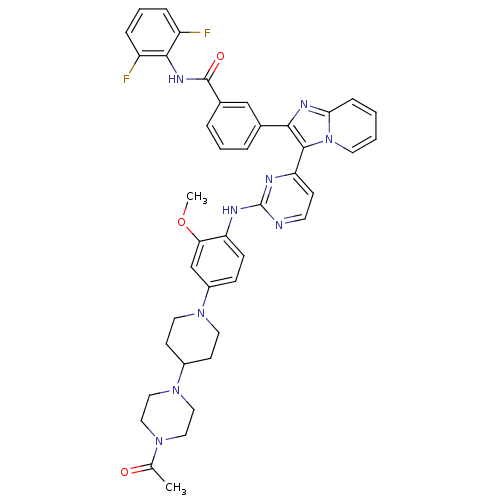
(3-(3-(2-(4-(4-(4-acetylpiperazin-1-yl)piperidin-1-...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)C(C)=O Show InChI InChI=1S/C42H41F2N9O3/c1-27(54)50-21-23-52(24-22-50)30-15-19-51(20-16-30)31-12-13-34(36(26-31)56-2)46-42-45-17-14-35(47-42)40-38(48-37-11-3-4-18-53(37)40)28-7-5-8-29(25-28)41(55)49-39-32(43)9-6-10-33(39)44/h3-14,17-18,25-26,30H,15-16,19-24H2,1-2H3,(H,49,55)(H,45,46,47) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256476
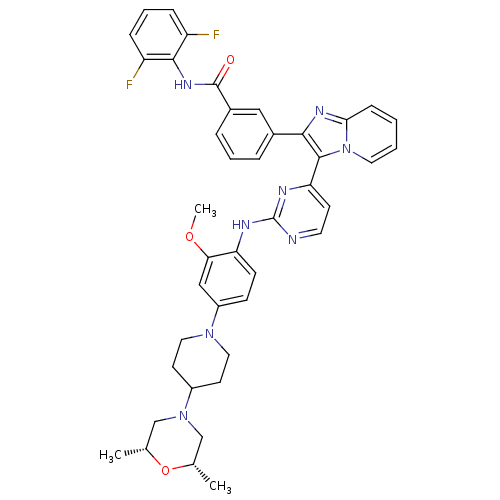
(CHEMBL454796 | N-(2,6-difluorophenyl)-3-(3-(2-(4-(...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1C[C@H](C)O[C@H](C)C1 |r| Show InChI InChI=1S/C42H42F2N8O3/c1-26-24-51(25-27(2)55-26)30-16-20-50(21-17-30)31-13-14-34(36(23-31)54-3)46-42-45-18-15-35(47-42)40-38(48-37-12-4-5-19-52(37)40)28-8-6-9-29(22-28)41(53)49-39-32(43)10-7-11-33(39)44/h4-15,18-19,22-23,26-27,30H,16-17,20-21,24-25H2,1-3H3,(H,49,53)(H,45,46,47)/t26-,27+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256468

(CHEMBL500003 | N-(2,6-difluorophenyl)-3-(3-(2-(4-(...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc(CCN3CCOCC3)cc2)n1 Show InChI InChI=1S/C36H31F2N7O2/c37-28-7-4-8-29(38)33(28)43-35(46)26-6-3-5-25(23-26)32-34(45-17-2-1-9-31(45)42-32)30-14-16-39-36(41-30)40-27-12-10-24(11-13-27)15-18-44-19-21-47-22-20-44/h1-14,16-17,23H,15,18-22H2,(H,43,46)(H,39,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged insulin receptor expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256466
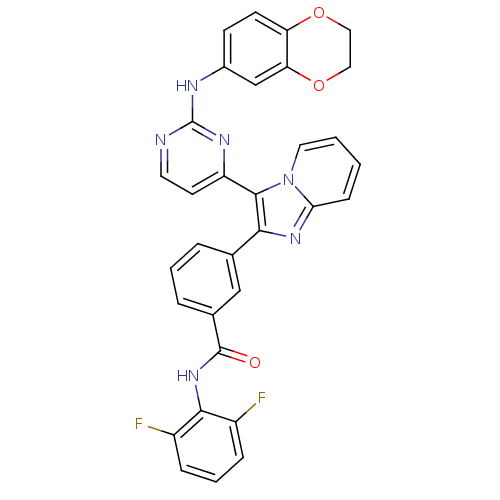
(CHEMBL444031 | N-(2,6-difluorophenyl)-3-(3-(2-(2,3...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc3OCCOc3c2)n1 Show InChI InChI=1S/C32H22F2N6O3/c33-22-7-4-8-23(34)29(22)39-31(41)20-6-3-5-19(17-20)28-30(40-14-2-1-9-27(40)38-28)24-12-13-35-32(37-24)36-21-10-11-25-26(18-21)43-16-15-42-25/h1-14,17-18H,15-16H2,(H,39,41)(H,35,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50256479
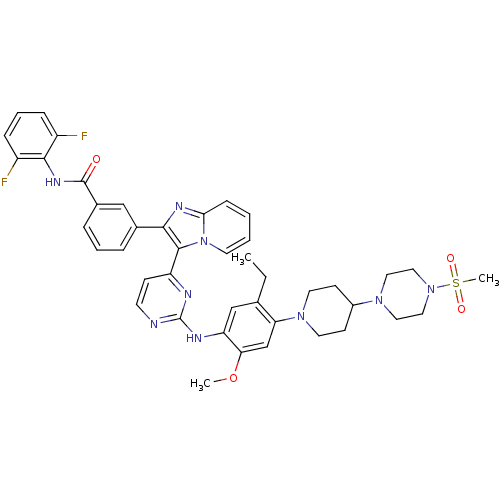
(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged insulin receptor expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50256470

(3-(3-(2-(4-(1,4'-bipiperidin-1'-yl)phenylamino)pyr...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc(cc2)N2CCC(CC2)N2CCCCC2)n1 Show InChI InChI=1S/C40H38F2N8O/c41-32-10-7-11-33(42)37(32)47-39(51)28-9-6-8-27(26-28)36-38(50-23-5-2-12-35(50)46-36)34-17-20-43-40(45-34)44-29-13-15-30(16-14-29)49-24-18-31(19-25-49)48-21-3-1-4-22-48/h2,5-17,20,23,26,31H,1,3-4,18-19,21-22,24-25H2,(H,47,51)(H,43,44,45) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256412
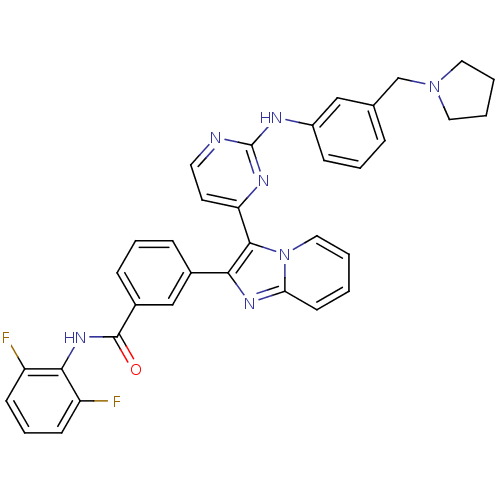
(CHEMBL447450 | N-(2,6-difluorophenyl)-3-(3-(2-(3-(...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2cccc(CN3CCCC3)c2)n1 Show InChI InChI=1S/C35H29F2N7O/c36-27-12-7-13-28(37)32(27)42-34(45)25-10-6-9-24(21-25)31-33(44-19-2-1-14-30(44)41-31)29-15-16-38-35(40-29)39-26-11-5-8-23(20-26)22-43-17-3-4-18-43/h1-2,5-16,19-21H,3-4,17-18,22H2,(H,42,45)(H,38,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human insulin receptor autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged insulin receptor expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256465
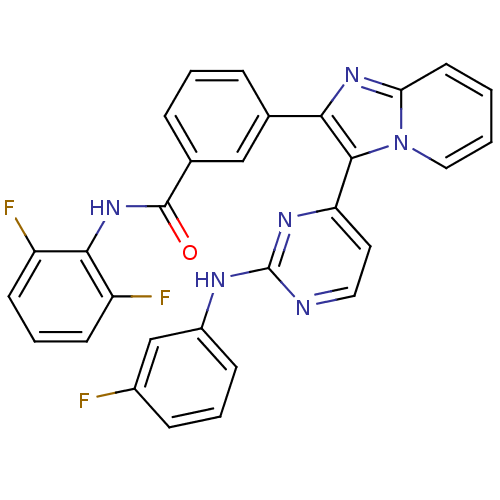
(CHEMBL480544 | N-(2,6-difluorophenyl)-3-(3-(2-(3-f...)Show SMILES Fc1cccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c1 Show InChI InChI=1S/C30H19F3N6O/c31-20-8-4-9-21(17-20)35-30-34-14-13-24(36-30)28-26(37-25-12-1-2-15-39(25)28)18-6-3-7-19(16-18)29(40)38-27-22(32)10-5-11-23(27)33/h1-17H,(H,38,40)(H,34,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of GST-tagged IGF1R (957-1367) (unknown origin) expressed in baculovirus by time-resolved fluorescence assay |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256475

(CHEMBL449110 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(C)CC1 Show InChI InChI=1S/C41H41F2N9O2/c1-49-21-23-51(24-22-49)29-15-19-50(20-16-29)30-12-13-33(35(26-30)54-2)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256473

(CHEMBL502198 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCCC1 Show InChI InChI=1S/C40H38F2N8O2/c1-52-34-25-29(49-22-16-28(17-23-49)48-19-4-5-20-48)13-14-32(34)44-40-43-18-15-33(45-40)38-36(46-35-12-2-3-21-50(35)38)26-8-6-9-27(24-26)39(51)47-37-30(41)10-7-11-31(37)42/h2-3,6-15,18,21,24-25,28H,4-5,16-17,19-20,22-23H2,1H3,(H,47,51)(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256471

(3-(3-(2-(4-(1,4'-bipiperidin-1'-yl)-2-methoxypheny...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCCCC1 Show InChI InChI=1S/C41H40F2N8O2/c1-53-35-26-30(50-23-17-29(18-24-50)49-20-4-2-5-21-49)14-15-33(35)45-41-44-19-16-34(46-41)39-37(47-36-13-3-6-22-51(36)39)27-9-7-10-28(25-27)40(52)48-38-31(42)11-8-12-32(38)43/h3,6-16,19,22,25-26,29H,2,4-5,17-18,20-21,23-24H2,1H3,(H,48,52)(H,44,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256474

((R)-N-(2,6-difluorophenyl)-3-(3-(2-(4-(4-(3-fluoro...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CC[C@@H](F)C1 |r| Show InChI InChI=1S/C40H37F3N8O2/c1-53-34-23-29(49-20-15-28(16-21-49)50-19-14-27(41)24-50)11-12-32(34)45-40-44-17-13-33(46-40)38-36(47-35-10-2-3-18-51(35)38)25-6-4-7-26(22-25)39(52)48-37-30(42)8-5-9-31(37)43/h2-13,17-18,22-23,27-28H,14-16,19-21,24H2,1H3,(H,48,52)(H,44,45,46)/t27-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256472

(CHEMBL502652 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCOCC1 Show InChI InChI=1S/C40H38F2N8O3/c1-52-34-25-29(48-18-14-28(15-19-48)49-20-22-53-23-21-49)11-12-32(34)44-40-43-16-13-33(45-40)38-36(46-35-10-2-3-17-50(35)38)26-6-4-7-27(24-26)39(51)47-37-30(41)8-5-9-31(37)42/h2-13,16-17,24-25,28H,14-15,18-23H2,1H3,(H,47,51)(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-2
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ErbB2 |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 57 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256477

(3-(3-(2-(4-(4-(4-acetylpiperazin-1-yl)piperidin-1-...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)C(C)=O Show InChI InChI=1S/C42H41F2N9O3/c1-27(54)50-21-23-52(24-22-50)30-15-19-51(20-16-30)31-12-13-34(36(26-31)56-2)46-42-45-17-14-35(47-42)40-38(48-37-11-3-4-18-53(37)40)28-7-5-8-29(25-28)41(55)49-39-32(43)9-6-10-33(39)44/h3-14,17-18,25-26,30H,15-16,19-24H2,1-2H3,(H,49,55)(H,45,46,47) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256476

(CHEMBL454796 | N-(2,6-difluorophenyl)-3-(3-(2-(4-(...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1C[C@H](C)O[C@H](C)C1 |r| Show InChI InChI=1S/C42H42F2N8O3/c1-26-24-51(25-27(2)55-26)30-16-20-50(21-17-30)31-13-14-34(36(23-31)54-3)46-42-45-18-15-35(47-42)40-38(48-37-12-4-5-19-52(37)40)28-8-6-9-29(22-28)41(53)49-39-32(43)10-7-11-33(39)44/h4-15,18-19,22-23,26-27,30H,16-17,20-21,24-25H2,1-3H3,(H,49,53)(H,45,46,47)/t26-,27+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-2
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 72 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ErbB2 |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-4
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ErbB4 |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256470

(3-(3-(2-(4-(1,4'-bipiperidin-1'-yl)phenylamino)pyr...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc(cc2)N2CCC(CC2)N2CCCCC2)n1 Show InChI InChI=1S/C40H38F2N8O/c41-32-10-7-11-33(42)37(32)47-39(51)28-9-6-8-27(26-28)36-38(50-23-5-2-12-35(50)46-36)34-17-20-43-40(45-34)44-29-13-15-30(16-14-29)49-24-18-31(19-25-49)48-21-3-1-4-22-48/h2,5-17,20,23,26,31H,1,3-4,18-19,21-22,24-25H2,(H,47,51)(H,43,44,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 87 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50256471

(3-(3-(2-(4-(1,4'-bipiperidin-1'-yl)-2-methoxypheny...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCCCC1 Show InChI InChI=1S/C41H40F2N8O2/c1-53-35-26-30(50-23-17-29(18-24-50)49-20-4-2-5-21-49)14-15-33(35)45-41-44-19-16-34(46-41)39-37(47-36-13-3-6-22-51(36)39)27-9-7-10-28(25-27)40(52)48-38-31(42)11-8-12-32(38)43/h3,6-16,19,22,25-26,29H,2,4-5,17-18,20-21,23-24H2,1H3,(H,48,52)(H,44,45,46) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-4
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ErbB4 |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256468

(CHEMBL500003 | N-(2,6-difluorophenyl)-3-(3-(2-(4-(...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc(CCN3CCOCC3)cc2)n1 Show InChI InChI=1S/C36H31F2N7O2/c37-28-7-4-8-29(38)33(28)43-35(46)26-6-3-5-25(23-26)32-34(45-17-2-1-9-31(45)42-32)30-14-16-39-36(41-30)40-27-12-10-24(11-13-27)15-18-44-19-21-47-22-20-44/h1-14,16-17,23H,15,18-22H2,(H,43,46)(H,39,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 174 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256467

(CHEMBL448668 | N-(2,6-difluorophenyl)-3-(3-(2-(3-(...)Show SMILES CN(C)CCOc1cccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c1 Show InChI InChI=1S/C34H29F2N7O2/c1-42(2)18-19-45-25-11-6-10-24(21-25)38-34-37-16-15-28(39-34)32-30(40-29-14-3-4-17-43(29)32)22-8-5-9-23(20-22)33(44)41-31-26(35)12-7-13-27(31)36/h3-17,20-21H,18-19H2,1-2H3,(H,41,44)(H,37,38,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 177 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Aurora B |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256469

((S)-N-(2,6-difluorophenyl)-3-(3-(2-(3-(3-(dimethyl...)Show SMILES CN(C)C[C@H](O)COc1cccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c1 |r| Show InChI InChI=1S/C35H31F2N7O3/c1-43(2)20-25(45)21-47-26-11-6-10-24(19-26)39-35-38-16-15-29(40-35)33-31(41-30-14-3-4-17-44(30)33)22-8-5-9-23(18-22)34(46)42-32-27(36)12-7-13-28(32)37/h3-19,25,45H,20-21H2,1-2H3,(H,42,46)(H,38,39,40)/t25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 244 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256412

(CHEMBL447450 | N-(2,6-difluorophenyl)-3-(3-(2-(3-(...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2cccc(CN3CCCC3)c2)n1 Show InChI InChI=1S/C35H29F2N7O/c36-27-12-7-13-28(37)32(27)42-34(45)25-10-6-9-24(21-25)31-33(44-19-2-1-14-30(44)41-31)29-15-16-38-35(40-29)39-26-11-5-8-23(20-26)22-43-17-3-4-18-43/h1-2,5-16,19-21H,3-4,17-18,22H2,(H,42,45)(H,38,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 247 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256410

(CHEMBL447668 | N-(2,6-difluorophenyl)-3-(3-(2-(3-(...)Show SMILES CN(C)CCCc1ccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)cc1 Show InChI InChI=1S/C35H31F2N7O/c1-43(2)20-7-8-23-14-16-26(17-15-23)39-35-38-19-18-29(40-35)33-31(41-30-13-3-4-21-44(30)33)24-9-5-10-25(22-24)34(45)42-32-27(36)11-6-12-28(32)37/h3-6,9-19,21-22H,7-8,20H2,1-2H3,(H,42,45)(H,38,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 344 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256466

(CHEMBL444031 | N-(2,6-difluorophenyl)-3-(3-(2-(2,3...)Show SMILES Fc1cccc(F)c1NC(=O)c1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2ccc3OCCOc3c2)n1 Show InChI InChI=1S/C32H22F2N6O3/c33-22-7-4-8-23(34)29(22)39-31(41)20-6-3-5-19(17-20)28-30(40-14-2-1-9-27(40)38-28)24-12-13-35-32(37-24)36-21-10-11-25-26(18-21)43-16-15-42-25/h1-14,17-18H,15-16H2,(H,39,41)(H,35,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 431 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256413
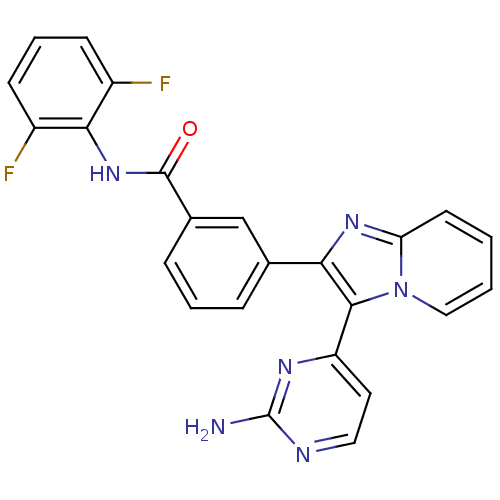
(3-(3-(2-aminopyrimidin-4-yl)imidazo[1,2-a]pyridin-...)Show SMILES Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F Show InChI InChI=1S/C24H16F2N6O/c25-16-7-4-8-17(26)21(16)31-23(33)15-6-3-5-14(13-15)20-22(18-10-11-28-24(27)29-18)32-12-2-1-9-19(32)30-20/h1-13H,(H,31,33)(H2,27,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256465

(CHEMBL480544 | N-(2,6-difluorophenyl)-3-(3-(2-(3-f...)Show SMILES Fc1cccc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c1 Show InChI InChI=1S/C30H19F3N6O/c31-20-8-4-9-21(17-20)35-30-34-14-13-24(36-30)28-26(37-25-12-1-2-15-39(25)28)18-6-3-7-19(16-18)29(40)38-27-22(32)10-5-11-23(27)33/h1-17H,(H,38,40)(H,34,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 509 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 710 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Aurora B |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50256478

(CHEMBL507714 | N-(2,6-difluorophenyl)-3-(3-(2-(2-m...)Show SMILES COc1cc(ccc1Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F)N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C41H41F2N9O4S/c1-56-35-26-30(49-19-15-29(16-20-49)50-21-23-51(24-22-50)57(2,54)55)12-13-33(35)45-41-44-17-14-34(46-41)39-37(47-36-11-3-4-18-52(36)39)27-7-5-8-28(25-27)40(53)48-38-31(42)9-6-10-32(38)43/h3-14,17-18,25-26,29H,15-16,19-24H2,1-2H3,(H,48,53)(H,44,45,46) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Aurora B |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256411
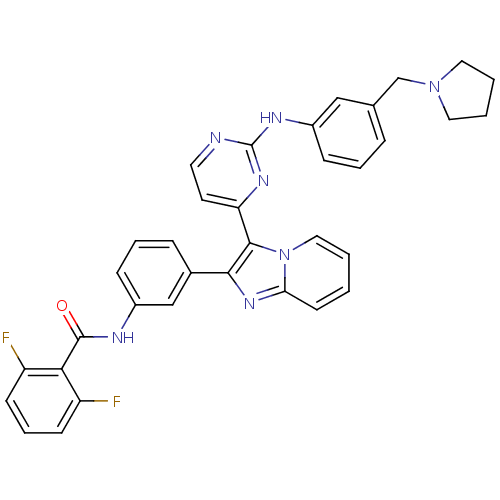
(2,6-difluoro-N-(3-(3-(2-(3-(pyrrolidin-1-ylmethyl)...)Show SMILES Fc1cccc(F)c1C(=O)Nc1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2cccc(CN3CCCC3)c2)n1 Show InChI InChI=1S/C35H29F2N7O/c36-27-12-7-13-28(37)31(27)34(45)39-26-11-6-9-24(21-26)32-33(44-19-2-1-14-30(44)42-32)29-15-16-38-35(41-29)40-25-10-5-8-23(20-25)22-43-17-3-4-18-43/h1-2,5-16,19-21H,3-4,17-18,22H2,(H,39,45)(H,38,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.93E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256411

(2,6-difluoro-N-(3-(3-(2-(3-(pyrrolidin-1-ylmethyl)...)Show SMILES Fc1cccc(F)c1C(=O)Nc1cccc(c1)-c1nc2ccccn2c1-c1ccnc(Nc2cccc(CN3CCCC3)c2)n1 Show InChI InChI=1S/C35H29F2N7O/c36-27-12-7-13-28(37)31(27)34(45)39-26-11-6-9-24(21-26)32-33(44-19-2-1-14-30(44)42-32)29-15-16-38-35(41-29)40-25-10-5-8-23(20-25)22-43-17-3-4-18-43/h1-2,5-16,19-21H,3-4,17-18,22H2,(H,39,45)(H,38,40,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.93E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50256479

(CHEMBL448929 | N-(2,6-difluorophenyl)-3-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2cccc(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C43H45F2N9O4S/c1-4-28-26-35(37(58-2)27-36(28)52-19-15-31(16-20-52)51-21-23-53(24-22-51)59(3,56)57)48-43-46-17-14-34(47-43)41-39(49-38-13-5-6-18-54(38)41)29-9-7-10-30(25-29)42(55)50-40-32(44)11-8-12-33(40)45/h5-14,17-18,25-27,31H,4,15-16,19-24H2,1-3H3,(H,50,55)(H,46,47,48) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >9.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Aurora A |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of EGFR |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-2
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ErbB2 |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-4
(Homo sapiens (Human)) | BDBM50256480

(CHEMBL466397 | N-(2,6-difluorophenyl)-5-(3-(2-(5-e...)Show SMILES CCc1cc(Nc2nccc(n2)-c2c(nc3ccccn23)-c2ccc(OC)c(c2)C(=O)Nc2c(F)cccc2F)c(OC)cc1N1CCC(CC1)N1CCN(CC1)S(C)(=O)=O Show InChI InChI=1S/C44H47F2N9O5S/c1-5-28-26-35(38(60-3)27-36(28)53-19-15-30(16-20-53)52-21-23-54(24-22-52)61(4,57)58)49-44-47-17-14-34(48-44)42-40(50-39-11-6-7-18-55(39)42)29-12-13-37(59-2)31(25-29)43(56)51-41-32(45)9-8-10-33(41)46/h6-14,17-18,25-27,30H,5,15-16,19-24H2,1-4H3,(H,51,56)(H,47,48,49) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ErbB4 |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50256413

(3-(3-(2-aminopyrimidin-4-yl)imidazo[1,2-a]pyridin-...)Show SMILES Nc1nccc(n1)-c1c(nc2ccccn12)-c1cccc(c1)C(=O)Nc1c(F)cccc1F Show InChI InChI=1S/C24H16F2N6O/c25-16-7-4-8-17(26)21(16)31-23(33)15-6-3-5-14(13-15)20-22(18-10-11-28-24(27)29-18)32-12-2-1-9-19(32)30-20/h1-13H,(H,31,33)(H2,27,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human IGF1R autophosphorylation in transfected mouse NIH3T3 cells |
Bioorg Med Chem Lett 19: 1004-8 (2009)
Article DOI: 10.1016/j.bmcl.2008.11.058
BindingDB Entry DOI: 10.7270/Q24T6J8M |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data