

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50300129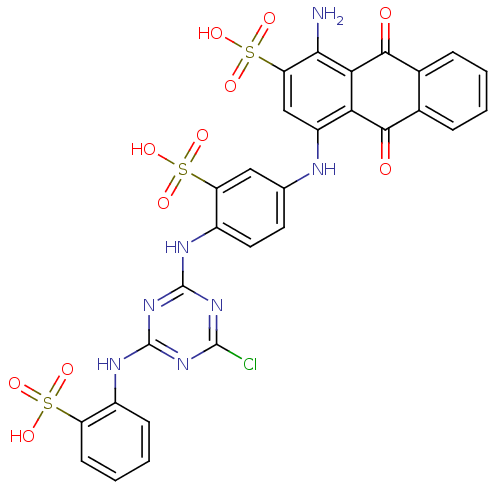 (CHEMBL572528 | CIBACRON BLUE | Cibacron Blue 3Ga) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50300128 (1-(3-(1H-tetrazol-5-yl)propyl)-4-(benzhydryloxy)pi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents | MMDB PDB Article PubMed | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50300127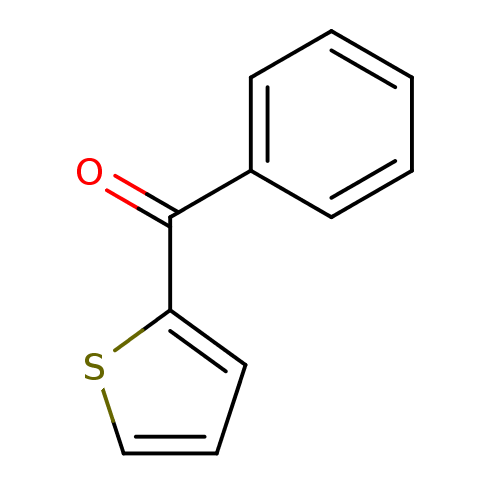 (CHEMBL567059 | NSC-4502 | phenyl(thiophen-2-yl)met...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 9.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50300126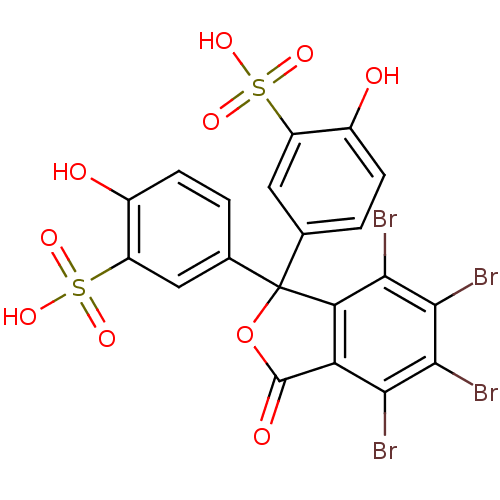 (CHEMBL574431 | Sulfobromophthalein) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50300125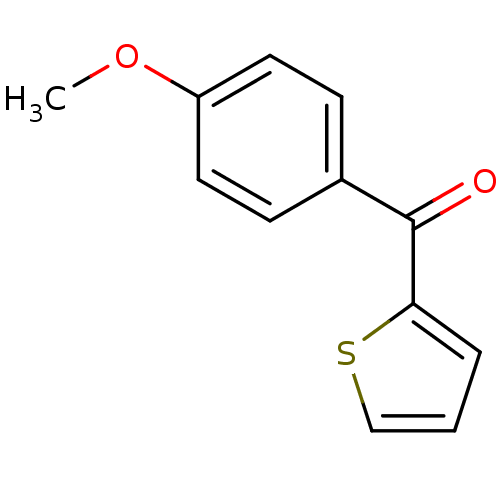 ((4-methoxyphenyl)(thiophen-2-yl)methanone | CHEMBL...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.05E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50186231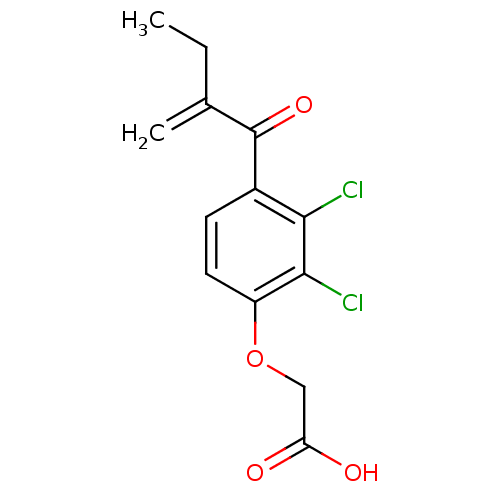 ((2,3-Dichloro-4-(2-methylene-1-oxobutyl)phenoxy)ac...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.22E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic prostaglandin D synthase (Homo sapiens (Human)) | BDBM50300124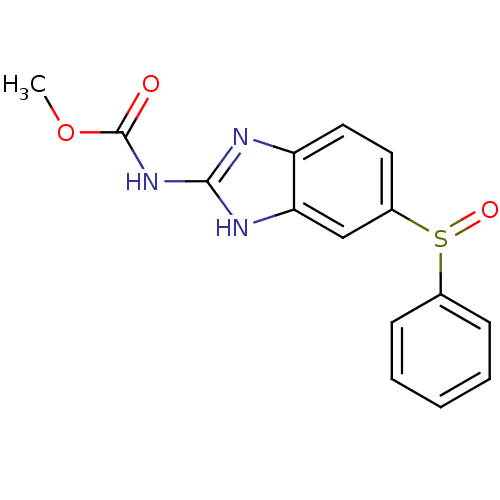 ((5-(phenylsulfinyl)-1H-benzimidazol-2-yl)carbamic ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland Curated by ChEMBL | Assay Description Inhibition of human H-PGDS expressed in Escherichia coli BL21 assessed as rate of glutathione-chloro-dinitro benzene conjugation | Eur J Med Chem 45: 447-54 (2010) Article DOI: 10.1016/j.ejmech.2009.10.025 BindingDB Entry DOI: 10.7270/Q2HD7VQT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||