 Found 47 hits of Enzyme Inhibition Constant Data
Found 47 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331795
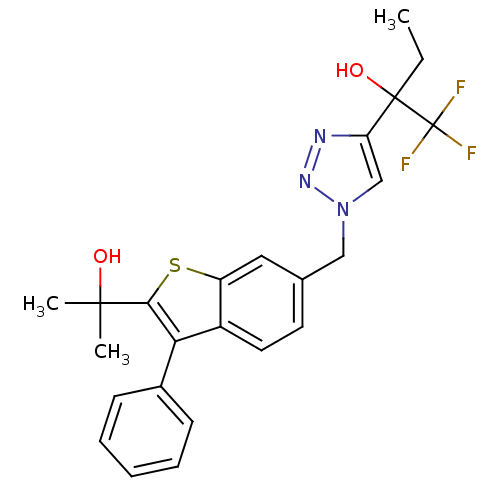
(1,1,1-trifluoro-2-(1-((2-(2-hydroxypropan-2-yl)-3-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)(C)O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C24H24F3N3O2S/c1-4-23(32,24(25,26)27)19-14-30(29-28-19)13-15-10-11-17-18(12-15)33-21(22(2,3)31)20(17)16-8-6-5-7-9-16/h5-12,14,31-32H,4,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331796

(1,1,1-trifluoro-2-(1-((2-(1-hydroxyethyl)-3-phenyl...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H22F3N3O2S/c1-3-22(31,23(24,25)26)19-13-29(28-27-19)12-15-9-10-17-18(11-15)32-21(14(2)30)20(17)16-7-5-4-6-8-16/h4-11,13-14,30-31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331797
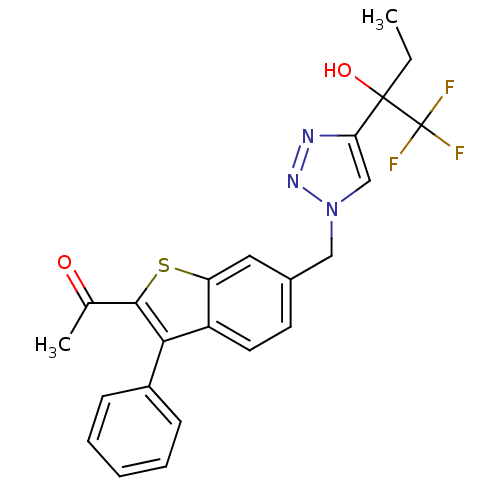
(1-(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H20F3N3O2S/c1-3-22(31,23(24,25)26)19-13-29(28-27-19)12-15-9-10-17-18(11-15)32-21(14(2)30)20(17)16-7-5-4-6-8-16/h4-11,13,31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331806
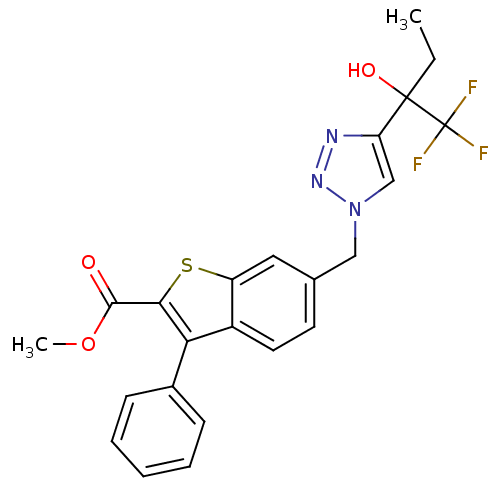
(CHEMBL1290649 | methyl 3-phenyl-6-((4-(1,1,1-trifl...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)OC)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H20F3N3O3S/c1-3-22(31,23(24,25)26)18-13-29(28-27-18)12-14-9-10-16-17(11-14)33-20(21(30)32-2)19(16)15-7-5-4-6-8-15/h4-11,13,31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331804

((R)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331803
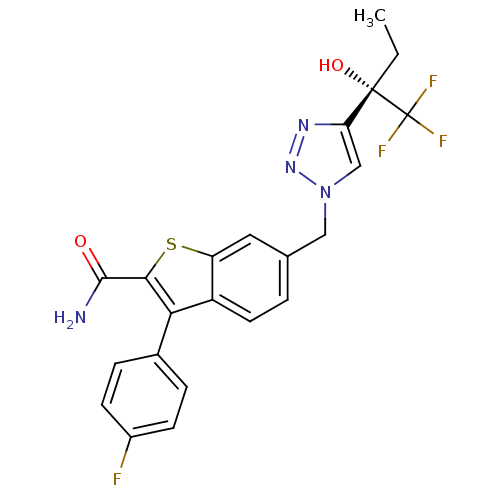
((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331794
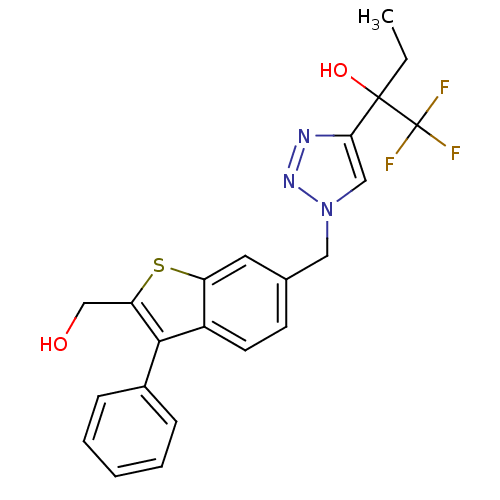
(1,1,1-trifluoro-2-(1-((2-(hydroxymethyl)-3-phenylb...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(CO)sc3c2)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H20F3N3O2S/c1-2-21(30,22(23,24)25)19-12-28(27-26-19)11-14-8-9-16-17(10-14)31-18(13-29)20(16)15-6-4-3-5-7-15/h3-10,12,29-30H,2,11,13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331802

((S)-3-(3-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2cccc(F)c2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-6-7-15-16(8-12)33-19(20(27)31)18(15)13-4-3-5-14(23)9-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331801

((S)-3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccccc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H19F3N4O2S/c1-2-21(31,22(23,24)25)17-12-29(28-27-17)11-13-8-9-15-16(10-13)32-19(20(26)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H2,26,30)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331800
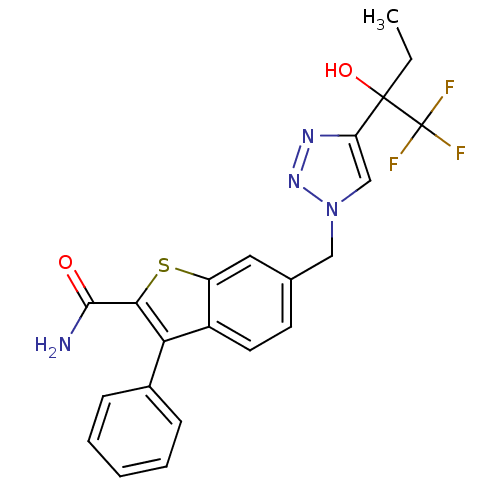
(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-2-y...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H19F3N4O2S/c1-2-21(31,22(23,24)25)17-12-29(28-27-17)11-13-8-9-15-16(10-13)32-19(20(26)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H2,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331798
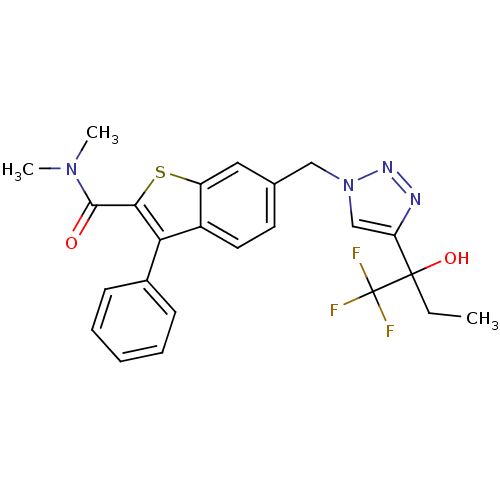
(CHEMBL1289328 | N,N-dimethyl-3-phenyl-6-((4-(1,1,1...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)N(C)C)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C24H23F3N4O2S/c1-4-23(33,24(25,26)27)19-14-31(29-28-19)13-15-10-11-17-18(12-15)34-21(22(32)30(2)3)20(17)16-8-6-5-7-9-16/h5-12,14,33H,4,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331793

(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-2-y...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(O)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H18F3N3O3S/c1-2-21(31,22(23,24)25)17-12-28(27-26-17)11-13-8-9-15-16(10-13)32-19(20(29)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331805

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(O)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H17F4N3O3S/c1-2-21(32,22(24,25)26)17-11-29(28-27-17)10-12-3-8-15-16(9-12)33-19(20(30)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H,30,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50331799

(CHEMBL1289329 | N-methyl-3-phenyl-6-((4-(1,1,1-tri...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)NC)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H21F3N4O2S/c1-3-22(32,23(24,25)26)18-13-30(29-28-18)12-14-9-10-16-17(11-14)33-20(21(31)27-2)19(16)15-7-5-4-6-8-15/h4-11,13,32H,3,12H2,1-2H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Displacement of labeled MK-499 from human ERG |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331806

(CHEMBL1290649 | methyl 3-phenyl-6-((4-(1,1,1-trifl...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)OC)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H20F3N3O3S/c1-3-22(31,23(24,25)26)18-13-29(28-27-18)12-14-9-10-16-17(11-14)33-20(21(30)32-2)19(16)15-7-5-4-6-8-15/h4-11,13,31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331797

(1-(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H20F3N3O2S/c1-3-22(31,23(24,25)26)19-13-29(28-27-19)12-15-9-10-17-18(11-15)32-21(14(2)30)20(17)16-7-5-4-6-8-16/h4-11,13,31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331794

(1,1,1-trifluoro-2-(1-((2-(hydroxymethyl)-3-phenylb...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(CO)sc3c2)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H20F3N3O2S/c1-2-21(30,22(23,24)25)19-12-28(27-26-19)11-14-8-9-16-17(10-14)31-18(13-29)20(16)15-6-4-3-5-7-15/h3-10,12,29-30H,2,11,13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331803

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331801

((S)-3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccccc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H19F3N4O2S/c1-2-21(31,22(23,24)25)17-12-29(28-27-17)11-13-8-9-15-16(10-13)32-19(20(26)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H2,26,30)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331802

((S)-3-(3-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2cccc(F)c2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-6-7-15-16(8-12)33-19(20(27)31)18(15)13-4-3-5-14(23)9-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331801

((S)-3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccccc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H19F3N4O2S/c1-2-21(31,22(23,24)25)17-12-29(28-27-17)11-13-8-9-15-16(10-13)32-19(20(26)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H2,26,30)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331802

((S)-3-(3-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2cccc(F)c2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-6-7-15-16(8-12)33-19(20(27)31)18(15)13-4-3-5-14(23)9-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331807

(1,1,1-trifluoro-2-(1-((3-phenylbenzo[b]thiophen-6-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(csc3c2)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C21H18F3N3OS/c1-2-20(28,21(22,23)24)19-12-27(26-25-19)11-14-8-9-16-17(13-29-18(16)10-14)15-6-4-3-5-7-15/h3-10,12-13,28H,2,11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331800

(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-2-y...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H19F3N4O2S/c1-2-21(31,22(23,24)25)17-12-29(28-27-17)11-13-8-9-15-16(10-13)32-19(20(26)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H2,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331803

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331794

(1,1,1-trifluoro-2-(1-((2-(hydroxymethyl)-3-phenylb...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(CO)sc3c2)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H20F3N3O2S/c1-2-21(30,22(23,24)25)19-12-28(27-26-19)11-14-8-9-16-17(10-14)31-18(13-29)20(16)15-6-4-3-5-7-15/h3-10,12,29-30H,2,11,13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331800

(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-2-y...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H19F3N4O2S/c1-2-21(31,22(23,24)25)17-12-29(28-27-17)11-13-8-9-15-16(10-13)32-19(20(26)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H2,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331797

(1-(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H20F3N3O2S/c1-3-22(31,23(24,25)26)19-13-29(28-27-19)12-15-9-10-17-18(11-15)32-21(14(2)30)20(17)16-7-5-4-6-8-16/h4-11,13,31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 84 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331796

(1,1,1-trifluoro-2-(1-((2-(1-hydroxyethyl)-3-phenyl...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H22F3N3O2S/c1-3-22(31,23(24,25)26)19-13-29(28-27-19)12-15-9-10-17-18(11-15)32-21(14(2)30)20(17)16-7-5-4-6-8-16/h4-11,13-14,30-31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 88 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331804

((R)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 154 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331796

(1,1,1-trifluoro-2-(1-((2-(1-hydroxyethyl)-3-phenyl...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H22F3N3O2S/c1-3-22(31,23(24,25)26)19-13-29(28-27-19)12-15-9-10-17-18(11-15)32-21(14(2)30)20(17)16-7-5-4-6-8-16/h4-11,13-14,30-31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 161 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331795

(1,1,1-trifluoro-2-(1-((2-(2-hydroxypropan-2-yl)-3-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)(C)O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C24H24F3N3O2S/c1-4-23(32,24(25,26)27)19-14-30(29-28-19)13-15-10-11-17-18(12-15)33-21(22(2,3)31)20(17)16-8-6-5-7-9-16/h5-12,14,31-32H,4,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 198 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331799

(CHEMBL1289329 | N-methyl-3-phenyl-6-((4-(1,1,1-tri...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)NC)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H21F3N4O2S/c1-3-22(32,23(24,25)26)18-13-30(29-28-18)12-14-9-10-16-17(11-14)33-20(21(31)27-2)19(16)15-7-5-4-6-8-15/h4-11,13,32H,3,12H2,1-2H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 223 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331806

(CHEMBL1290649 | methyl 3-phenyl-6-((4-(1,1,1-trifl...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)OC)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H20F3N3O3S/c1-3-22(31,23(24,25)26)18-13-29(28-27-18)12-14-9-10-16-17(11-14)33-20(21(30)32-2)19(16)15-7-5-4-6-8-15/h4-11,13,31H,3,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331799

(CHEMBL1289329 | N-methyl-3-phenyl-6-((4-(1,1,1-tri...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)NC)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C23H21F3N4O2S/c1-3-22(32,23(24,25)26)18-13-30(29-28-18)12-14-9-10-16-17(11-14)33-20(21(31)27-2)19(16)15-7-5-4-6-8-15/h4-11,13,32H,3,12H2,1-2H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 263 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331795

(1,1,1-trifluoro-2-(1-((2-(2-hydroxypropan-2-yl)-3-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(C)(C)O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C24H24F3N3O2S/c1-4-23(32,24(25,26)27)19-14-30(29-28-19)13-15-10-11-17-18(12-15)33-21(22(2,3)31)20(17)16-8-6-5-7-9-16/h5-12,14,31-32H,4,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 392 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331807

(1,1,1-trifluoro-2-(1-((3-phenylbenzo[b]thiophen-6-...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(csc3c2)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C21H18F3N3OS/c1-2-20(28,21(22,23)24)19-12-27(26-25-19)11-14-8-9-16-17(13-29-18(16)10-14)15-6-4-3-5-7-15/h3-10,12-13,28H,2,11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 598 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331804

((R)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331798

(CHEMBL1289328 | N,N-dimethyl-3-phenyl-6-((4-(1,1,1...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)N(C)C)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C24H23F3N4O2S/c1-4-23(33,24(25,26)27)19-14-31(29-28-19)13-15-10-11-17-18(12-15)34-21(22(32)30(2)3)20(17)16-8-6-5-7-9-16/h5-12,14,33H,4,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331798

(CHEMBL1289328 | N,N-dimethyl-3-phenyl-6-((4-(1,1,1...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(=O)N(C)C)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C24H23F3N4O2S/c1-4-23(33,24(25,26)27)19-14-31(29-28-19)13-15-10-11-17-18(12-15)34-21(22(32)30(2)3)20(17)16-8-6-5-7-9-16/h5-12,14,33H,4,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.89E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331793

(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-2-y...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(O)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H18F3N3O3S/c1-2-21(31,22(23,24)25)17-12-28(27-26-17)11-13-8-9-15-16(10-13)32-19(20(29)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331805

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(O)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H17F4N3O3S/c1-2-21(32,22(24,25)26)17-11-29(28-27-17)10-12-3-8-15-16(9-12)33-19(20(30)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H,30,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant 5-lipoxygenase assessed as arachidonic acid oxidation |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Arachidonate 5-lipoxygenase-activating protein
(Homo sapiens (Human)) | BDBM50331803

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase activating protein |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid lipoxygenase ALOX12
(Homo sapiens (Human)) | BDBM50331803

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 12-lipoxygenase |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331805

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(O)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H17F4N3O3S/c1-2-21(32,22(24,25)26)17-11-29(28-27-17)10-12-3-8-15-16(9-12)33-19(20(30)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H,30,31)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid 5-lipoxygenase
(Homo sapiens (Human)) | BDBM50331793

(3-phenyl-6-((4-(1,1,1-trifluoro-2-hydroxybutan-2-y...)Show SMILES CCC(O)(c1cn(Cc2ccc3c(c(sc3c2)C(O)=O)-c2ccccc2)nn1)C(F)(F)F Show InChI InChI=1S/C22H18F3N3O3S/c1-2-21(31,22(23,24)25)17-12-28(27-26-17)11-13-8-9-15-16(10-13)32-19(20(29)30)18(15)14-6-4-3-5-7-14/h3-10,12,31H,2,11H2,1H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 5-lipoxygenase in human whole blood assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |
Polyunsaturated fatty acid lipoxygenase ALOX15
(Homo sapiens (Human)) | BDBM50331803

((S)-3-(4-fluorophenyl)-6-((4-(1,1,1-trifluoro-2-hy...)Show SMILES CC[C@](O)(c1cn(Cc2ccc3c(c(sc3c2)C(N)=O)-c2ccc(F)cc2)nn1)C(F)(F)F |r| Show InChI InChI=1S/C22H18F4N4O2S/c1-2-21(32,22(24,25)26)17-11-30(29-28-17)10-12-3-8-15-16(9-12)33-19(20(27)31)18(15)13-4-6-14(23)7-5-13/h3-9,11,32H,2,10H2,1H3,(H2,27,31)/t21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibition of 15-lipoxygenase |
Bioorg Med Chem Lett 20: 7440-3 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.024
BindingDB Entry DOI: 10.7270/Q2XG9RC8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data