

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50210557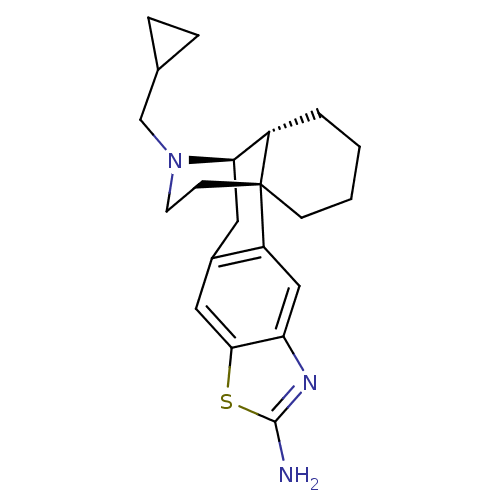 (CHEMBL242756 | MCL-147) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50135806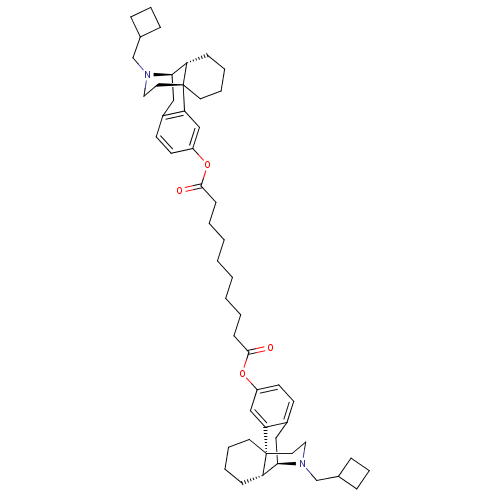 (CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343625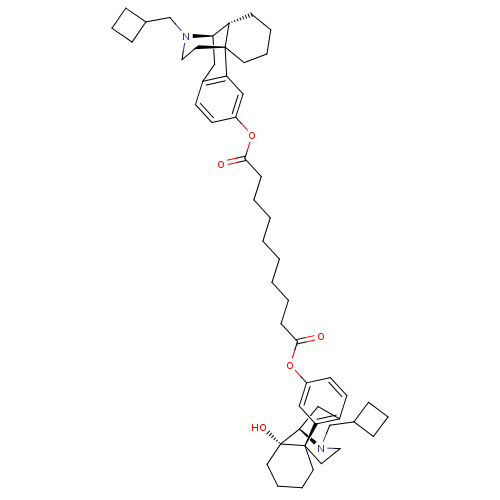 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50135808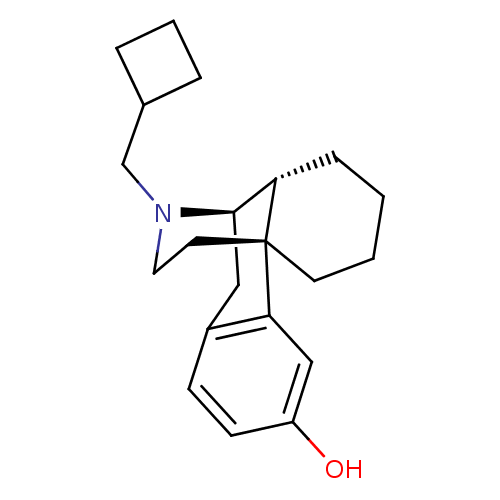 ((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50135797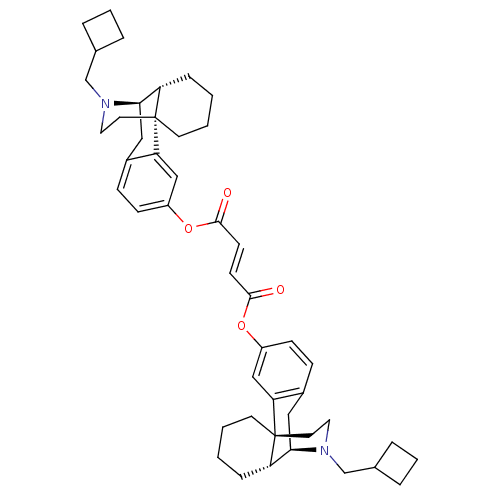 (CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343625 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0890 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50135806 (CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50240437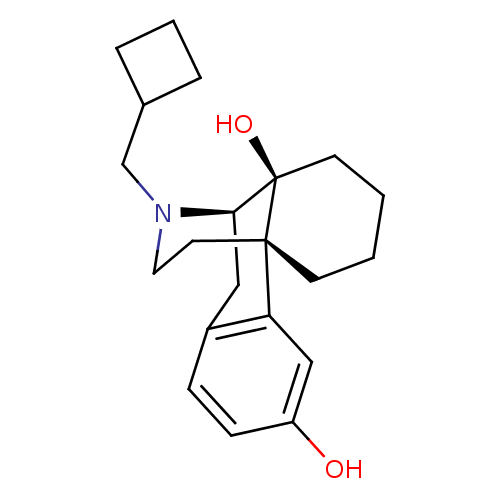 ((-)-17-(cyclobutylmethyl)morphinan-3,14-diol | (-)...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343626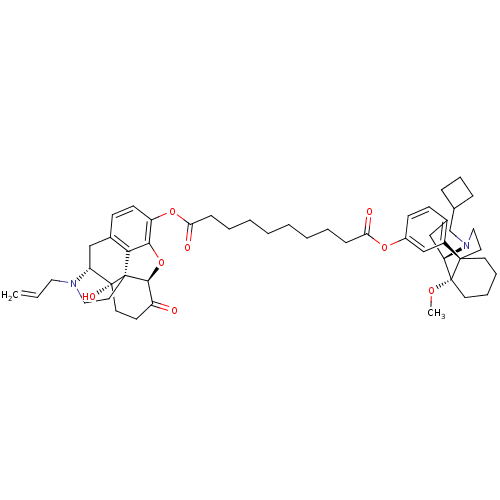 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343629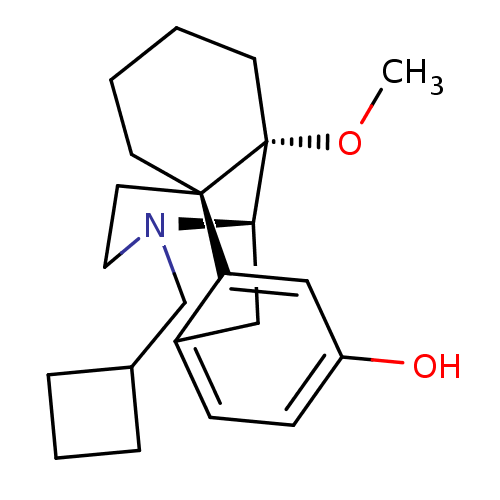 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343629 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50135797 (CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50240437 ((-)-17-(cyclobutylmethyl)morphinan-3,14-diol | (-)...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50135808 ((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50000788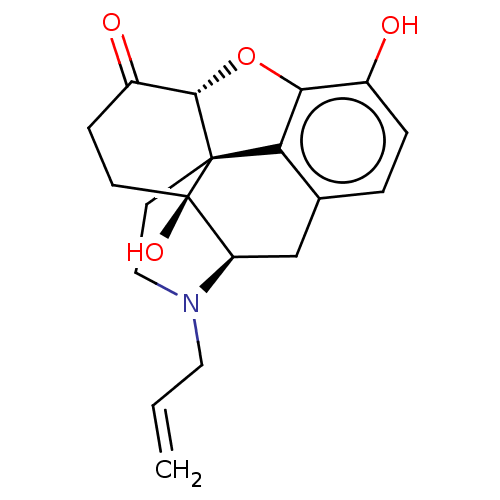 ((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50000788 ((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343628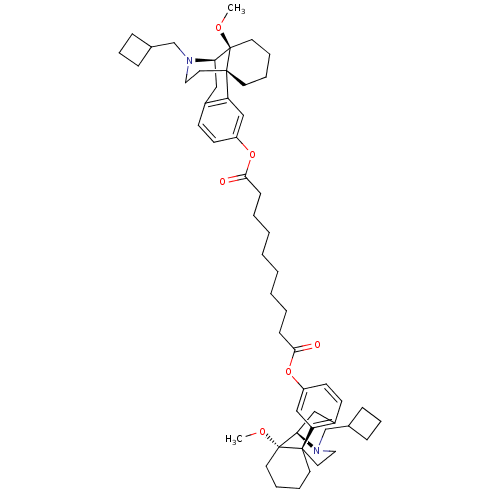 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343623 ((-)-3,30-(Decane-1,10-diaminothiazolo)bis(cyclopro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343628 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343630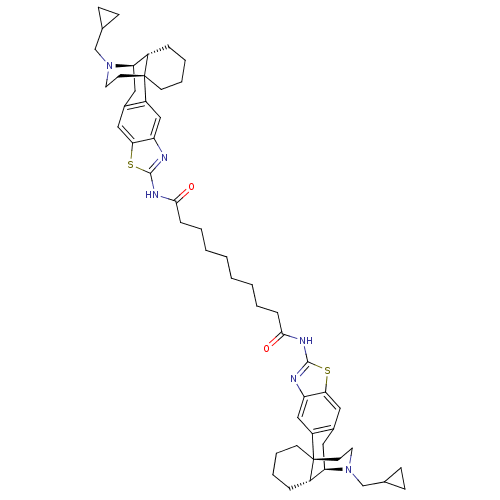 (Bis((-)-3-aminothiazolo-N-cyclopropylmethylmorphin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]U69563 from human kappa-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343626 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50210557 (CHEMBL242756 | MCL-147) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343623 ((-)-3,30-(Decane-1,10-diaminothiazolo)bis(cyclopro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50135806 (CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50135808 ((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 5.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343625 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343629 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 8.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343630 (Bis((-)-3-aminothiazolo-N-cyclopropylmethylmorphin...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu-opioid receptor expressed in CHO cells after 60 mins by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50135797 (CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50240437 ((-)-17-(cyclobutylmethyl)morphinan-3,14-diol | (-)...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343628 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50210557 (CHEMBL242756 | MCL-147) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50000788 ((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343626 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343623 ((-)-3,30-(Decane-1,10-diaminothiazolo)bis(cyclopro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50343630 (Bis((-)-3-aminothiazolo-N-cyclopropylmethylmorphin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Displacement of [3H]naltrindole from human delta-opioid receptor expressed in CHO cells after 3 hrs by scintillation counting | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343625 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343625 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343626 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343629 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343628 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343626 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343629 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human kappa-opioid receptor expressed in CHO cells assessed as inhibition of U69593-induced [35S]GTPgammaS binding after 60 mi... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343628 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343623 ((-)-3,30-(Decane-1,10-diaminothiazolo)bis(cyclopro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 390 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Antagonist activity at human mu-opioid receptor expressed in CHO cells assessed as inhibition of DAMGO-induced [35S]GTPgammaS binding after 60 mins b... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343629 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343628 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343626 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 18 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343630 (Bis((-)-3-aminothiazolo-N-cyclopropylmethylmorphin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343625 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.840 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343630 (Bis((-)-3-aminothiazolo-N-cyclopropylmethylmorphin...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 130 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343624 (5alpha-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphina...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343625 ((-)-17-N-Cyclobutylmethyl)morphinan-3-yl-(-)-14-hy...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343623 ((-)-3,30-(Decane-1,10-diaminothiazolo)bis(cyclopro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 22 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343623 ((-)-3,30-(Decane-1,10-diaminothiazolo)bis(cyclopro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 7.30 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343627 (Benzyl-(-)-14-methoxy-butorphanol-3-yl-decanedioat...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50343626 ((5alpha)-17-Allyl-14-hydroxy-6-oxo-4,5-epoxymorphi...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human mu-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillation ... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343629 ((-)-14-Methoxybutorphanol | CHEMBL1774946) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.80 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50343628 (Bis((-)-14-methoxy-17-N-cyclobutylmethyl)morphinan...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a |
Harvard Medical School Curated by ChEMBL | Assay Description Agonist activity at human kappa-opioid receptor expressed in CHO cells assessed as stimulation of [35S]GTPgammaS binding after 60 mins by scintillati... | Bioorg Med Chem 19: 2808-16 (2011) Article DOI: 10.1016/j.bmc.2011.03.052 BindingDB Entry DOI: 10.7270/Q2KW5GB1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||