

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aurora kinase A (Homo sapiens (Human)) | BDBM81552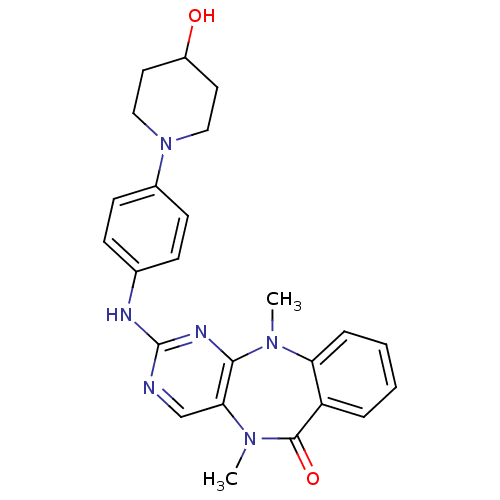 (Benzo[e]pyrimido[5,4-b][1,4]diazepin-6(11H)-one, 1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase B (Homo sapiens (Human)) | BDBM81552 (Benzo[e]pyrimido[5,4-b][1,4]diazepin-6(11H)-one, 1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aurora kinase C (Homo sapiens (Human)) | BDBM81552 (Benzo[e]pyrimido[5,4-b][1,4]diazepin-6(11H)-one, 1...) | PDB NCI pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase pim-2 (Homo sapiens (Human)) | BDBM81550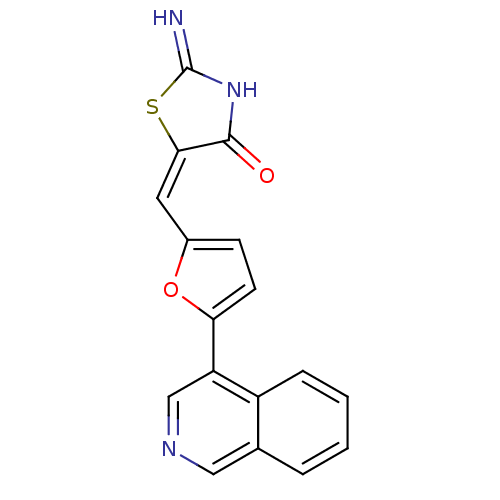 (Furan thiazolidinediones, A47) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 49 | 23 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase pim-1 (Homo sapiens (Human)) | BDBM81550 (Furan thiazolidinediones, A47) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 49 | 40 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Homeodomain-interacting protein kinase 2 (Homo sapiens (Human)) | BDBM81551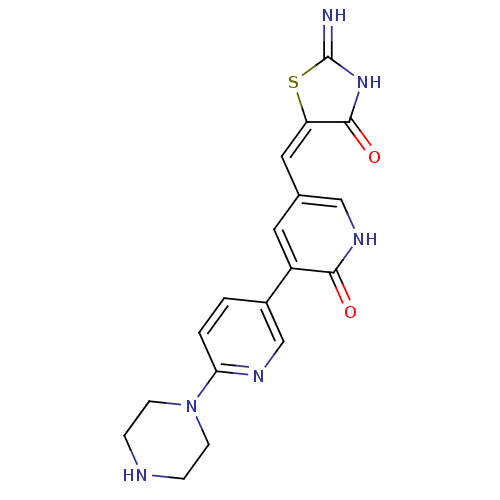 (Furan thiazolidinediones, A64) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 74 | 9.5 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dual specificity protein kinase TTK (Homo sapiens (Human)) | BDBM36486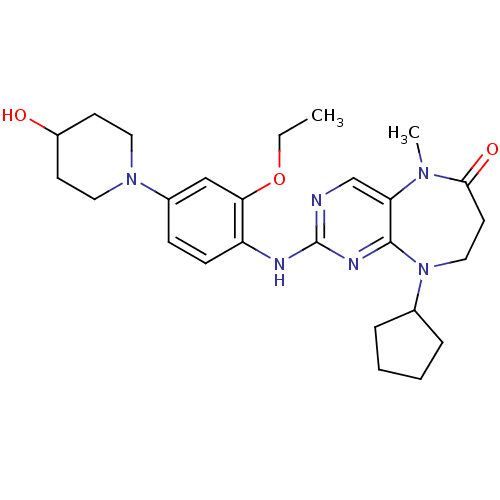 (9-Cyclopentyl-2-(2-ethoxy-4-(4-hydroxypiperidin-1-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 145 | 26 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 7 (Homo sapiens (Human)) | BDBM50337134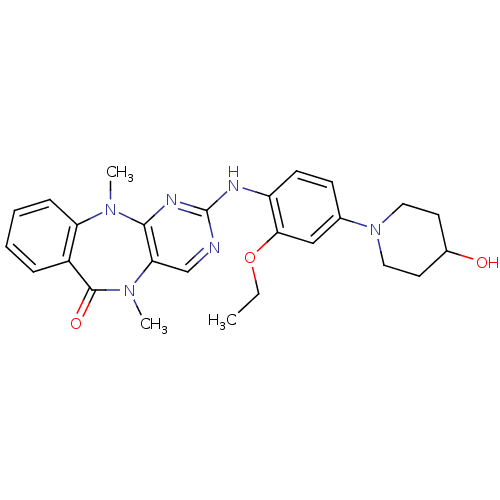 (2-((2-ethoxy-4-(4-hydroxypiperidin-1-yl)phenyl)ami...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Serine/threonine-protein kinase pim-3 (Homo sapiens (Human)) | BDBM81550 (Furan thiazolidinediones, A47) | KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Activated CDC42 kinase 1 (Homo sapiens (Human)) | BDBM50337126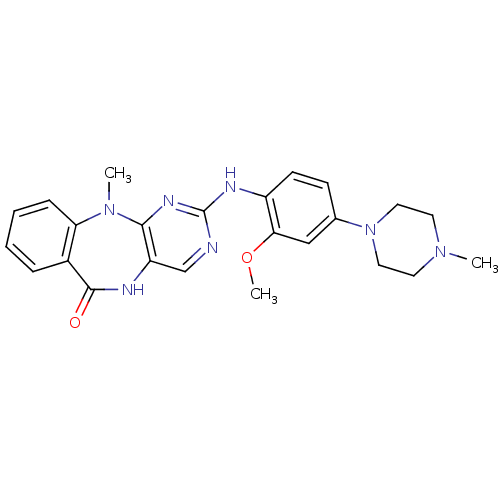 (2-(2-methoxy-4-(4-methylpiperazin-1-yl)phenylamino...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School | Assay Description In vitro biochemical assays were performed in parallel to determine the most potent tool compound. | Chem Biol 18: 868-79 (2011) Article DOI: 10.1016/j.chembiol.2011.05.010 BindingDB Entry DOI: 10.7270/Q2HD7T57 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||