

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50149381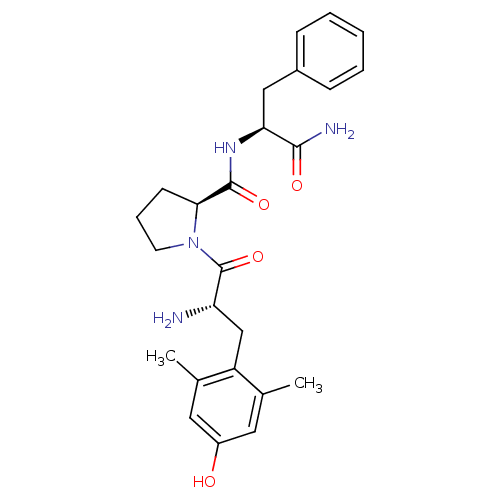 (1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-phenyl)-p...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor (unknown origin) | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009271 (CHEMBL3233014) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009272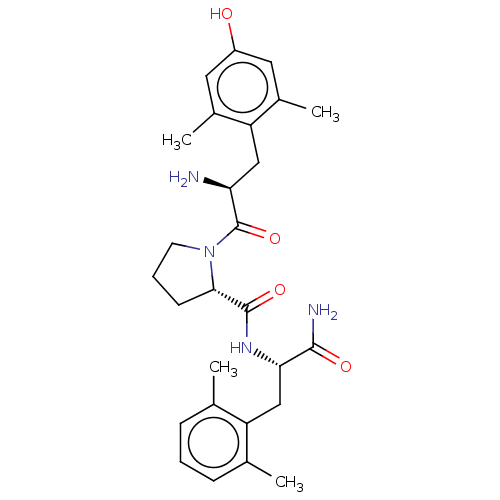 (CHEMBL3233200) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009268 (CHEMBL3233197) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009266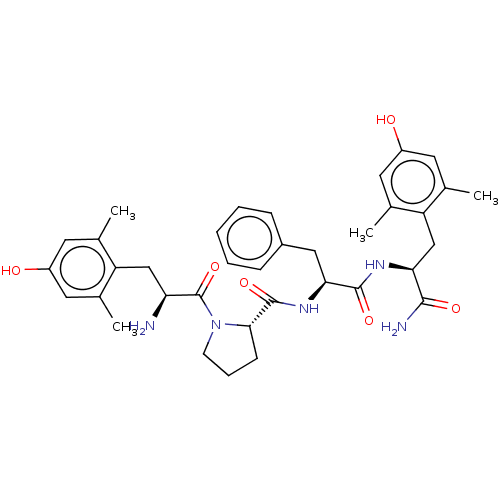 (CHEMBL3233195) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009251 (CHEMBL3233191) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009265 (CHEMBL3233194) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50166065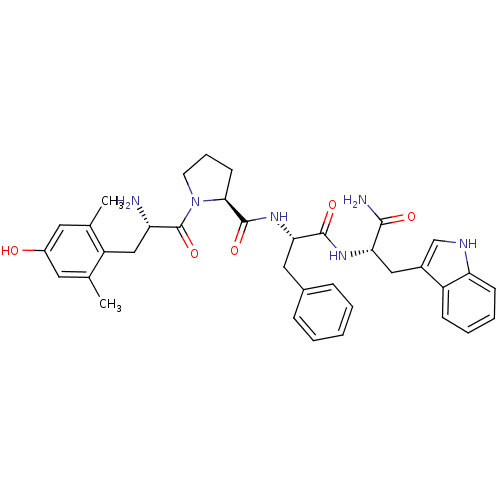 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-pheny...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009264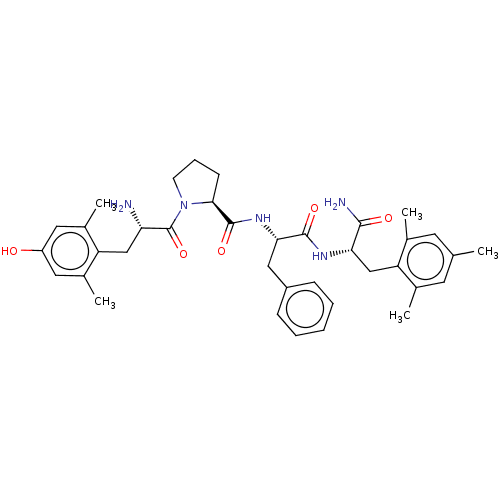 (CHEMBL3233193) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009267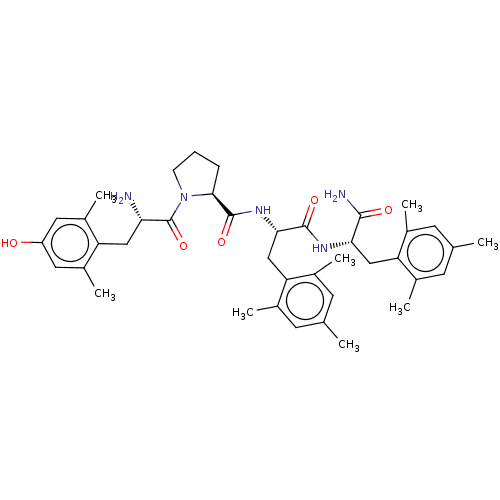 (CHEMBL3233196) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009269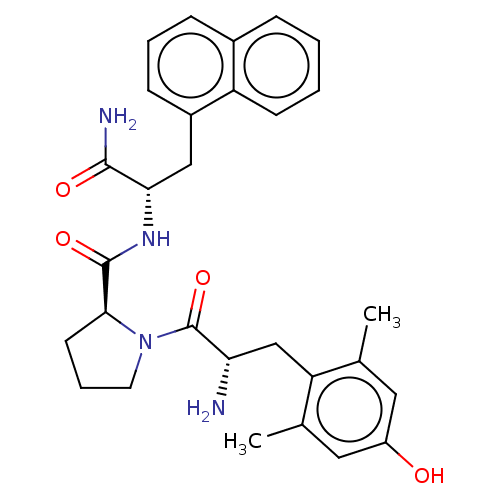 (CHEMBL3233198) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009254 (CHEMBL3233192) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50139013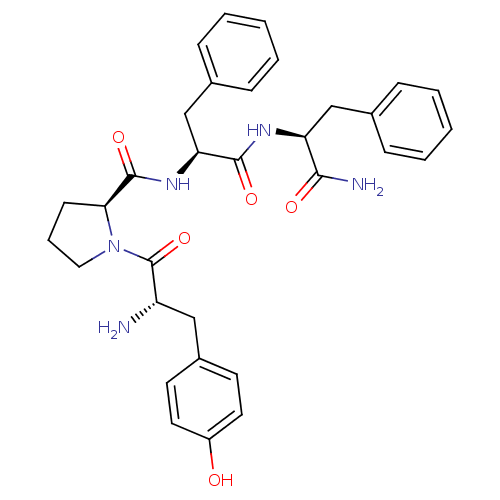 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Binding affinity to mu opioid receptor (unknown origin) | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009267 (CHEMBL3233196) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM21025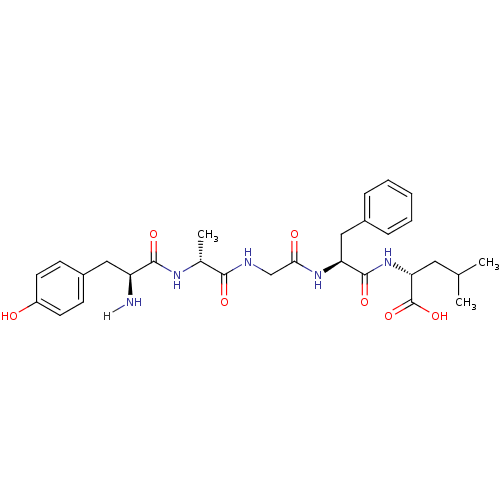 ((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in guinea pig brain membranes after 1 hr | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009271 (CHEMBL3233014) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009270 (CHEMBL3233199) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from mu opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009254 (CHEMBL3233192) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50166065 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-pheny...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009264 (CHEMBL3233193) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009272 (CHEMBL3233200) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009265 (CHEMBL3233194) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009251 (CHEMBL3233191) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50149381 (1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-phenyl)-p...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from delta opioid receptor (unknown origin) | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009268 (CHEMBL3233197) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 66 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009270 (CHEMBL3233199) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009266 (CHEMBL3233195) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 87 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50009269 (CHEMBL3233198) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 121 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Displacement of [3H]deltorphin-2 from delta opioid receptor in Sprague-Dawley rat brain P2 synaptosomal membranes | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50139013 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 6.09E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Binding affinity to delta opioid receptor (unknown origin) | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009272 (CHEMBL3233200) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.274 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM21025 ((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50149381 (1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-phenyl)-p...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009271 (CHEMBL3233014) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009269 (CHEMBL3233198) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009267 (CHEMBL3233196) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009268 (CHEMBL3233197) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM21025 ((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009251 (CHEMBL3233191) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009254 (CHEMBL3233192) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009266 (CHEMBL3233195) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009264 (CHEMBL3233193) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50009268 (CHEMBL3233197) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | <100 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor (unknown origin) expressed in HEK293 cells assessed as inhibition of forskolin-induced cAMP accumulation af... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50149381 (1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-phenyl)-p...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 113 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50166065 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-pheny...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 207 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009265 (CHEMBL3233194) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 249 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009272 (CHEMBL3233200) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 745 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009269 (CHEMBL3233198) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009268 (CHEMBL3233197) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50009270 (CHEMBL3233199) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at mu opioid receptor in guinea pig ileum assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009254 (CHEMBL3233192) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009271 (CHEMBL3233014) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009265 (CHEMBL3233194) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009251 (CHEMBL3233191) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009267 (CHEMBL3233196) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50166065 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-2,6-dimethyl-pheny...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009264 (CHEMBL3233193) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009266 (CHEMBL3233195) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50009270 (CHEMBL3233199) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at delta opioid receptor in mouse vas deferens assessed as inhibition of electrically-stimulated muscle contraction | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM21025 ((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at GFP-fused delta opioid receptor (unknown origin) expressed in HEK293 cells assessed as recruitment of beta-arrestin-2 after 10 mi... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50009271 (CHEMBL3233014) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 19 | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at GFP-fused mu opioid receptor (unknown origin) expressed in HEK293 cells assessed as recruitment of beta-arrestin-2 after 10 mins ... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM21015 ((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 378 | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at GFP-fused mu opioid receptor (unknown origin) expressed in HEK293 cells assessed as recruitment of beta-arrestin-2 after 10 mins ... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM21025 ((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at GFP-fused mu opioid receptor (unknown origin) expressed in HEK293 cells assessed as recruitment of beta-arrestin-2 after 10 mins ... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50009268 (CHEMBL3233197) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 6.30 | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at GFP-fused mu opioid receptor (unknown origin) expressed in HEK293 cells assessed as recruitment of beta-arrestin-2 after 10 mins ... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50381677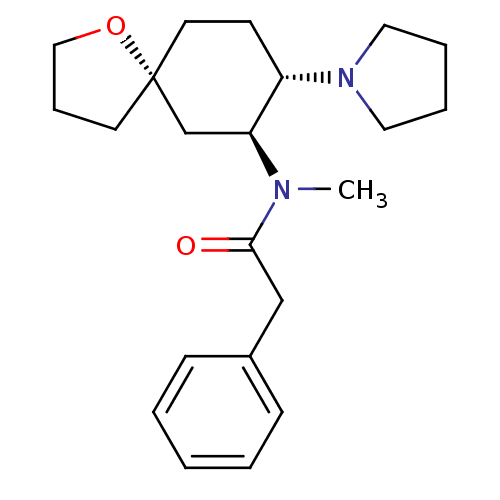 (CHEMBL1256748 | U-69593) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 534 | n/a | n/a | n/a | n/a |
Nanjing Medical University Curated by ChEMBL | Assay Description Agonist activity at GFP-fused kappa opioid receptor (unknown origin) expressed in HEK293 cells assessed as recruitment of beta-arrestin-2 after 10 mi... | Bioorg Med Chem 22: 2208-19 (2014) Article DOI: 10.1016/j.bmc.2014.02.015 BindingDB Entry DOI: 10.7270/Q29Z96DB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||