 Found 51 hits of Enzyme Inhibition Constant Data
Found 51 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400781
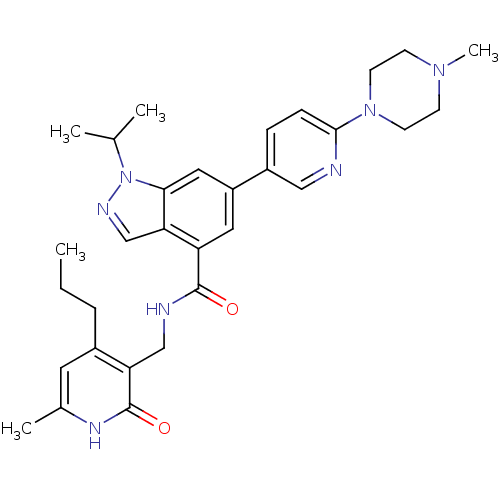
(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 79 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 174 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400779
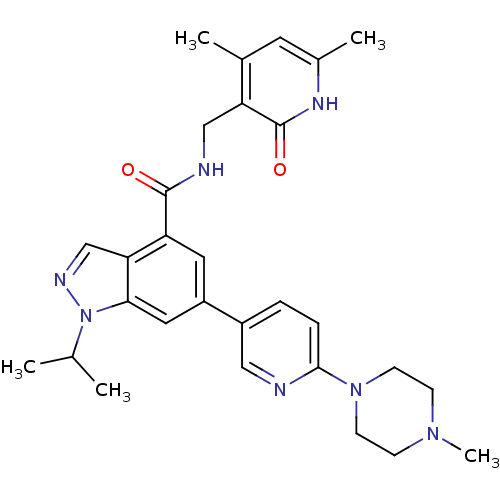
(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 324 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400780
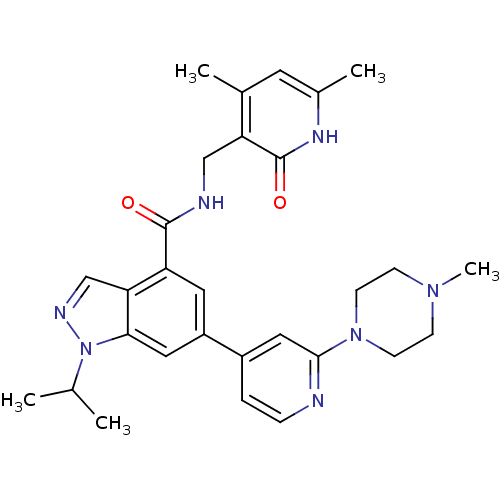
(CHEMBL2204996)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccnc(c1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-30-27(15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400782
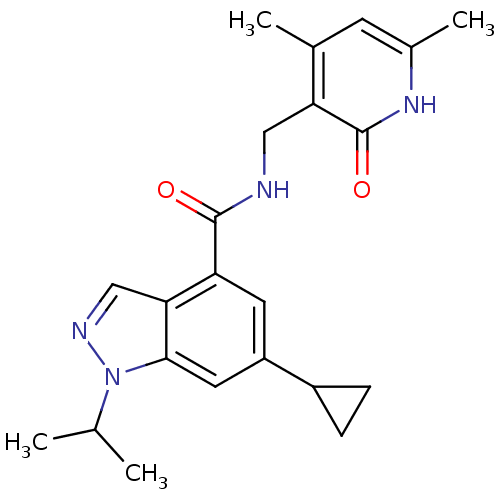
(CHEMBL2204999)Show SMILES CC(C)n1ncc2c(cc(cc12)C1CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C22H26N4O2/c1-12(2)26-20-9-16(15-5-6-15)8-17(19(20)11-24-26)21(27)23-10-18-13(3)7-14(4)25-22(18)28/h7-9,11-12,15H,5-6,10H2,1-4H3,(H,23,27)(H,25,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EZH2
(Homo sapiens (Human)) | BDBM50400783
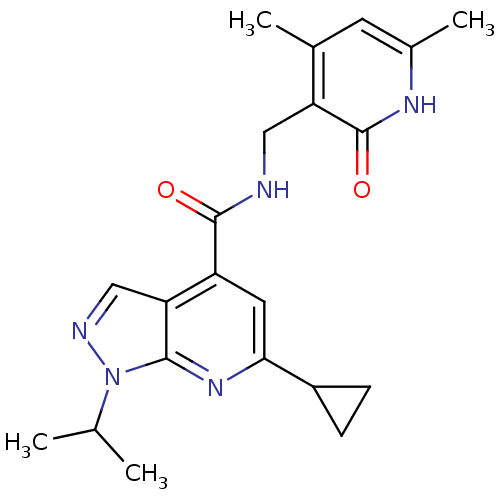
(CHEMBL1608462)Show SMILES CC(C)n1ncc2c(cc(nc12)C1CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C21H25N5O2/c1-11(2)26-19-17(10-23-26)15(8-18(25-19)14-5-6-14)20(27)22-9-16-12(3)7-13(4)24-21(16)28/h7-8,10-11,14H,5-6,9H2,1-4H3,(H,22,27)(H,24,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.06E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of EZH2-mediated proliferation of human LNCaP cells after 6 days by chemiluminescence analysis |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SETD7
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SET7 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 3 [N508S]
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT3 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SETD7
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SET7 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 3 [N508S]
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT3 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H2
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H2 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase 2B
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MLL4 using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 3B
(Mus musculus) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of mouse DNMT3b1 using lambda DNA as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SETMAR
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SETMAR using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 5
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT5 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 1 [11-371]
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT1 using histone H4 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EHMT2
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of G9a using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 3A
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DNMT3a using lambda DNA as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 1
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DNMT1 using polydi-dc as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 3 [N508S]
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT3 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 3B
(Mus musculus) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of mouse DNMT3b1 using lambda DNA as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 3A
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DNMT3a using lambda DNA as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 1
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DNMT1 using polydi-dc as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DOT1L using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EHMT2
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of G9a using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase 2C
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MLL3 using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 1 [11-371]
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT1 using histone H4 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H1
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H1 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H2
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H2 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 3B
(Mus musculus) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of mouse DNMT3b1 using lambda DNA as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 1 [11-371]
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT1 using histone H4 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-arginine methyltransferase CARM1
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT4 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase EHMT2
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of G9a using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase 2D
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MLL2 using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SETMAR
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SETMAR using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SETMAR
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SETMAR using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 3A
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DNMT3a using lambda DNA as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 5
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT5 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase 2A
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of MLL1 using core histone as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Protein arginine N-methyltransferase 5
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT5 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H1
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H1 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H1
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H1 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-arginine methyltransferase CARM1
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.16E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT4 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
DNA (cytosine-5)-methyltransferase 1
(Homo sapiens (Human)) | BDBM50400781

(CHEMBL2204997)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccc(nc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-22-14-21(4)35-31(40)26(22)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)23-8-9-29(32-17-23)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.21E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of DNMT1 using polydi-dc as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SUV39H2
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.23E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SUV39H2 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-arginine methyltransferase CARM1
(Homo sapiens (Human)) | BDBM50400778

(CHEMBL2204995)Show SMILES CCCc1cc(C)[nH]c(=O)c1CNC(=O)c1cc(cc2n(ncc12)C(C)C)-c1ccnc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H39N7O2/c1-6-7-23-14-21(4)35-31(40)26(23)18-33-30(39)25-15-24(16-28-27(25)19-34-38(28)20(2)3)22-8-9-32-29(17-22)37-12-10-36(5)11-13-37/h8-9,14-17,19-20H,6-7,10-13,18H2,1-5H3,(H,33,39)(H,35,40) | PDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.28E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PRMT4 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SETD7
(Homo sapiens (Human)) | BDBM50400779

(CHEMBL2204998)Show SMILES CC(C)n1ncc2c(cc(cc12)-c1ccc(nc1)N1CCN(C)CC1)C(=O)NCc1c(C)cc(C)[nH]c1=O Show InChI InChI=1S/C29H35N7O2/c1-18(2)36-26-14-22(21-6-7-27(30-15-21)35-10-8-34(5)9-11-35)13-23(25(26)17-32-36)28(37)31-16-24-19(3)12-20(4)33-29(24)38/h6-7,12-15,17-18H,8-11,16H2,1-5H3,(H,31,37)(H,33,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.14E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of SET7 using histone H3 as substrate preincubated for 10 mins followed by addition of [3H]SAM and incubated for 60 mins |
ACS Med Chem Lett 3: 1091-1096 (2012)
Article DOI: 10.1021/ml3003346
BindingDB Entry DOI: 10.7270/Q2NK3G60 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data