 Found 9 hits of Enzyme Inhibition Constant Data
Found 9 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059910
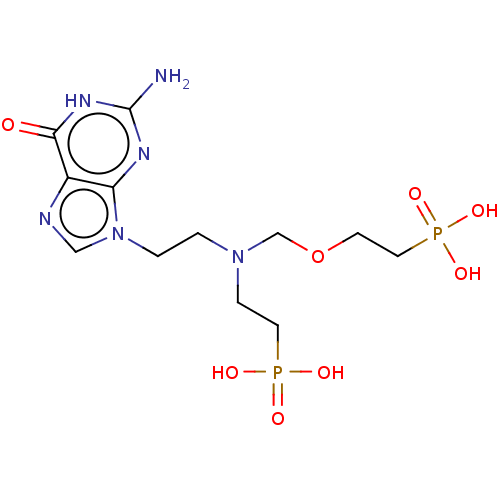
(CHEMBL3394315)Show SMILES Nc1nc2n(CCN(CCP(O)(O)=O)COCCP(O)(O)=O)cnc2c(=O)[nH]1 Show InChI InChI=1S/C12H22N6O8P2/c13-12-15-10-9(11(19)16-12)14-7-18(10)2-1-17(3-5-27(20,21)22)8-26-4-6-28(23,24)25/h7H,1-6,8H2,(H2,20,21,22)(H2,23,24,25)(H3,13,15,16,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059908

(CHEMBL3394327)Show SMILES Nc1nc2n(CCN(CCOCCP(O)(O)=O)CCP(O)(O)=O)c(Br)nc2c(=O)[nH]1 Show InChI InChI=1S/C13H23BrN6O8P2/c14-12-16-9-10(17-13(15)18-11(9)21)20(12)2-1-19(4-7-29(22,23)24)3-5-28-6-8-30(25,26)27/h1-8H2,(H2,22,23,24)(H2,25,26,27)(H3,15,17,18,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50392261
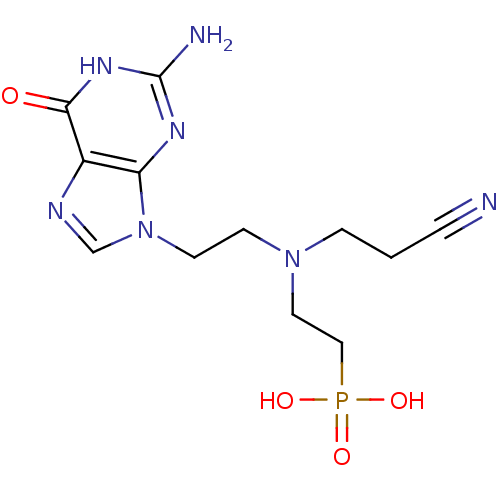
(CHEMBL2153480)Show SMILES Nc1nc2n(CCN(CCC#N)CCP(O)(O)=O)cnc2c(=O)[nH]1 Show InChI InChI=1S/C12H18N7O4P/c13-2-1-3-18(6-7-24(21,22)23)4-5-19-8-15-9-10(19)16-12(14)17-11(9)20/h8H,1,3-7H2,(H2,21,22,23)(H3,14,16,17,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059909
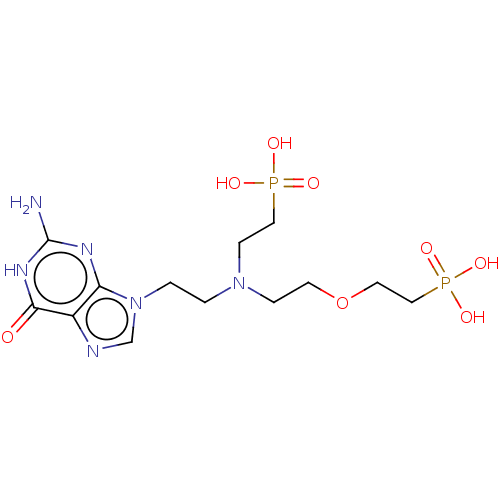
(CHEMBL3394316)Show SMILES Nc1nc2n(CCN(CCOCCP(O)(O)=O)CCP(O)(O)=O)cnc2c(=O)[nH]1 Show InChI InChI=1S/C13H24N6O8P2/c14-13-16-11-10(12(20)17-13)15-9-19(11)2-1-18(4-7-28(21,22)23)3-5-27-6-8-29(24,25)26/h9H,1-8H2,(H2,21,22,23)(H2,24,25,26)(H3,14,16,17,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059913
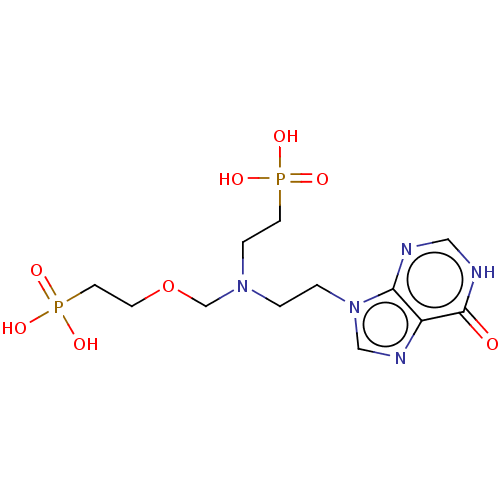
(CHEMBL3394312)Show SMILES OP(O)(=O)CCOCN(CCn1cnc2c1nc[nH]c2=O)CCP(O)(O)=O Show InChI InChI=1S/C12H21N5O8P2/c18-12-10-11(13-7-14-12)17(8-15-10)2-1-16(3-5-26(19,20)21)9-25-4-6-27(22,23)24/h7-8H,1-6,9H2,(H,13,14,18)(H2,19,20,21)(H2,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059911

(CHEMBL3394314)Show SMILES Nc1nc2n(CCN(CCCCP(O)(O)=O)CCP(O)(O)=O)cnc2c(=O)[nH]1 Show InChI InChI=1S/C13H24N6O7P2/c14-13-16-11-10(12(20)17-13)15-9-19(11)5-4-18(6-8-28(24,25)26)3-1-2-7-27(21,22)23/h9H,1-8H2,(H2,21,22,23)(H2,24,25,26)(H3,14,16,17,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059912

(CHEMBL3394313)Show SMILES OP(O)(=O)CCOCCN(CCn1cnc2c1nc[nH]c2=O)CCP(O)(O)=O Show InChI InChI=1S/C13H23N5O8P2/c19-13-11-12(14-9-15-13)18(10-16-11)2-1-17(4-7-27(20,21)22)3-5-26-6-8-28(23,24)25/h9-10H,1-8H2,(H,14,15,19)(H2,20,21,22)(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50392251
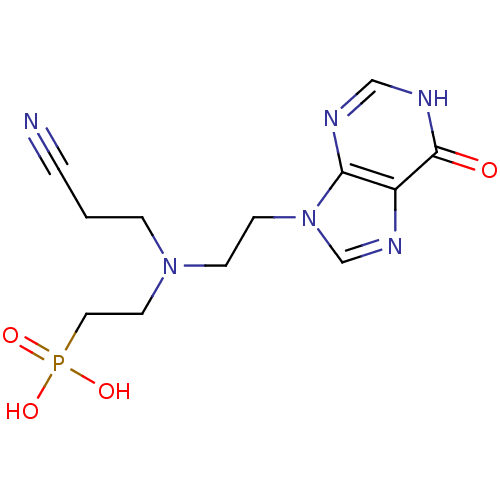
(CHEMBL2153490)Show InChI InChI=1S/C12H17N6O4P/c13-2-1-3-17(6-7-23(20,21)22)4-5-18-9-16-10-11(18)14-8-15-12(10)19/h8-9H,1,3-7H2,(H,14,15,19)(H2,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |
Hypoxanthine-guanine phosphoribosyltransferase
(Homo sapiens (Human)) | BDBM50059914

(CHEMBL3394311)Show SMILES OP(O)(=O)CCCCN(CCn1cnc2c1nc[nH]c2=O)CCP(O)(O)=O Show InChI InChI=1S/C13H23N5O7P2/c19-13-11-12(14-9-15-13)18(10-16-11)5-4-17(6-8-27(23,24)25)3-1-2-7-26(20,21)22/h9-10H,1-8H2,(H,14,15,19)(H2,20,21,22)(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human HGPRT |
J Med Chem 58: 827-46 (2015)
Article DOI: 10.1021/jm501416t
BindingDB Entry DOI: 10.7270/Q2HM5B3V |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data