

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Farnesyl pyrophosphate synthase (Homo sapiens (Human)) | BDBM12576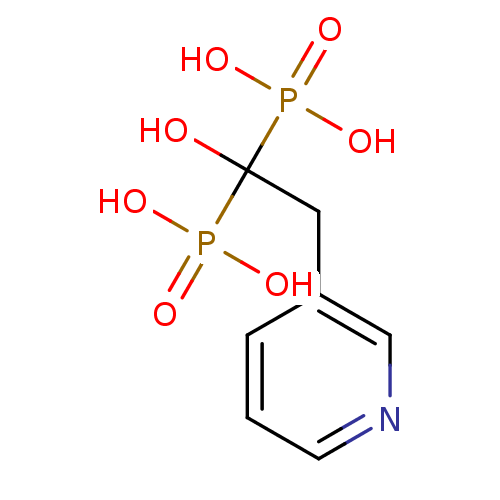 (Bisphosphonate 1 | CHEMBL923 | JMC515594 Compound ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Iowa Curated by ChEMBL | Assay Description Inhibition of FDPS (unknown origin) | Bioorg Med Chem Lett 25: 2331-4 (2015) Article DOI: 10.1016/j.bmcl.2015.04.021 BindingDB Entry DOI: 10.7270/Q2M61MZN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Farnesyl pyrophosphate synthase (Homo sapiens (Human)) | BDBM50226002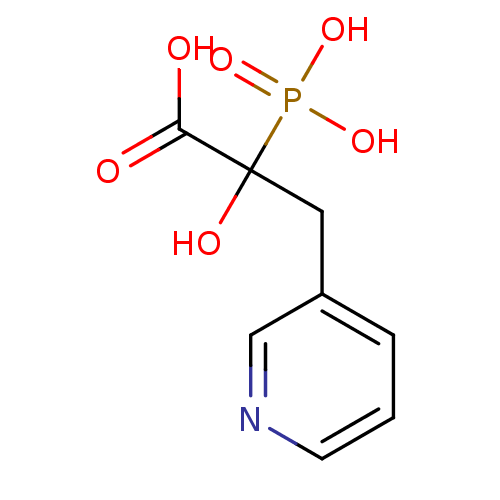 ((R)-2-hydroxy-2-phosphono-3-(pyridin-3-yl)propanoi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Iowa Curated by ChEMBL | Assay Description Inhibition of recombinant FDPS (unknown origin) assessed as decrease in radiolabeld GGPP level using GPP and [14C]IPP as substrate treated with enzym... | Bioorg Med Chem Lett 25: 2331-4 (2015) Article DOI: 10.1016/j.bmcl.2015.04.021 BindingDB Entry DOI: 10.7270/Q2M61MZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Farnesyl pyrophosphate synthase (Homo sapiens (Human)) | BDBM50083066 (CHEMBL240776) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Iowa Curated by ChEMBL | Assay Description Inhibition of recombinant FDPS (unknown origin) assessed as decrease in radiolabeld GGPP level using GPP and [14C]IPP as substrate treated with enzym... | Bioorg Med Chem Lett 25: 2331-4 (2015) Article DOI: 10.1016/j.bmcl.2015.04.021 BindingDB Entry DOI: 10.7270/Q2M61MZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Farnesyl pyrophosphate synthase (Homo sapiens (Human)) | BDBM50083067 (CHEMBL3422846) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Iowa Curated by ChEMBL | Assay Description Inhibition of recombinant FDPS (unknown origin) assessed as decrease in radiolabeld GGPP level using GPP and [14C]IPP as substrate treated with enzym... | Bioorg Med Chem Lett 25: 2331-4 (2015) Article DOI: 10.1016/j.bmcl.2015.04.021 BindingDB Entry DOI: 10.7270/Q2M61MZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Farnesyl pyrophosphate synthase (Homo sapiens (Human)) | BDBM50083065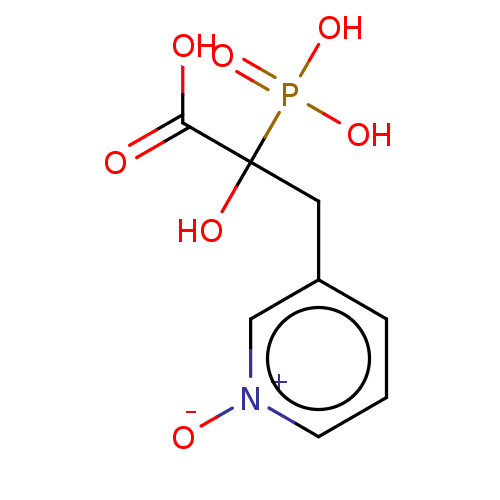 (CHEMBL3422845) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Iowa Curated by ChEMBL | Assay Description Inhibition of recombinant FDPS (unknown origin) assessed as decrease in radiolabeld GGPP level using GPP and [14C]IPP as substrate treated with enzym... | Bioorg Med Chem Lett 25: 2331-4 (2015) Article DOI: 10.1016/j.bmcl.2015.04.021 BindingDB Entry DOI: 10.7270/Q2M61MZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||