

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50346601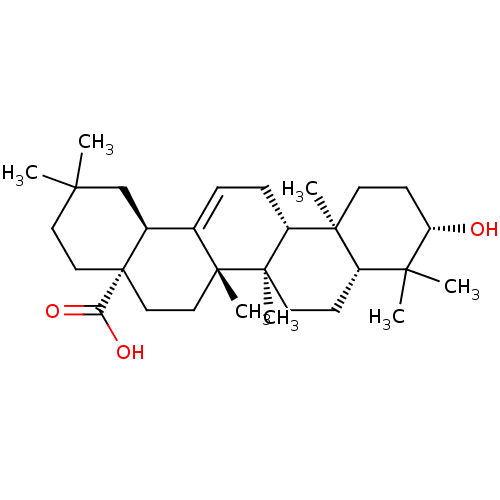 (NSC-114945 | OLEANOLIC_ACID | Oleanolic acid | Ole...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092798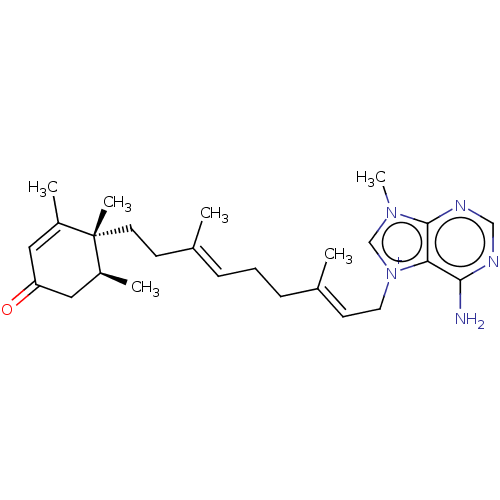 (CHEMBL3586407) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092796 (CHEMBL3586406) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092801 (CHEMBL3586409) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092800 (CHEMBL3586408) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092807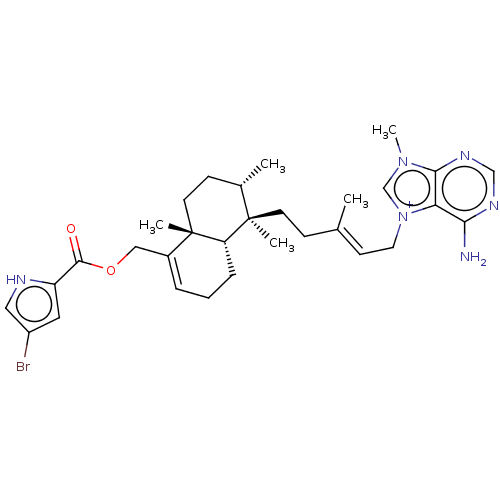 (CHEMBL3586411) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092805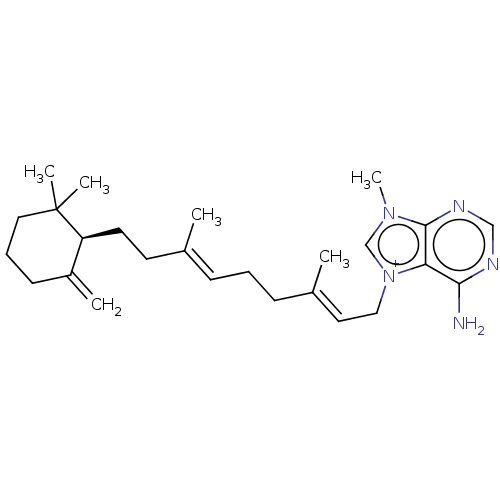 (CHEMBL3586410) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092808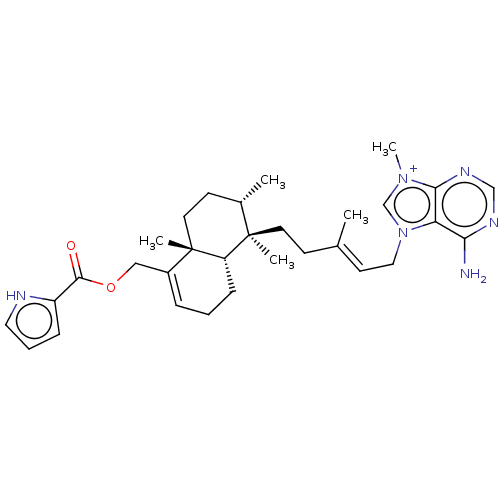 (AGELINE B) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092804 (Agelasine D) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092803 (AGELASINE B) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092806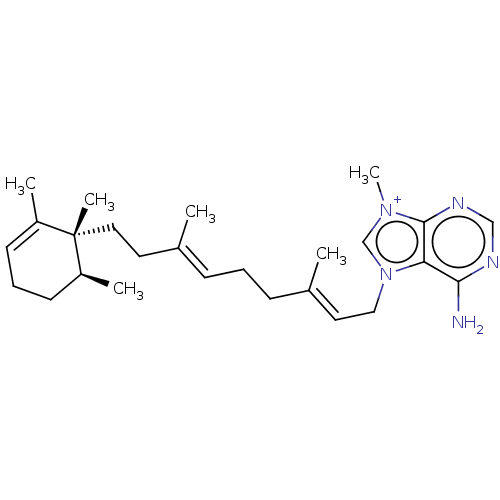 (AGELINE A) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein phosphatase non-receptor type 1 (Homo sapiens (Human)) | BDBM50092802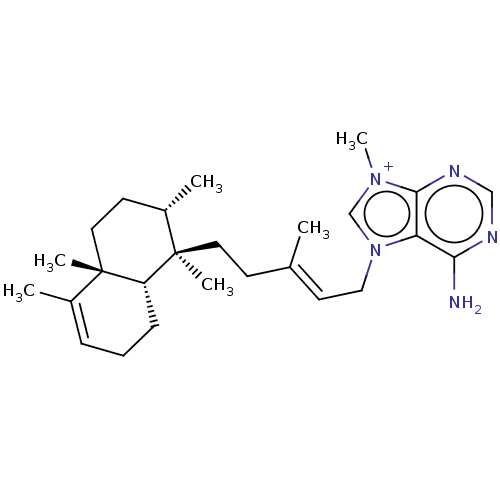 (AGELASINE A) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B using pNPP as substrate assessed as rate of hydrolysis incubated for 10 mins prior to substrate addition measur... | J Nat Prod 78: 1428-33 (2015) Article DOI: 10.1021/acs.jnatprod.5b00375 BindingDB Entry DOI: 10.7270/Q2VH5QM6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||