 Found 85 hits of Enzyme Inhibition Constant Data
Found 85 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142700
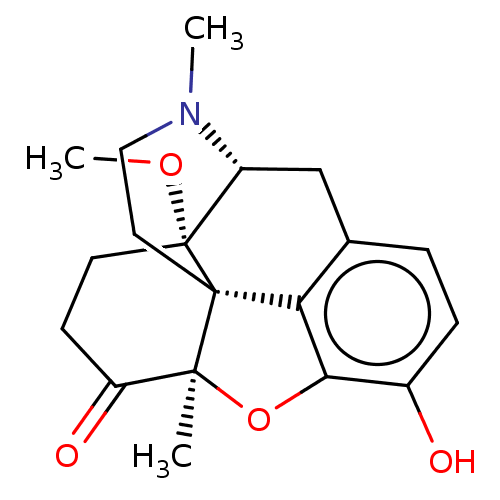
(CHEMBL326684)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)C(=O)CC[C@]1(OC)[C@@]5(CCN2C)c34 |r,TLB:23:22:16:2.3.24| Show InChI InChI=1S/C19H23NO4/c1-17-14(22)6-7-19(23-3)13-10-11-4-5-12(21)16(24-17)15(11)18(17,19)8-9-20(13)2/h4-5,13,21H,6-10H2,1-3H3/t13-,17+,18+,19-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50299217
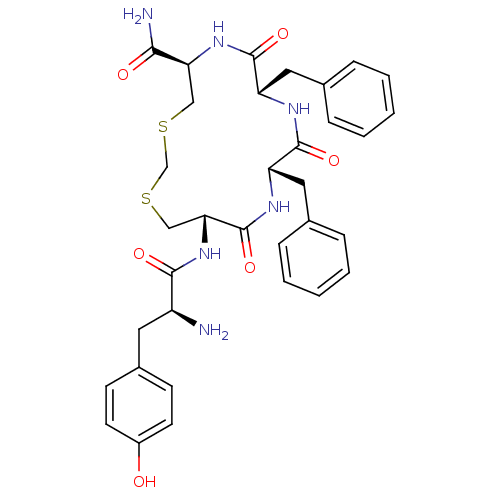
((5R,8S,11S,14S)-14-((S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H]1CSCSC[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C34H40N6O6S2/c35-25(15-23-11-13-24(41)14-12-23)31(43)40-29-19-48-20-47-18-28(30(36)42)39-33(45)27(17-22-9-5-2-6-10-22)37-32(44)26(38-34(29)46)16-21-7-3-1-4-8-21/h1-14,25-29,41H,15-20,35H2,(H2,36,42)(H,37,44)(H,38,46)(H,39,45)(H,40,43)/t25-,26-,27-,28-,29+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142622
(CHEMBL3758292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCc1nc2ccccc2[nH]1 |r| Show InChI InChI=1S/C29H31N5O3/c1-17-11-21(35)12-18(2)22(17)14-23(30)29(37)34-16-20-8-4-3-7-19(20)13-26(34)28(36)31-15-27-32-24-9-5-6-10-25(24)33-27/h3-12,23,26,35H,13-16,30H2,1-2H3,(H,31,36)(H,32,33)/t23-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142621
(CHEMBL3759292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C30H34N4O4/c1-19-12-24(35)13-20(2)25(19)15-26(31)30(38)34-18-23-11-7-6-10-22(23)14-27(34)29(37)33-17-28(36)32-16-21-8-4-3-5-9-21/h3-13,26-27,35H,14-18,31H2,1-2H3,(H,32,36)(H,33,37)/t26-,27+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142620
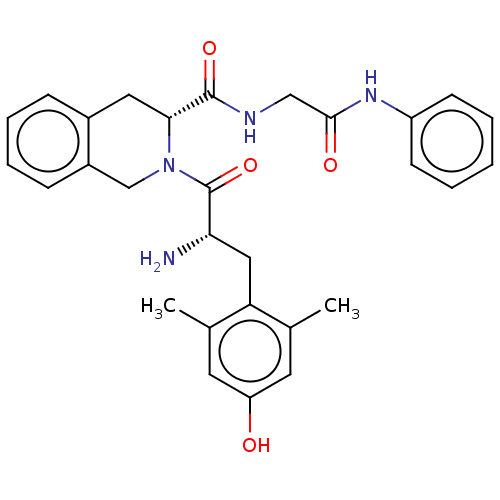
(CHEMBL3758712)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)Nc1ccccc1 |r| Show InChI InChI=1S/C29H32N4O4/c1-18-12-23(34)13-19(2)24(18)15-25(30)29(37)33-17-21-9-7-6-8-20(21)14-26(33)28(36)31-16-27(35)32-22-10-4-3-5-11-22/h3-13,25-26,34H,14-17,30H2,1-2H3,(H,31,36)(H,32,35)/t25-,26+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135806
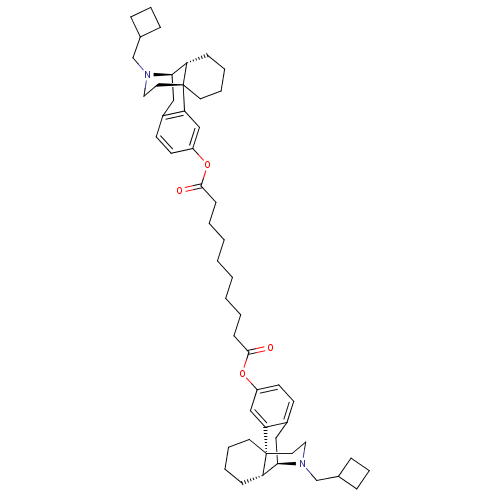
(CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C52H72N2O4/c55-49(57-41-23-21-39-31-47-43-17-7-9-25-51(43,45(39)33-41)27-29-53(47)35-37-13-11-14-37)19-5-3-1-2-4-6-20-50(56)58-42-24-22-40-32-48-44-18-8-10-26-52(44,46(40)34-42)28-30-54(48)36-38-15-12-16-38/h21-24,33-34,37-38,43-44,47-48H,1-20,25-32,35-36H2/t43-,44-,47+,48+,51+,52+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50142694
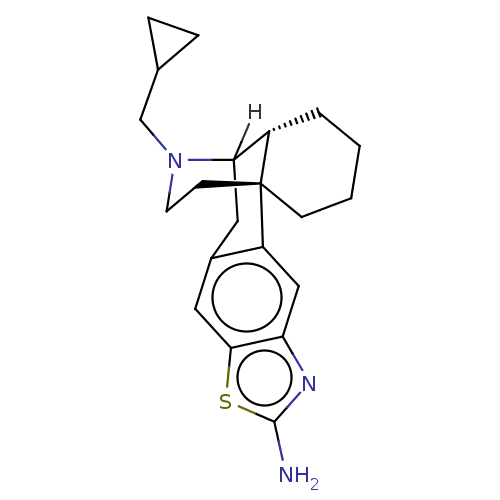
(CHEMBL3759660)Show SMILES [H]C12Cc3cc4sc(N)nc4cc3[C@]3(CCCC[C@@]13[H])CCN2CC1CC1 |r,TLB:23:22:3.12.2:18| Show InChI InChI=1S/C21H27N3S/c22-20-23-17-11-16-14(10-19(17)25-20)9-18-15-3-1-2-6-21(15,16)7-8-24(18)12-13-4-5-13/h10-11,13,15,18H,1-9,12H2,(H2,22,23)/t15-,18?,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808
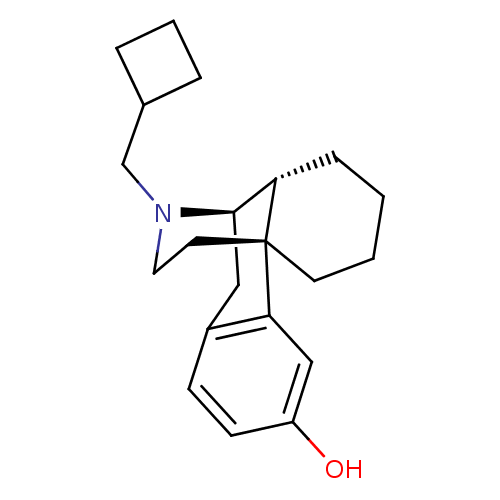
((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity to kappa opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135797
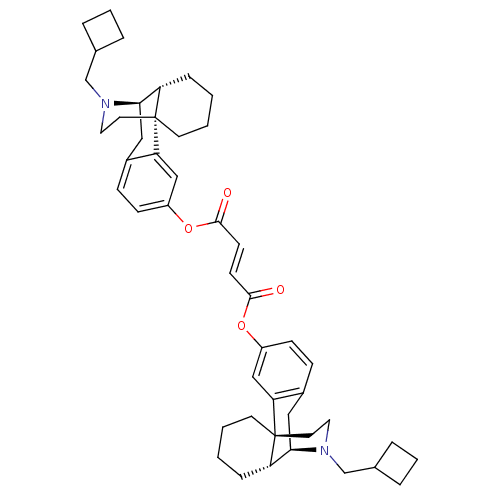
(CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...)Show SMILES O=C(Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)\C=C\C(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H58N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-18,27-28,31-32,37-38,41-42H,1-12,19-26,29-30H2/b18-17+/t37-,38-,41+,42+,45+,46+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135806

(CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C52H72N2O4/c55-49(57-41-23-21-39-31-47-43-17-7-9-25-51(43,45(39)33-41)27-29-53(47)35-37-13-11-14-37)19-5-3-1-2-4-6-20-50(56)58-42-24-22-40-32-48-44-18-8-10-26-52(44,46(40)34-42)28-30-54(48)36-38-15-12-16-38/h21-24,33-34,37-38,43-44,47-48H,1-20,25-32,35-36H2/t43-,44-,47+,48+,51+,52+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50010483
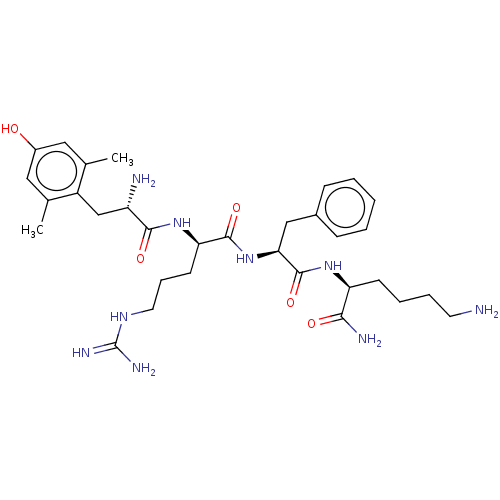
(CHEMBL2181202)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(N)=O |r| Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142621

(CHEMBL3759292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C30H34N4O4/c1-19-12-24(35)13-20(2)25(19)15-26(31)30(38)34-18-23-11-7-6-10-22(23)14-27(34)29(37)33-17-28(36)32-16-21-8-4-3-5-9-21/h3-13,26-27,35H,14-18,31H2,1-2H3,(H,32,36)(H,33,37)/t26-,27+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.151 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPN from human delta opioid receptor expressed in CHO cells by liquid scintillation counting |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142621

(CHEMBL3759292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C30H34N4O4/c1-19-12-24(35)13-20(2)25(19)15-26(31)30(38)34-18-23-11-7-6-10-22(23)14-27(34)29(37)33-17-28(36)32-16-21-8-4-3-5-9-21/h3-13,26-27,35H,14-18,31H2,1-2H3,(H,32,36)(H,33,37)/t26-,27+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142620

(CHEMBL3758712)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)Nc1ccccc1 |r| Show InChI InChI=1S/C29H32N4O4/c1-18-12-23(34)13-19(2)24(18)15-25(30)29(37)33-17-21-9-7-6-8-20(21)14-26(33)28(36)31-16-27(35)32-22-10-4-3-5-11-22/h3-13,25-26,34H,14-17,30H2,1-2H3,(H,31,36)(H,32,35)/t25-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50123658
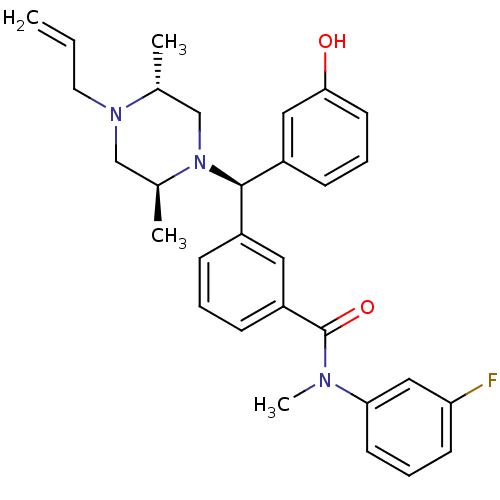
(3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...)Show SMILES C[C@@H]1CN([C@@H](c2cccc(O)c2)c2cccc(c2)C(=O)N(C)c2cccc(F)c2)[C@@H](C)CN1CC=C Show InChI InChI=1S/C30H34FN3O2/c1-5-15-33-19-22(3)34(20-21(33)2)29(24-10-7-14-28(35)17-24)23-9-6-11-25(16-23)30(36)32(4)27-13-8-12-26(31)18-27/h5-14,16-18,21-22,29,35H,1,15,19-20H2,2-4H3/t21-,22+,29-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from mu opioid receptor in albino Sprague-Dawley rat after 90 mins by liquid scintillation |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50142693
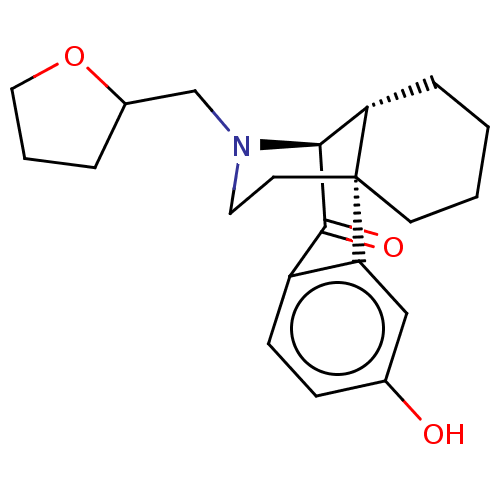
(CHEMBL3759238)Show SMILES [H][C@@]12CCCC[C@@]11CCN(CC3CCCO3)[C@]2([H])C(=O)c2ccc(O)cc12 |r,TLB:25:26:1:7.9.8,19:18:1:7.9.8,THB:10:9:20.26.18:1,2:1:20.26.18:7.9.8| Show InChI InChI=1S/C21H27NO3/c23-14-6-7-16-18(12-14)21-8-2-1-5-17(21)19(20(16)24)22(10-9-21)13-15-4-3-11-25-15/h6-7,12,15,17,19,23H,1-5,8-11,13H2/t15?,17-,19-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity to kappa opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135797

(CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...)Show SMILES O=C(Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)\C=C\C(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H58N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-18,27-28,31-32,37-38,41-42H,1-12,19-26,29-30H2/b18-17+/t37-,38-,41+,42+,45+,46+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142705
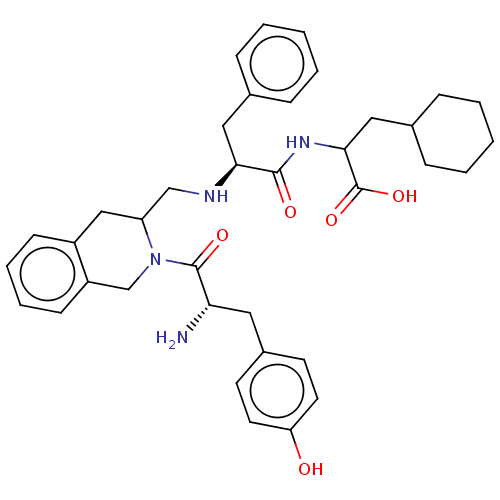
(CHEMBL3758257)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2CC1CN[C@@H](Cc1ccccc1)C(=O)NC(CC1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H46N4O5/c38-32(19-27-15-17-31(42)18-16-27)36(44)41-24-29-14-8-7-13-28(29)22-30(41)23-39-33(20-25-9-3-1-4-10-25)35(43)40-34(37(45)46)21-26-11-5-2-6-12-26/h1,3-4,7-10,13-18,26,30,32-34,39,42H,2,5-6,11-12,19-24,38H2,(H,40,43)(H,45,46)/t30?,32-,33-,34?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.259 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142695
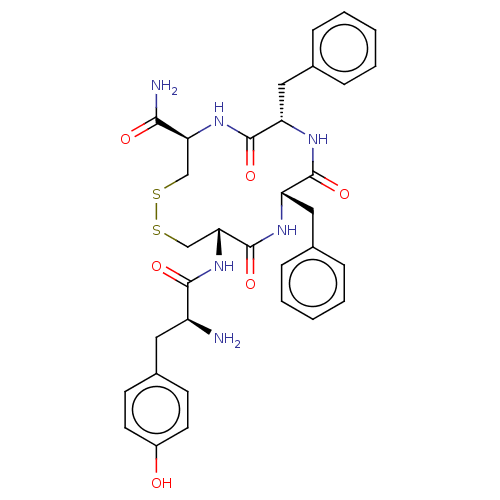
(CHEMBL3759663)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H]1CSSC[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C33H38N6O6S2/c34-24(15-22-11-13-23(40)14-12-22)30(42)39-28-19-47-46-18-27(29(35)41)38-32(44)26(17-21-9-5-2-6-10-21)36-31(43)25(37-33(28)45)16-20-7-3-1-4-8-20/h1-14,24-28,40H,15-19,34H2,(H2,35,41)(H,36,43)(H,37,45)(H,38,44)(H,39,42)/t24-,25-,26-,27-,28+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat mu opioid receptor transfected in C6 cells by liquid scintillation counting assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142693

(CHEMBL3759238)Show SMILES [H][C@@]12CCCC[C@@]11CCN(CC3CCCO3)[C@]2([H])C(=O)c2ccc(O)cc12 |r,TLB:25:26:1:7.9.8,19:18:1:7.9.8,THB:10:9:20.26.18:1,2:1:20.26.18:7.9.8| Show InChI InChI=1S/C21H27NO3/c23-14-6-7-16-18(12-14)21-8-2-1-5-17(21)19(20(16)24)22(10-9-21)13-15-4-3-11-25-15/h6-7,12,15,17,19,23H,1-5,8-11,13H2/t15?,17-,19-,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142619
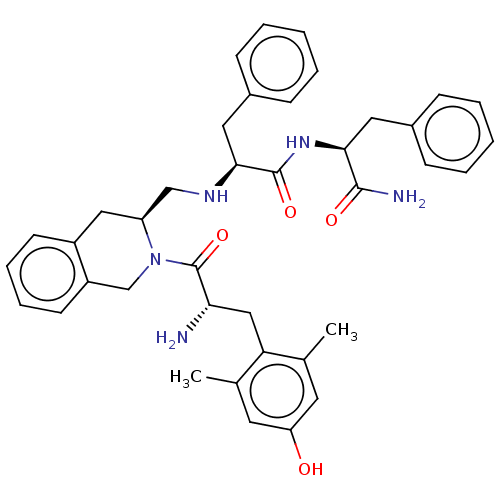
(CHEMBL3759383)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1CN[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H45N5O4/c1-25-17-32(45)18-26(2)33(25)22-34(40)39(48)44-24-30-16-10-9-15-29(30)21-31(44)23-42-36(20-28-13-7-4-8-14-28)38(47)43-35(37(41)46)19-27-11-5-3-6-12-27/h3-18,31,34-36,42,45H,19-24,40H2,1-2H3,(H2,41,46)(H,43,47)/t31-,34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50123658

(3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...)Show SMILES C[C@@H]1CN([C@@H](c2cccc(O)c2)c2cccc(c2)C(=O)N(C)c2cccc(F)c2)[C@@H](C)CN1CC=C Show InChI InChI=1S/C30H34FN3O2/c1-5-15-33-19-22(3)34(20-21(33)2)29(24-10-7-14-28(35)17-24)23-9-6-11-25(16-23)30(36)32(4)27-13-8-12-26(31)18-27/h5-14,16-18,21-22,29,35H,1,15,19-20H2,2-4H3/t21-,22+,29-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from kappa opioid receptor in albino Sprague-Dawley rat after 90 mins by liquid scintillation |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142696
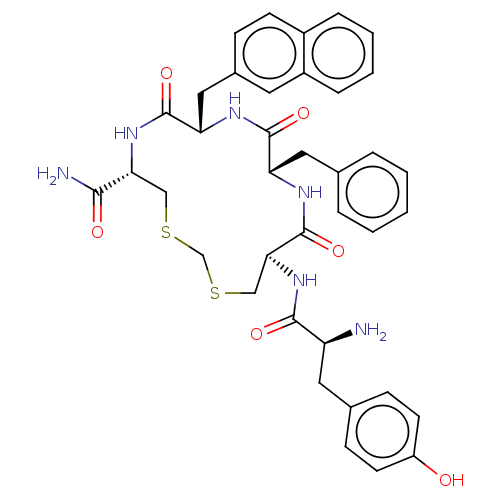
(CHEMBL3758259)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@H]1CSCSC[C@@H](NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C38H42N6O6S2/c39-29(17-24-11-14-28(45)15-12-24)35(47)44-33-21-52-22-51-20-32(34(40)46)43-37(49)31(19-25-10-13-26-8-4-5-9-27(26)16-25)41-36(48)30(42-38(33)50)18-23-6-2-1-3-7-23/h1-16,29-33,45H,17-22,39H2,(H2,40,46)(H,41,48)(H,42,50)(H,43,49)(H,44,47)/t29-,30-,31-,32+,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142696

(CHEMBL3758259)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@H]1CSCSC[C@@H](NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C38H42N6O6S2/c39-29(17-24-11-14-28(45)15-12-24)35(47)44-33-21-52-22-51-20-32(34(40)46)43-37(49)31(19-25-10-13-26-8-4-5-9-27(26)16-25)41-36(48)30(42-38(33)50)18-23-6-2-1-3-7-23/h1-16,29-33,45H,17-22,39H2,(H2,40,46)(H,41,48)(H,42,50)(H,43,49)(H,44,47)/t29-,30-,31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142622

(CHEMBL3758292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCc1nc2ccccc2[nH]1 |r| Show InChI InChI=1S/C29H31N5O3/c1-17-11-21(35)12-18(2)22(17)14-23(30)29(37)34-16-20-8-4-3-7-19(20)13-26(34)28(36)31-15-27-32-24-9-5-6-10-25(24)33-27/h3-12,23,26,35H,13-16,30H2,1-2H3,(H,31,36)(H,32,33)/t23-,26+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142697
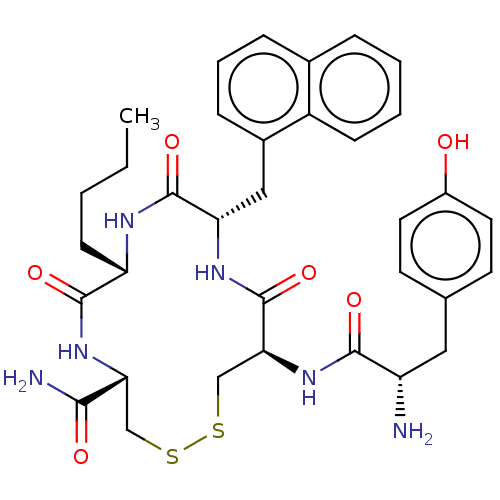
(CHEMBL3759294)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H42N6O6S2/c1-2-3-11-26-32(44)39-28(30(36)42)18-47-48-19-29(40-31(43)25(35)16-20-12-14-23(41)15-13-20)34(46)38-27(33(45)37-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,41H,2-3,11,16-19,35H2,1H3,(H2,36,42)(H,37,45)(H,38,46)(H,39,44)(H,40,43)/t25-,26-,27-,28+,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat mu opioid receptor transfected in C6 cells by liquid scintillation counting assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142704
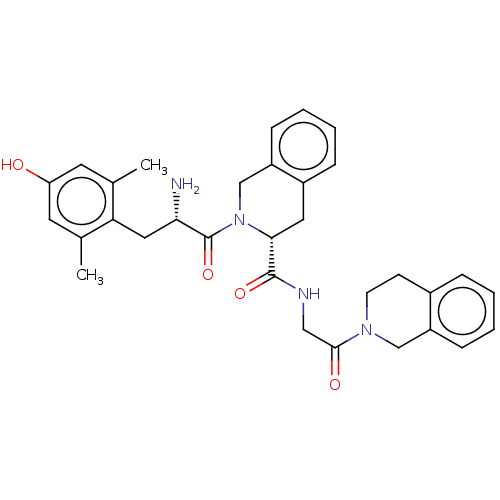
(CHEMBL3758563)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N1CCc2ccccc2C1 |r| Show InChI InChI=1S/C32H36N4O4/c1-20-13-26(37)14-21(2)27(20)16-28(33)32(40)36-19-25-10-6-4-8-23(25)15-29(36)31(39)34-17-30(38)35-12-11-22-7-3-5-9-24(22)18-35/h3-10,13-14,28-29,37H,11-12,15-19,33H2,1-2H3,(H,34,39)/t28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from human delta opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142695

(CHEMBL3759663)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H]1CSSC[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C33H38N6O6S2/c34-24(15-22-11-13-23(40)14-12-22)30(42)39-28-19-47-46-18-27(29(35)41)38-32(44)26(17-21-9-5-2-6-10-21)36-31(43)25(37-33(28)45)16-20-7-3-1-4-8-20/h1-14,24-28,40H,15-19,34H2,(H2,35,41)(H,36,43)(H,37,45)(H,38,44)(H,39,42)/t24-,25-,26-,27-,28+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat delta opioid receptor transfected in C6 cells by liquid scintillation counting assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142692
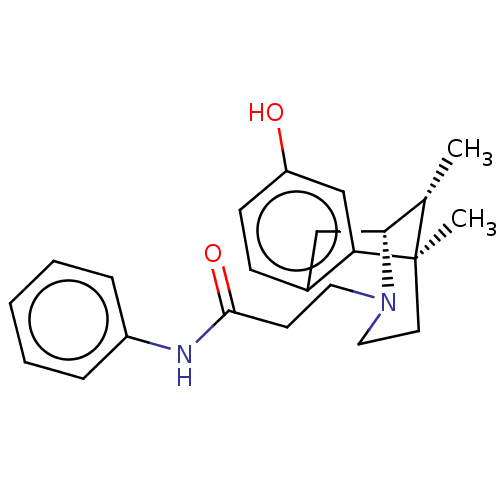
(CHEMBL3759092)Show SMILES [H][C@]12Cc3ccc(O)cc3[C@](C)(CCN1CCC(=O)Nc1ccccc1)[C@H]2C |r,THB:8:9:26:12.14.13,15:14:26:3.9.2| Show InChI InChI=1S/C23H28N2O2/c1-16-21-14-17-8-9-19(26)15-20(17)23(16,2)11-13-25(21)12-10-22(27)24-18-6-4-3-5-7-18/h3-9,15-16,21,26H,10-14H2,1-2H3,(H,24,27)/t16-,21+,23+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142703
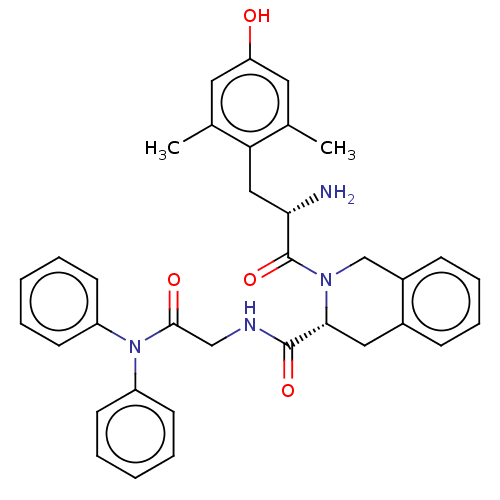
(CHEMBL3759541)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C35H36N4O4/c1-23-17-29(40)18-24(2)30(23)20-31(36)35(43)38-22-26-12-10-9-11-25(26)19-32(38)34(42)37-21-33(41)39(27-13-5-3-6-14-27)28-15-7-4-8-16-28/h3-18,31-32,40H,19-22,36H2,1-2H3,(H,37,42)/t31-,32+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from human delta opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142619

(CHEMBL3759383)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1CN[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H45N5O4/c1-25-17-32(45)18-26(2)33(25)22-34(40)39(48)44-24-30-16-10-9-15-29(30)21-31(44)23-42-36(20-28-13-7-4-8-14-28)38(47)43-35(37(41)46)19-27-11-5-3-6-12-27/h3-18,31,34-36,42,45H,19-24,40H2,1-2H3,(H2,41,46)(H,43,47)/t31-,34-,35-,36-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142693

(CHEMBL3759238)Show SMILES [H][C@@]12CCCC[C@@]11CCN(CC3CCCO3)[C@]2([H])C(=O)c2ccc(O)cc12 |r,TLB:25:26:1:7.9.8,19:18:1:7.9.8,THB:10:9:20.26.18:1,2:1:20.26.18:7.9.8| Show InChI InChI=1S/C21H27NO3/c23-14-6-7-16-18(12-14)21-8-2-1-5-17(21)19(20(16)24)22(10-9-21)13-15-4-3-11-25-15/h6-7,12,15,17,19,23H,1-5,8-11,13H2/t15?,17-,19-,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50001157
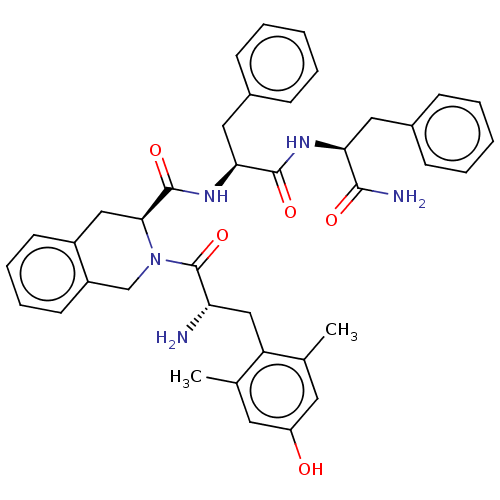
(CHEMBL538700)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C39H43N5O5/c1-24-17-30(45)18-25(2)31(24)22-32(40)39(49)44-23-29-16-10-9-15-28(29)21-35(44)38(48)43-34(20-27-13-7-4-8-14-27)37(47)42-33(36(41)46)19-26-11-5-3-6-12-26/h3-18,32-35,45H,19-23,40H2,1-2H3,(H2,41,46)(H,42,47)(H,43,48)/t32-,33-,34-,35-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50142696

(CHEMBL3758259)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@H]1CSCSC[C@@H](NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C38H42N6O6S2/c39-29(17-24-11-14-28(45)15-12-24)35(47)44-33-21-52-22-51-20-32(34(40)46)43-37(49)31(19-25-10-13-26-8-4-5-9-27(26)16-25)41-36(48)30(42-38(33)50)18-23-6-2-1-3-7-23/h1-16,29-33,45H,17-22,39H2,(H2,40,46)(H,41,48)(H,42,50)(H,43,49)(H,44,47)/t29-,30-,31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity to kappa opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50135808

((-)-3-Hydroxy-N-cyclobutylmethylmorphinan S(+)-Man...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 |r,TLB:16:15:4.21.5:7| Show InChI InChI=1S/C21H29NO/c23-17-8-7-16-12-20-18-6-1-2-9-21(18,19(16)13-17)10-11-22(20)14-15-4-3-5-15/h7-8,13,15,18,20,23H,1-6,9-12,14H2/t18-,20+,21+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142694

(CHEMBL3759660)Show SMILES [H]C12Cc3cc4sc(N)nc4cc3[C@]3(CCCC[C@@]13[H])CCN2CC1CC1 |r,TLB:23:22:3.12.2:18| Show InChI InChI=1S/C21H27N3S/c22-20-23-17-11-16-14(10-19(17)25-20)9-18-15-3-1-2-6-21(15,16)7-8-24(18)12-13-4-5-13/h10-11,13,15,18H,1-9,12H2,(H2,22,23)/t15-,18?,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]D-Ala2,N-Me-Phe4,Gly5-ol]-Enkephalin from human mu opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142703

(CHEMBL3759541)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C35H36N4O4/c1-23-17-29(40)18-24(2)30(23)20-31(36)35(43)38-22-26-12-10-9-11-25(26)19-32(38)34(42)37-21-33(41)39(27-13-5-3-6-14-27)28-15-7-4-8-16-28/h3-18,31-32,40H,19-22,36H2,1-2H3,(H,37,42)/t31-,32+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50299217

((5R,8S,11S,14S)-14-((S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H]1CSCSC[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C34H40N6O6S2/c35-25(15-23-11-13-24(41)14-12-23)31(43)40-29-19-48-20-47-18-28(30(36)42)39-33(45)27(17-22-9-5-2-6-10-22)37-32(44)26(38-34(29)46)16-21-7-3-1-4-8-21/h1-14,25-29,41H,15-20,35H2,(H2,36,42)(H,37,44)(H,38,46)(H,39,45)(H,40,43)/t25-,26-,27-,28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142690
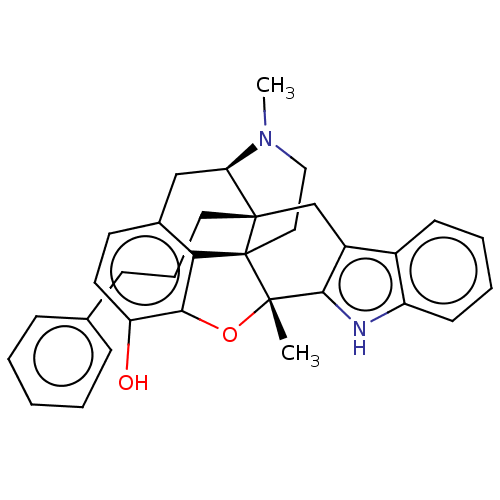
(CHEMBL3759489)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)c6[nH]c7ccccc7c6C[C@]1(CCCc1ccccc1)[C@@]5(CCN2C)c34 |r,TLB:36:35:3.37.2:22| Show InChI InChI=1S/C33H34N2O2/c1-31-30-24(23-12-6-7-13-25(23)34-30)20-32(16-8-11-21-9-4-3-5-10-21)27-19-22-14-15-26(36)29(37-31)28(22)33(31,32)17-18-35(27)2/h3-7,9-10,12-15,27,34,36H,8,11,16-20H2,1-2H3/t27-,31+,32+,33+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50299217

((5R,8S,11S,14S)-14-((S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H]1CSCSC[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C34H40N6O6S2/c35-25(15-23-11-13-24(41)14-12-23)31(43)40-29-19-48-20-47-18-28(30(36)42)39-33(45)27(17-22-9-5-2-6-10-22)37-32(44)26(38-34(29)46)16-21-7-3-1-4-8-21/h1-14,25-29,41H,15-20,35H2,(H2,36,42)(H,37,44)(H,38,46)(H,39,45)(H,40,43)/t25-,26-,27-,28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity to kappa opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50041144
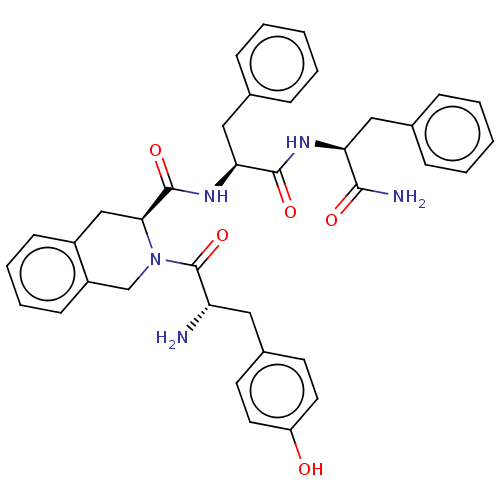
((S)-2-Amino-N-((5S,8S)-5-benzyl-4,7,13,16-tetraoxo...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C37H39N5O5/c38-30(19-26-15-17-29(43)18-16-26)37(47)42-23-28-14-8-7-13-27(28)22-33(42)36(46)41-32(21-25-11-5-2-6-12-25)35(45)40-31(34(39)44)20-24-9-3-1-4-10-24/h1-18,30-33,43H,19-23,38H2,(H2,39,44)(H,40,45)(H,41,46)/t30-,31-,32-,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from delta opioid receptor in rat brain membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142691
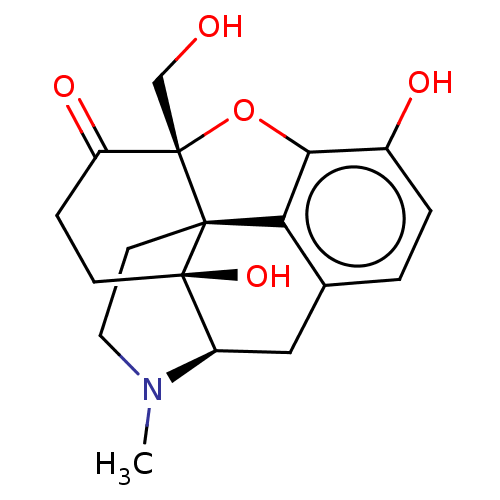
(CHEMBL3758348)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(CO)C(=O)CC[C@]1(O)[C@@]5(CCN2C)c34 |r,THB:23:22:3.24.2:17| Show InChI InChI=1S/C18H21NO5/c1-19-7-6-16-14-10-2-3-11(21)15(14)24-18(16,9-20)13(22)4-5-17(16,23)12(19)8-10/h2-3,12,20-21,23H,4-9H2,1H3/t12-,16-,17-,18+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142704

(CHEMBL3758563)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N1CCc2ccccc2C1 |r| Show InChI InChI=1S/C32H36N4O4/c1-20-13-26(37)14-21(2)27(20)16-28(33)32(40)36-19-25-10-6-4-8-23(25)15-29(36)31(39)34-17-30(38)35-12-11-22-7-3-5-9-24(22)18-35/h3-10,13-14,28-29,37H,11-12,15-19,33H2,1-2H3,(H,34,39)/t28-,29+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cell membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142699
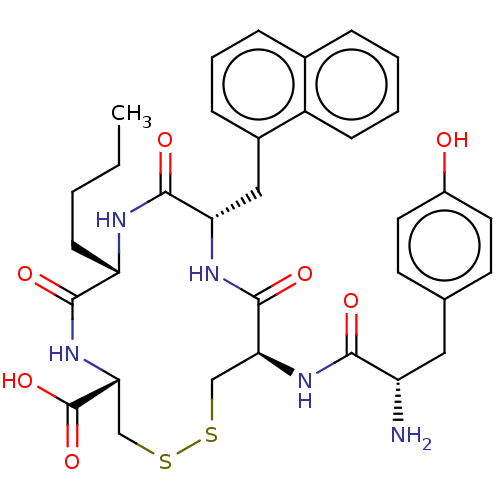
(CHEMBL3759831)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(O)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H41N5O7S2/c1-2-3-11-26-31(42)39-29(34(45)46)19-48-47-18-28(38-30(41)25(35)16-20-12-14-23(40)15-13-20)33(44)37-27(32(43)36-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,40H,2-3,11,16-19,35H2,1H3,(H,36,43)(H,37,44)(H,38,41)(H,39,42)(H,45,46)/t25-,26-,27-,28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat mu opioid receptor transfected in C6 cells by liquid scintillation counting assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142702
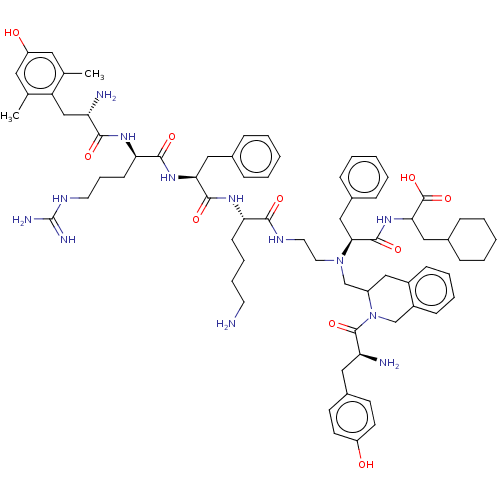
(CHEMBL3758231)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(=O)NCCN(CC1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1ccc(O)cc1)[C@@H](Cc1ccccc1)C(=O)NC(CC1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C71H97N13O10/c1-45-35-55(86)36-46(2)56(45)42-57(73)64(87)79-60(26-16-32-78-71(75)76)66(89)81-61(38-47-17-6-3-7-18-47)67(90)80-59(25-14-15-31-72)65(88)77-33-34-83(63(40-49-21-10-5-11-22-49)68(91)82-62(70(93)94)39-48-19-8-4-9-20-48)44-53-41-51-23-12-13-24-52(51)43-84(53)69(92)58(74)37-50-27-29-54(85)30-28-50/h3,5-7,10-13,17-18,21-24,27-30,35-36,48,53,57-63,85-86H,4,8-9,14-16,19-20,25-26,31-34,37-44,72-74H2,1-2H3,(H,77,88)(H,79,87)(H,80,90)(H,81,89)(H,82,91)(H,93,94)(H4,75,76,78)/t53?,57-,58-,59-,60+,61-,62?,63-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142690

(CHEMBL3759489)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)c6[nH]c7ccccc7c6C[C@]1(CCCc1ccccc1)[C@@]5(CCN2C)c34 |r,TLB:36:35:3.37.2:22| Show InChI InChI=1S/C33H34N2O2/c1-31-30-24(23-12-6-7-13-25(23)34-30)20-32(16-8-11-21-9-4-3-5-10-21)27-19-22-14-15-26(36)29(37-31)28(22)33(31,32)17-18-35(27)2/h3-7,9-10,12-15,27,34,36H,8,11,16-20H2,1-2H3/t27-,31+,32+,33+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142699

(CHEMBL3759831)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(O)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H41N5O7S2/c1-2-3-11-26-31(42)39-29(34(45)46)19-48-47-18-28(38-30(41)25(35)16-20-12-14-23(40)15-13-20)33(44)37-27(32(43)36-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,40H,2-3,11,16-19,35H2,1H3,(H,36,43)(H,37,44)(H,38,41)(H,39,42)(H,45,46)/t25-,26-,27-,28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat delta opioid receptor transfected in C6 cells by liquid scintillation counting assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142697

(CHEMBL3759294)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H42N6O6S2/c1-2-3-11-26-32(44)39-28(30(36)42)18-47-48-19-29(40-31(43)25(35)16-20-12-14-23(41)15-13-20)34(46)38-27(33(45)37-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,41H,2-3,11,16-19,35H2,1H3,(H2,36,42)(H,37,45)(H,38,46)(H,39,44)(H,40,43)/t25-,26-,27-,28+,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]diprenorphine from rat delta opioid receptor transfected in C6 cells by liquid scintillation counting assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142700

(CHEMBL326684)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)C(=O)CC[C@]1(OC)[C@@]5(CCN2C)c34 |r,TLB:23:22:16:2.3.24| Show InChI InChI=1S/C19H23NO4/c1-17-14(22)6-7-19(23-3)13-10-11-4-5-12(21)16(24-17)15(11)18(17,19)8-9-20(13)2/h4-5,13,21H,6-10H2,1-3H3/t13-,17+,18+,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142702

(CHEMBL3758231)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(=O)NCCN(CC1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1ccc(O)cc1)[C@@H](Cc1ccccc1)C(=O)NC(CC1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C71H97N13O10/c1-45-35-55(86)36-46(2)56(45)42-57(73)64(87)79-60(26-16-32-78-71(75)76)66(89)81-61(38-47-17-6-3-7-18-47)67(90)80-59(25-14-15-31-72)65(88)77-33-34-83(63(40-49-21-10-5-11-22-49)68(91)82-62(70(93)94)39-48-19-8-4-9-20-48)44-53-41-51-23-12-13-24-52(51)43-84(53)69(92)58(74)37-50-27-29-54(85)30-28-50/h3,5-7,10-13,17-18,21-24,27-30,35-36,48,53,57-63,85-86H,4,8-9,14-16,19-20,25-26,31-34,37-44,72-74H2,1-2H3,(H,77,88)(H,79,87)(H,80,90)(H,81,89)(H,82,91)(H,93,94)(H4,75,76,78)/t53?,57-,58-,59-,60+,61-,62?,63-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142621

(CHEMBL3759292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C30H34N4O4/c1-19-12-24(35)13-20(2)25(19)15-26(31)30(38)34-18-23-11-7-6-10-22(23)14-27(34)29(37)33-17-28(36)32-16-21-8-4-3-5-9-21/h3-13,26-27,35H,14-18,31H2,1-2H3,(H,32,36)(H,33,37)/t26-,27+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPN from human mu opioid receptor expressed in CHO cells by liquid scintillation counting |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50142700

(CHEMBL326684)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)C(=O)CC[C@]1(OC)[C@@]5(CCN2C)c34 |r,TLB:23:22:16:2.3.24| Show InChI InChI=1S/C19H23NO4/c1-17-14(22)6-7-19(23-3)13-10-11-4-5-12(21)16(24-17)15(11)18(17,19)8-9-20(13)2/h4-5,13,21H,6-10H2,1-3H3/t13-,17+,18+,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity to kappa opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142692

(CHEMBL3759092)Show SMILES [H][C@]12Cc3ccc(O)cc3[C@](C)(CCN1CCC(=O)Nc1ccccc1)[C@H]2C |r,THB:8:9:26:12.14.13,15:14:26:3.9.2| Show InChI InChI=1S/C23H28N2O2/c1-16-21-14-17-8-9-19(26)15-20(17)23(16,2)11-13-25(21)12-10-22(27)24-18-6-4-3-5-7-18/h3-9,15-16,21,26H,10-14H2,1-2H3,(H,24,27)/t16-,21+,23+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50041144

((S)-2-Amino-N-((5S,8S)-5-benzyl-4,7,13,16-tetraoxo...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r| Show InChI InChI=1S/C37H39N5O5/c38-30(19-26-15-17-29(43)18-16-26)37(47)42-23-28-14-8-7-13-27(28)22-33(42)36(46)41-32(21-25-11-5-2-6-12-25)35(45)40-31(34(39)44)20-24-9-3-1-4-10-24/h1-18,30-33,43H,19-23,38H2,(H2,39,44)(H,40,45)(H,41,46)/t30-,31-,32-,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAGO from mu opioid receptor in rat brain membrane |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142691

(CHEMBL3758348)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(CO)C(=O)CC[C@]1(O)[C@@]5(CCN2C)c34 |r,THB:23:22:3.24.2:17| Show InChI InChI=1S/C18H21NO5/c1-19-7-6-16-14-10-2-3-11(21)15(14)24-18(16,9-20)13(22)4-5-17(16,23)12(19)8-10/h2-3,12,20-21,23H,4-9H2,1H3/t12-,16-,17-,18+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 208 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPDPE from human delta opioid receptor expressed in CHO cells |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142705

(CHEMBL3758257)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1Cc2ccccc2CC1CN[C@@H](Cc1ccccc1)C(=O)NC(CC1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H46N4O5/c38-32(19-27-15-17-31(42)18-16-27)36(44)41-24-29-14-8-7-13-28(29)22-30(41)23-39-33(20-25-9-3-1-4-10-25)35(43)40-34(37(45)46)21-26-11-5-2-6-12-26/h1,3-4,7-10,13-18,26,30,32-34,39,42H,2,5-6,11-12,19-24,38H2,(H,40,43)(H,45,46)/t30?,32-,33-,34?/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50010483

(CHEMBL2181202)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(N)=O |r| Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50123658

(3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...)Show SMILES C[C@@H]1CN([C@@H](c2cccc(O)c2)c2cccc(c2)C(=O)N(C)c2cccc(F)c2)[C@@H](C)CN1CC=C Show InChI InChI=1S/C30H34FN3O2/c1-5-15-33-19-22(3)34(20-21(33)2)29(24-10-7-14-28(35)17-24)23-9-6-11-25(16-23)30(36)32(4)27-13-8-12-26(31)18-27/h5-14,16-18,21-22,29,35H,1,15,19-20H2,2-4H3/t21-,22+,29-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at mu-opioid receptor in guinea pig ileum assessed as inhibition of electrically induced twitches |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50010483

(CHEMBL2181202)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(N)=O |r| Show InChI InChI=1S/C32H49N9O5/c1-19-15-22(42)16-20(2)23(19)18-24(34)29(44)40-26(12-8-14-38-32(36)37)30(45)41-27(17-21-9-4-3-5-10-21)31(46)39-25(28(35)43)11-6-7-13-33/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,44)(H,41,45)(H4,36,37,38)/t24-,25-,26+,27-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at mu-opioid receptor in guinea pig ileum |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142704

(CHEMBL3758563)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N1CCc2ccccc2C1 |r| Show InChI InChI=1S/C32H36N4O4/c1-20-13-26(37)14-21(2)27(20)16-28(33)32(40)36-19-25-10-6-4-8-23(25)15-29(36)31(39)34-17-30(38)35-12-11-22-7-3-5-9-24(22)18-35/h3-10,13-14,28-29,37H,11-12,15-19,33H2,1-2H3,(H,34,39)/t28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Antagonist activity at human delta opioid receptor expressed in CHO cell membrane assessed as inhibition of DAMGO-induced [35S]-GTP-gammaS binding |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50123658

(3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...)Show SMILES C[C@@H]1CN([C@@H](c2cccc(O)c2)c2cccc(c2)C(=O)N(C)c2cccc(F)c2)[C@@H](C)CN1CC=C Show InChI InChI=1S/C30H34FN3O2/c1-5-15-33-19-22(3)34(20-21(33)2)29(24-10-7-14-28(35)17-24)23-9-6-11-25(16-23)30(36)32(4)27-13-8-12-26(31)18-27/h5-14,16-18,21-22,29,35H,1,15,19-20H2,2-4H3/t21-,22+,29-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Antagonist activity at human delta opioid receptor in CD1 mouse assessed as electrically induced twitches |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142703

(CHEMBL3759541)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C35H36N4O4/c1-23-17-29(40)18-24(2)30(23)20-31(36)35(43)38-22-26-12-10-9-11-25(26)19-32(38)34(42)37-21-33(41)39(27-13-5-3-6-14-27)28-15-7-4-8-16-28/h3-18,31-32,40H,19-22,36H2,1-2H3,(H,37,42)/t31-,32+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Antagonist activity at human delta opioid receptor expressed in CHO cell membrane assessed as inhibition of DAMGO-induced [35S]-GTP-gammaS binding |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142701
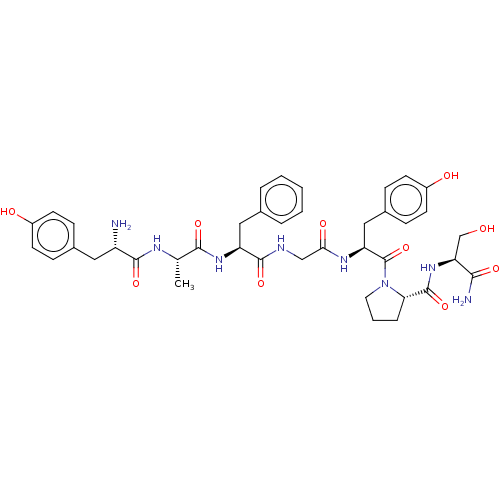
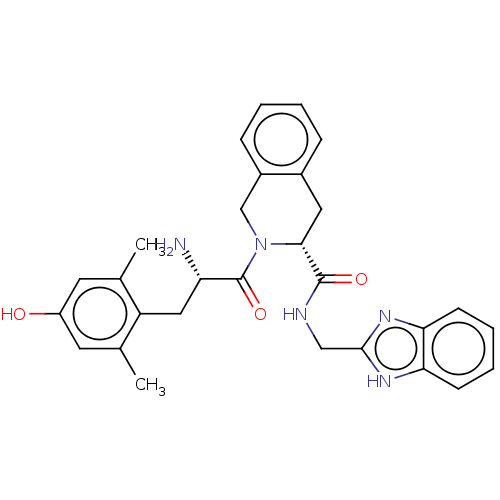
(CHEMBL285479)Show SMILES C[C@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(N)=O Show InChI InChI=1S/C40H50N8O10/c1-23(44-37(55)29(41)18-25-9-13-27(50)14-10-25)36(54)46-30(19-24-6-3-2-4-7-24)38(56)43-21-34(52)45-31(20-26-11-15-28(51)16-12-26)40(58)48-17-5-8-33(48)39(57)47-32(22-49)35(42)53/h2-4,6-7,9-16,23,29-33,49-51H,5,8,17-22,41H2,1H3,(H2,42,53)(H,43,56)(H,44,55)(H,45,52)(H,46,54)(H,47,57)/t23-,29-,30-,31-,32-,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at mu opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50142702

(CHEMBL3758231)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCCN)C(=O)NCCN(CC1Cc2ccccc2CN1C(=O)[C@@H](N)Cc1ccc(O)cc1)[C@@H](Cc1ccccc1)C(=O)NC(CC1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C71H97N13O10/c1-45-35-55(86)36-46(2)56(45)42-57(73)64(87)79-60(26-16-32-78-71(75)76)66(89)81-61(38-47-17-6-3-7-18-47)67(90)80-59(25-14-15-31-72)65(88)77-33-34-83(63(40-49-21-10-5-11-22-49)68(91)82-62(70(93)94)39-48-19-8-4-9-20-48)44-53-41-51-23-12-13-24-52(51)43-84(53)69(92)58(74)37-50-27-29-54(85)30-28-50/h3,5-7,10-13,17-18,21-24,27-30,35-36,48,53,57-63,85-86H,4,8-9,14-16,19-20,25-26,31-34,37-44,72-74H2,1-2H3,(H,77,88)(H,79,87)(H,80,90)(H,81,89)(H,82,91)(H,93,94)(H4,75,76,78)/t53?,57-,58-,59-,60+,61-,62?,63-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at mu-opioid receptor in guinea pig ileum |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142701

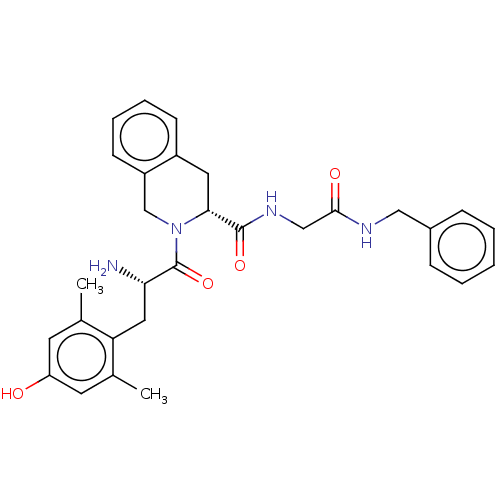
(CHEMBL285479)Show SMILES C[C@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(N)=O Show InChI InChI=1S/C40H50N8O10/c1-23(44-37(55)29(41)18-25-9-13-27(50)14-10-25)36(54)46-30(19-24-6-3-2-4-7-24)38(56)43-21-34(52)45-31(20-26-11-15-28(51)16-12-26)40(58)48-17-5-8-33(48)39(57)47-32(22-49)35(42)53/h2-4,6-7,9-16,23,29-33,49-51H,5,8,17-22,41H2,1H3,(H2,42,53)(H,43,56)(H,44,55)(H,45,52)(H,46,54)(H,47,57)/t23-,29-,30-,31-,32-,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Binding affinity at delta opioid receptor (unknown origin) |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142696

(CHEMBL3758259)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@H]1CSCSC[C@@H](NC(=O)[C@H](Cc2ccc3ccccc3c2)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(N)=O |r| Show InChI InChI=1S/C38H42N6O6S2/c39-29(17-24-11-14-28(45)15-12-24)35(47)44-33-21-52-22-51-20-32(34(40)46)43-37(49)31(19-25-10-13-26-8-4-5-9-27(26)16-25)41-36(48)30(42-38(33)50)18-23-6-2-1-3-7-23/h1-16,29-33,45H,17-22,39H2,(H2,40,46)(H,41,48)(H,42,50)(H,43,49)(H,44,47)/t29-,30-,31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 36 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Partial agonist activity at delta opioid receptor (unknown origin) assessed as inhibition of adenylyl cyclase activity |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142697

(CHEMBL3759294)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H42N6O6S2/c1-2-3-11-26-32(44)39-28(30(36)42)18-47-48-19-29(40-31(43)25(35)16-20-12-14-23(41)15-13-20)34(46)38-27(33(45)37-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,41H,2-3,11,16-19,35H2,1H3,(H2,36,42)(H,37,45)(H,38,46)(H,39,44)(H,40,43)/t25-,26-,27-,28+,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.10 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at rat mu opioid receptor transfected in C6 cells by [35S]GTPgammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50142621

(CHEMBL3759292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C30H34N4O4/c1-19-12-24(35)13-20(2)25(19)15-26(31)30(38)34-18-23-11-7-6-10-22(23)14-27(34)29(37)33-17-28(36)32-16-21-8-4-3-5-9-21/h3-13,26-27,35H,14-18,31H2,1-2H3,(H,32,36)(H,33,37)/t26-,27+/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 32 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at mu-opioid receptor in guinea pig ileum assessed as inhibition of electrically induced twitches |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50135806

(CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C52H72N2O4/c55-49(57-41-23-21-39-31-47-43-17-7-9-25-51(43,45(39)33-41)27-29-53(47)35-37-13-11-14-37)19-5-3-1-2-4-6-20-50(56)58-42-24-22-40-32-48-44-18-8-10-26-52(44,46(40)34-42)28-30-54(48)36-38-15-12-16-38/h21-24,33-34,37-38,43-44,47-48H,1-20,25-32,35-36H2/t43-,44-,47+,48+,51+,52+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Partial agonist activity at human mu opioid receptor expressed in CHO cells by [35S]GTPgammaS binding |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142691

(CHEMBL3758348)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(CO)C(=O)CC[C@]1(O)[C@@]5(CCN2C)c34 |r,THB:23:22:3.24.2:17| Show InChI InChI=1S/C18H21NO5/c1-19-7-6-16-14-10-2-3-11(21)15(14)24-18(16,9-20)13(22)4-5-17(16,23)12(19)8-10/h2-3,12,20-21,23H,4-9H2,1H3/t12-,16-,17-,18+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cells by [35S]GTP-gammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50142622

(CHEMBL3758292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCc1nc2ccccc2[nH]1 |r| Show InChI InChI=1S/C29H31N5O3/c1-17-11-21(35)12-18(2)22(17)14-23(30)29(37)34-16-20-8-4-3-7-19(20)13-26(34)28(36)31-15-27-32-24-9-5-6-10-25(24)33-27/h3-12,23,26,35H,13-16,30H2,1-2H3,(H,31,36)(H,32,33)/t23-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at delta-opioid receptor in mouse vas deferens |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142694

(CHEMBL3759660)Show SMILES [H]C12Cc3cc4sc(N)nc4cc3[C@]3(CCCC[C@@]13[H])CCN2CC1CC1 |r,TLB:23:22:3.12.2:18| Show InChI InChI=1S/C21H27N3S/c22-20-23-17-11-16-14(10-19(17)25-20)9-18-15-3-1-2-6-21(15,16)7-8-24(18)12-13-4-5-13/h10-11,13,15,18H,1-9,12H2,(H2,22,23)/t15-,18?,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 73 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Partial agonist activity at mu opioid receptor (unknown origin) expressed in CHO cell membrane assessed as stimulation of [35S]GTP-gamma-S binding |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50142621

(CHEMBL3759292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C30H34N4O4/c1-19-12-24(35)13-20(2)25(19)15-26(31)30(38)34-18-23-11-7-6-10-22(23)14-27(34)29(37)33-17-28(36)32-16-21-8-4-3-5-9-21/h3-13,26-27,35H,14-18,31H2,1-2H3,(H,32,36)(H,33,37)/t26-,27+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at delta-opioid receptor in mouse vas deferens |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142697

(CHEMBL3759294)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(N)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H42N6O6S2/c1-2-3-11-26-32(44)39-28(30(36)42)18-47-48-19-29(40-31(43)25(35)16-20-12-14-23(41)15-13-20)34(46)38-27(33(45)37-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,41H,2-3,11,16-19,35H2,1H3,(H2,36,42)(H,37,45)(H,38,46)(H,39,44)(H,40,43)/t25-,26-,27-,28+,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 22 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at rat delta opioid receptor transfected in C6 cells by [35S]GTPgammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142703

(CHEMBL3759541)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C35H36N4O4/c1-23-17-29(40)18-24(2)30(23)20-31(36)35(43)38-22-26-12-10-9-11-25(26)19-32(38)34(42)37-21-33(41)39(27-13-5-3-6-14-27)28-15-7-4-8-16-28/h3-18,31-32,40H,19-22,36H2,1-2H3,(H,37,42)/t31-,32+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cells by [35S]GTP-gammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142622

(CHEMBL3758292)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCc1nc2ccccc2[nH]1 |r| Show InChI InChI=1S/C29H31N5O3/c1-17-11-21(35)12-18(2)22(17)14-23(30)29(37)34-16-20-8-4-3-7-19(20)13-26(34)28(36)31-15-27-32-24-9-5-6-10-25(24)33-27/h3-12,23,26,35H,13-16,30H2,1-2H3,(H,31,36)(H,32,33)/t23-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.0813 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at human delta-opioid receptor expressed in CHO cells by [35S]GTPgammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135797

(CHEMBL146756 | di[17-cyclobutylmethyl-(1R,9R,10R)-...)Show SMILES O=C(Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)\C=C\C(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C46H58N2O4/c49-43(51-35-15-13-33-25-41-37-11-1-3-19-45(37,39(33)27-35)21-23-47(41)29-31-7-5-8-31)17-18-44(50)52-36-16-14-34-26-42-38-12-2-4-20-46(38,40(34)28-36)22-24-48(42)30-32-9-6-10-32/h13-18,27-28,31-32,37-38,41-42H,1-12,19-26,29-30H2/b18-17+/t37-,38-,41+,42+,45+,46+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.30 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142699

(CHEMBL3759831)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(O)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H41N5O7S2/c1-2-3-11-26-31(42)39-29(34(45)46)19-48-47-18-28(38-30(41)25(35)16-20-12-14-23(40)15-13-20)33(44)37-27(32(43)36-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,40H,2-3,11,16-19,35H2,1H3,(H,36,43)(H,37,44)(H,38,41)(H,39,42)(H,45,46)/t25-,26-,27-,28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.10 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at rat mu opioid receptor transfected in C6 cells by [35S]GTPgammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50142699

(CHEMBL3759831)Show SMILES CCCC[C@@H]1NC(=O)[C@H](Cc2cccc3ccccc23)NC(=O)[C@H](CSSC[C@@H](NC1=O)C(O)=O)NC(=O)[C@@H](N)Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C34H41N5O7S2/c1-2-3-11-26-31(42)39-29(34(45)46)19-48-47-18-28(38-30(41)25(35)16-20-12-14-23(40)15-13-20)33(44)37-27(32(43)36-26)17-22-9-6-8-21-7-4-5-10-24(21)22/h4-10,12-15,25-29,40H,2-3,11,16-19,35H2,1H3,(H,36,43)(H,37,44)(H,38,41)(H,39,42)(H,45,46)/t25-,26-,27-,28-,29+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 22 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at rat delta opioid receptor transfected in C6 cells by [35S]GTPgammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50135806

(CHEMBL147511 | MCL-144 | di[17-cyclobutylmethyl-(1...)Show SMILES O=C(CCCCCCCCC(=O)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1)Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CCC3)c2c1 Show InChI InChI=1S/C52H72N2O4/c55-49(57-41-23-21-39-31-47-43-17-7-9-25-51(43,45(39)33-41)27-29-53(47)35-37-13-11-14-37)19-5-3-1-2-4-6-20-50(56)58-42-24-22-40-32-48-44-18-8-10-26-52(44,46(40)34-42)28-30-54(48)36-38-15-12-16-38/h21-24,33-34,37-38,43-44,47-48H,1-20,25-32,35-36H2/t43-,44-,47+,48+,51+,52+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.850 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142704

(CHEMBL3758563)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)N1CCc2ccccc2C1 |r| Show InChI InChI=1S/C32H36N4O4/c1-20-13-26(37)14-21(2)27(20)16-28(33)32(40)36-19-25-10-6-4-8-23(25)15-29(36)31(39)34-17-30(38)35-12-11-22-7-3-5-9-24(22)18-35/h3-10,13-14,28-29,37H,11-12,15-19,33H2,1-2H3,(H,34,39)/t28-,29+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 22 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cells by [35S]GTP-gammaS binding assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50142620

(CHEMBL3758712)Show SMILES Cc1cc(O)cc(C)c1C[C@H](N)C(=O)N1Cc2ccccc2C[C@@H]1C(=O)NCC(=O)Nc1ccccc1 |r| Show InChI InChI=1S/C29H32N4O4/c1-18-12-23(34)13-19(2)24(18)15-25(30)29(37)33-17-21-9-7-6-8-20(21)14-26(33)28(36)31-16-27(35)32-22-10-4-3-5-11-22/h3-13,25-26,34H,14-17,30H2,1-2H3,(H,31,36)(H,32,35)/t25-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at delta-opioid receptor in mouse vas deferens |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50142690

(CHEMBL3759489)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)c6[nH]c7ccccc7c6C[C@]1(CCCc1ccccc1)[C@@]5(CCN2C)c34 |r,TLB:36:35:3.37.2:22| Show InChI InChI=1S/C33H34N2O2/c1-31-30-24(23-12-6-7-13-25(23)34-30)20-32(16-8-11-21-9-4-3-5-10-21)27-19-22-14-15-26(36)29(37-31)28(22)33(31,32)17-18-35(27)2/h3-7,9-10,12-15,27,34,36H,8,11,16-20H2,1-2H3/t27-,31+,32+,33+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.870 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Partial agonist activity at delta opioid receptor (unknown origin) assessed as [35S]GTPgammaS binding by cell based assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50142690

(CHEMBL3759489)Show SMILES [H][C@@]12Cc3ccc(O)c4O[C@@]5(C)c6[nH]c7ccccc7c6C[C@]1(CCCc1ccccc1)[C@@]5(CCN2C)c34 |r,TLB:36:35:3.37.2:22| Show InChI InChI=1S/C33H34N2O2/c1-31-30-24(23-12-6-7-13-25(23)34-30)20-32(16-8-11-21-9-4-3-5-10-21)27-19-22-14-15-26(36)29(37-31)28(22)33(31,32)17-18-35(27)2/h3-7,9-10,12-15,27,34,36H,8,11,16-20H2,1-2H3/t27-,31+,32+,33+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 45 | n/a | n/a | n/a | n/a |
University of Catania
Curated by ChEMBL
| Assay Description
Agonist activity at mu opioid receptor (unknown origin) assessed as [35S]GTPgammaS binding by cell based assay |
Eur J Med Chem 108: 211-28 (2016)
Article DOI: 10.1016/j.ejmech.2015.11.028
BindingDB Entry DOI: 10.7270/Q2R2137W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data