

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Monoglyceride lipase (Rattus norvegicus (Rat)) | BDBM50160284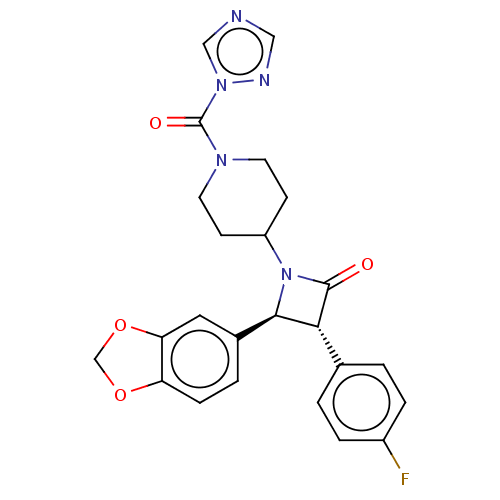 (CHEMBL3785379) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of rat MAGL | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160282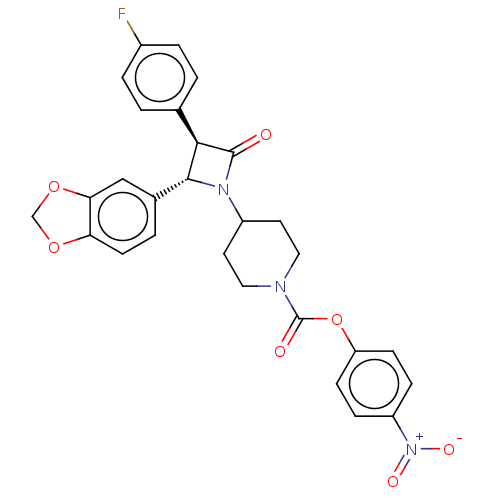 (CHEMBL3785760) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Rattus norvegicus (Rat)) | BDBM50160284 (CHEMBL3785379) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of MAGL in rat brain membranes preincubated for 20 mins followed by fluorophosphonate-rhodamine addition measured after 30 mins by competi... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160280 (CHEMBL3787340) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160277 (CHEMBL3785536) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160283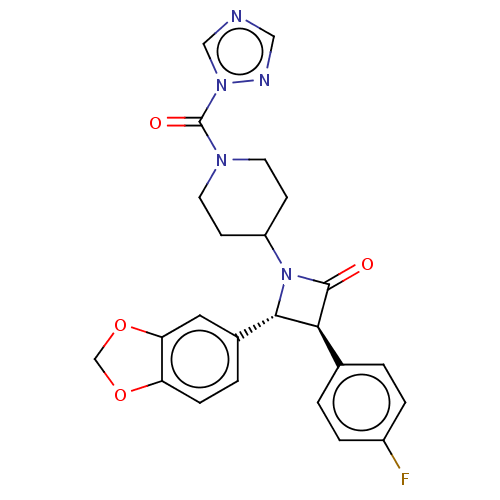 (CHEMBL3787346) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160279 (CHEMBL3787224) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 61 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160281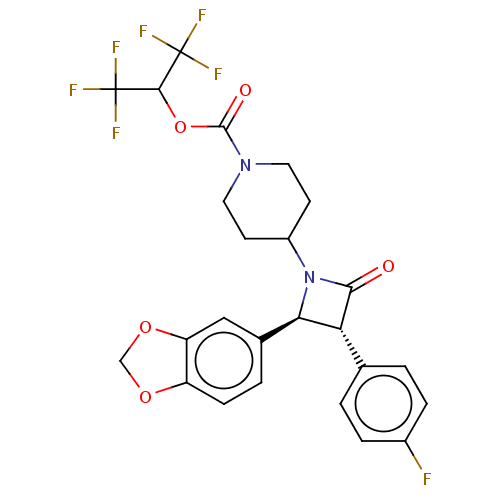 (CHEMBL3787592) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 98 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of human MAGL expressed in African green monkey COS cells using 2-arachidonoyl-[3H]-glycerol as substrate incubated for 20 mins by scintil... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 195 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate preincubate... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 196 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate preincubate... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160278 (CHEMBL3786142) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 251 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM60622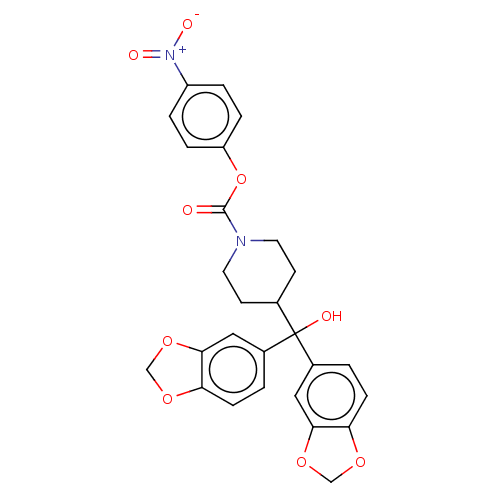 (BDBM50300355 | US11753371, Compound JZL-184 | US91...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 285 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160287 (CHEMBL3787453) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 376 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 [30-579] (Rattus norvegicus (rat)) | BDBM50160279 (CHEMBL3787224) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 725 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of FAAH in rat brain membranes using using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as reduction in [14C]ethanolamine produ... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160283 (CHEMBL3787346) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 729 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate preincubate... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 [30-579] (Rattus norvegicus (rat)) | BDBM50160280 (CHEMBL3787340) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of FAAH in rat brain membranes using using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as reduction in [14C]ethanolamine produ... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 [30-579] (Rattus norvegicus (rat)) | BDBM50160277 (CHEMBL3785536) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of FAAH in rat brain membranes using using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as reduction in [14C]ethanolamine produ... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His-tagged human FAAH expressed in sf21 cells using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as r... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His-tagged human FAAH expressed in sf21 cells using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as r... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM60622 (BDBM50300355 | US11753371, Compound JZL-184 | US91...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.44E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate preincubate... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50160283 (CHEMBL3787346) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7.74E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His-tagged human FAAH expressed in sf21 cells using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as r... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Mus musculus (mouse)) | BDBM60622 (BDBM50300355 | US11753371, Compound JZL-184 | US91...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of FAAH in mouse brain membrane using [13C]oleamide as substrate by LC-MS analysis | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160285 (CHEMBL3786786) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160290 (CHEMBL3786806) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Displacement of [3H]-CP-55940 from human recombinant CB1 receptor expressed in HEK293 cells after 90 mins | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160286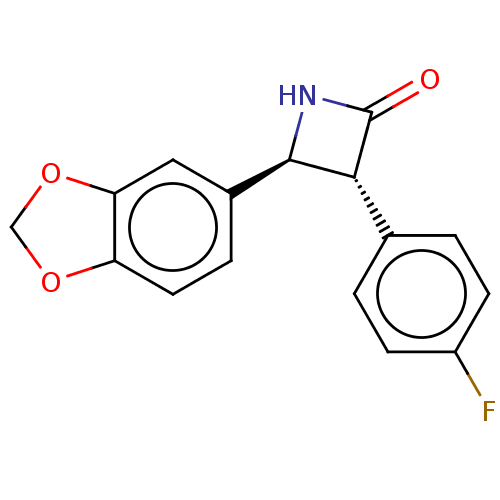 (CHEMBL3787456) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160288 (CHEMBL3787437) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 [30-579] (Rattus norvegicus (rat)) | BDBM50160278 (CHEMBL3786142) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of FAAH in rat brain membranes using using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as reduction in [14C]ethanolamine produ... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM50160289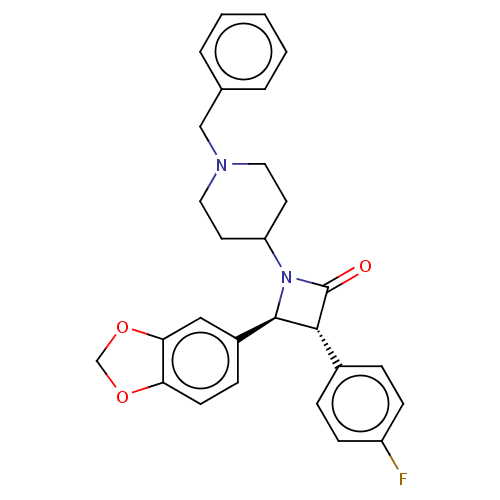 (CHEMBL3785148) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His6-tagged human MAGL expressed in Escherichia coli using 2-arachidonoyl-[3H]-glycerol as substrate incubated f... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (Homo sapiens (Human)) | BDBM50160282 (CHEMBL3785760) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of recombinant C-terminal His-tagged human FAAH expressed in sf21 cells using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as r... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM50160284 (CHEMBL3785379) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Displacement of [3H]-CP-55940 from human recombinant CB2 receptor expressed in HEK293 cells after 90 mins | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 [30-579] (Rattus norvegicus (rat)) | BDBM50160281 (CHEMBL3787592) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena Curated by ChEMBL | Assay Description Inhibition of FAAH in rat brain membranes using using N-arachidonoyl-[14C]-ethanolamine as substrate assessed as reduction in [14C]ethanolamine produ... | J Med Chem 59: 2612-32 (2016) Article DOI: 10.1021/acs.jmedchem.5b01812 BindingDB Entry DOI: 10.7270/Q21R6SC7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||